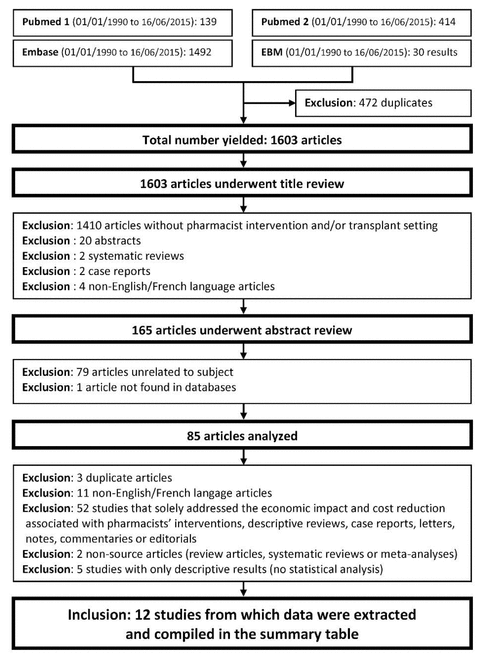
Figure 1 Selection of studies for a systematic review of the roles and impacts of transplant pharmacists.
Sébastien Sam, Aurélie Guérin, André Rieutord, Stéphanie Belaiche, Jean-François BussièresABSTRACT
Background
Pharmacists have been involved in the care of transplant recipients for several decades, and a growing body of literature shows the beneficial effects of clinical pharmacist care on important outcomes for these patients.
Objectives
The primary objective was to describe the roles and impacts of pharmacists in a solid organ transplant setting. The secondary objective was to describe and rate the pharmacists’ interventions.
Data Sources
Three databases—PubMed, Embase, and Evidence-Based Medicine Reviews—were searched from January 1, 1990, to June 16, 2015.
Study Selection and Data Extraction
All studies addressing the roles of pharmacists and the impacts of clinical pharmacy services on the care of solid organ transplant recipients were considered. Only studies providing a statistical analysis were included. Design, setting, sample size, patient characteristics, pharmacists’ interventions, study bias, and outcomes were extracted for analysis.
Data Synthesis
Four randomized controlled trials, 4 cohort studies, 3 pre–post studies, and 1 quasi-randomized controlled trial were included in the review, representing a total of 1837 patients. Of the 12 studies included, 8 specifically focused on renal transplant, and 1 each focused on liver, lung, abdominal organ, and general solid organ transplant. The pivotal pharmacist activities leading to the main patient outcomes were medication counselling (n = 8 studies), medication reconciliation (n = 5), and reviewing and optimizing drug therapy (n = 3). Improvements to medication adherence (n = 6 studies), morbidity (n = 4), costs (n = 2), and medication errors (n = 2) were reported.
Conclusion
Currently available evidence suggests that pharmacists can improve patient outcomes in the solid organ transplant setting. Adherence, morbidity, costs, and medication errors were identified as the main outcomes that were improved by pharmaceutical interventions. Transplant programs need to invest more in this resource.
KEYWORDS: pharmacist, organ transplantation, impact, clinical pharmacy, outcome-based research
RÉSUMÉ
Contexte
Les pharmaciens participent aux soins des greffés depuis plusieurs décennies et un nombre croissant de publications révèlent les effets bénéfiques des soins prodigués par les pharmaciens cliniciens quant aux résultats thérapeutiques importants pour ces patients.
Objectifs
L’objectif principal était de décrire les rôles des pharmaciens et leurs influences par rapport aux greffes d’organes solides. L’objectif secondaire était de décrire et d’évaluer les interventions des pharmaciens.
Sources des données
Les bases de données PubMed, Embase et Evidence-Based Medicine Reviews ont été interrogées pour la période allant du 1cr janvier 1990 au 16 juin 2015.
Sélection des études et extraction des données
Toutes les études abordant les rôles des pharmaciens et l’influence des services de pharmacie clinique sur les soins des receveurs d’organes solides ont été prises en considération. Seules les études présentant des analyses statistiques ont été retenues. Le plan d’étude, le contexte, la taille de l’échantillon, les caractéristiques des patients, les interventions des pharmaciens, les biais et les résultats thérapeutiques ont servi à l’analyse.
Synthèse des données
Quatre études contrôlées à répartition aléatoire, 4 études de cohorte, 3 études avant-après et 1 essai comparatif à répartition quasi-aléatoire ont été retenus pour l’analyse, ce qui représentait au total 1837 patients. Parmi les 12 études retenues, 8 abordaient spécifiquement la greffe rénale et chacune des 4 autres concernait respectivement une greffe hépatique, une greffe pulmonaire, une greffe d’organe abdominal et une greffe d’organe solide. Les activités clés des pharmaciens menant aux principaux résultats thérapeutiques étaient les conseils sur les médicaments (n = 8 études), l’établissement du bilan comparatif des médicaments (n = 5) ainsi que l’examen et l’optimisation de la pharmacothérapie (n = 3). On a constaté des améliorations des taux d’observance pharmacothérapeutique (n = 6 études), des taux de morbidité (n = 4), des coûts (n = 2) et des taux d’erreurs de médicaments (n = 2).
Conclusion
Les données probantes disponibles laissent croire que les pharmaciens peuvent améliorer les résultats thérapeutiques en ce qui concerne les greffes d’organes solides. Les taux d’observance pharmacothérapeutique, les taux de morbidité, les coûts et les taux d’erreurs de médicaments ont été désignés comme les résultats principaux qui ont été améliorés par les interventions pharmaceutiques. Les programmes de greffe doivent investir davantage dans cette ressource.
MOTS CLÉS: pharmacien, greffe d’organe, effet, pharmacie clinique, recherche axée sur les résultats
Solid organ transplant has been one of the most important-therapeutic advances in medicine over the past 60 years. Since the first transplants were performed, it has become the recommended therapeutic approach for many end-stage chronic diseases. In Canada, 2835 transplant procedures were done in 2016.1
Patients who have received a solid organ transplant require lifelong immunosuppressive treatments. Nonadherence to post-transplant drug therapy and recommendations is a major issue that can lead to misdiagnosis of subsequent health problems, poor health affecting quality of life, graft rejection, or death.2,3
Pharmacists have been involved in direct patient care since the early 1970s. The first report outlining specific activities of a dedicated transplant pharmacist was published in 1976.4 This article introduced the transplant pharmacist as an individual with specific expertise in transplantation pharmacology who actively participated in the medical management of organ transplant recipients and provided direct patient medication counselling. Since that time, the overall pharmacy practice model has evolved from a product-oriented to a patient-oriented model, and there have been advances in the field of transplant pharmacy as well. In the United States, for example, a “pharmacology expert” is now mandatory in transplant centres.5
A growing body of literature has shown the beneficial effects of clinical pharmacist care on important outcomes for both hospitalized and ambulatory patients; however, in the context of solid organ transplant, the majority of published studies have focused on renal transplant recipients.
There is high heterogeneity among the interventions described in studies evaluating the impact of clinical pharmacy services. Several authors have characterized the descriptions of interventions in pharmacy practice studies as inconsistent or even poor.6,7 Authors have therefore recommended that interventions be clearly reported, with a detailed explanation of the intervention, a description of the pharmacist–patient and pharmacist–provider relationships, and details about the setting where the study took place.8 A more comprehensive understanding of clinical pharmacy interventions for transplant patients would help in achieving better outcomes.
The primary objective of this systematic review was to describe the roles and impacts of pharmacists in a solid organ transplant setting. The secondary objectives were to describe and rate pharmacists’ interventions.
All specifications of the PRISMA 2009 checklist9 were followed for reporting this systematic review.
Four systematic searches were carried out in 3 databases (PubMed, Embase, and Evidence-Based Medicine Reviews) for articles published between January 1, 1990, and June 16, 2015. Manual reference checks were performed to search for potentially missing studies. Search strategies are presented in Appendix 1 (available at https://www.cjhp-online.ca/index.php/cjhp/issue/view/186/showToc).
All studies addressing the impact of clinical pharmacy services on the care of patients with solid organ transplant were considered. Studies providing a statistical analysis on the impact of pharmaceutical activities were included. Studies that presented only descriptive results, studies addressing only the economic impact of transplant services, descriptive reviews, case reports, journal letters, journal notes, commentaries, and editorials were all excluded. Also excluded were secondary sources such as literature reviews, systematic reviews, and meta-analyses. Articles in either English or French were included.
All references were screened by 2 independents reviewers (A.G., J.F.B.). If there were any discrepancies in the decision to include or exclude studies, a third researcher was consulted (S.B.). Study selection was accomplished through 3 phases of screening. During the first phase, titles were reviewed for relevance. During the second phase, abstracts from articles retained in the first phase were reviewed for relevance. In the third and final phase, the full texts of articles retained in the second phase were reviewed.
Data extraction was performed by 2 authors (A.G., S.S.), under the supervision of 1 reviewer (J.F.B.). Data from the included studies were synthesized into summary tables.
The DEPICT tool10 was used to evaluate the description of pharmaceutical interventions. Rating was performed by 2 authors (A.G., S.S.), under the supervision of 1 reviewer (J.F.B.), and a DEPICT score was assigned to each study. The DEPICT score evaluates studies according to 12 sections, with multiple items per section. For each section, a score of 1 is assigned if the reviewers answer “yes” for at least 1 item within the section; otherwise, a score of 0 is assigned for that section. The DEPICT score is determined by summing the number of sections with a score of 1 (maximum score = 12).
Individual study limitations, including risk of bias, were reported as described by the authors of each included article. The risk of bias across studies was assessed informally by the authors of the current systematic review.
The search yielded 1603 articles. Of these, 1518 were excluded after review of titles and abstracts. Of the 85 potentially eligible studies, 73 were excluded after review of the full-text articles. Twelve studies involving a total of 1837 patients were included in the analysis (Figure 1).11–22 Manual searching of the reference lists of these included articles yielded no additional eligible articles.
|
|
||
|
Figure 1 Selection of studies for a systematic review of the roles and impacts of transplant pharmacists. |
||
Eight studies focused on kidney transplant, one on liver transplant, one on lung transplant, one on abdominal transplant, and one on general solid organ transplant. The studies were conducted in the United States (n = 8 studies), Canada (n = 2), and Germany (n = 2). No differences were observed in terms of pharmacist roles or patient outcomes in relation to the geographic location of the studies.
The study characteristics are presented in Table 1 and the outcomes of individual studies in Table 2.
Table 2 Outcomes of Individual Studies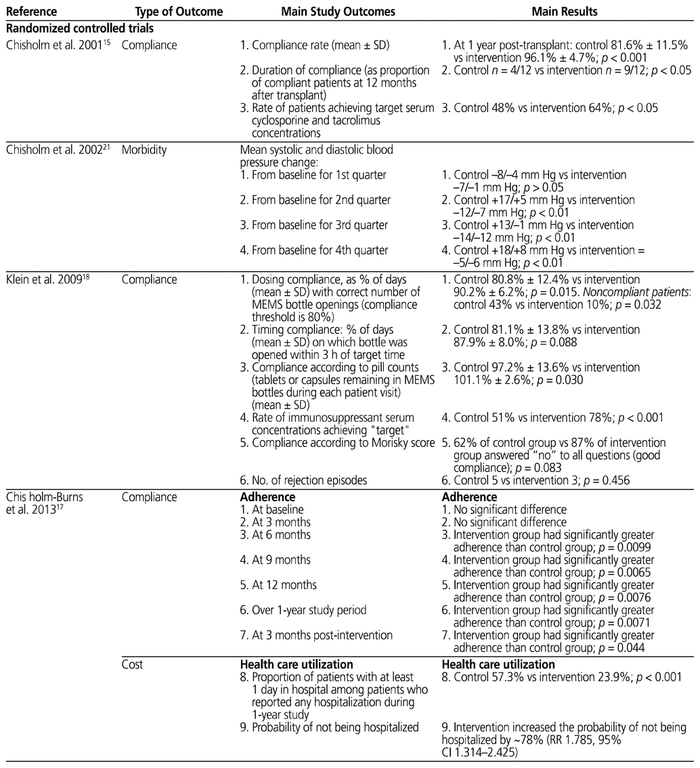
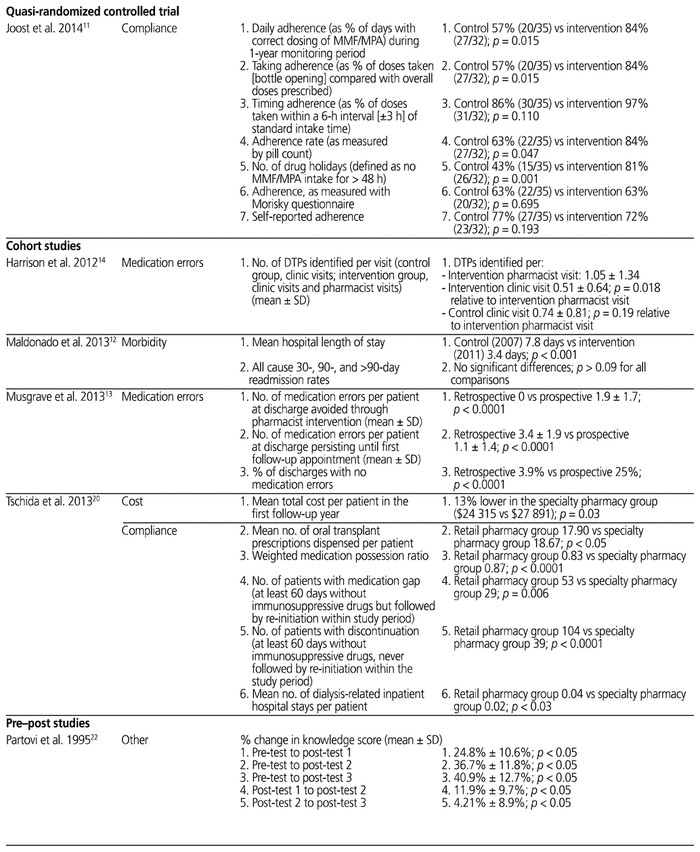
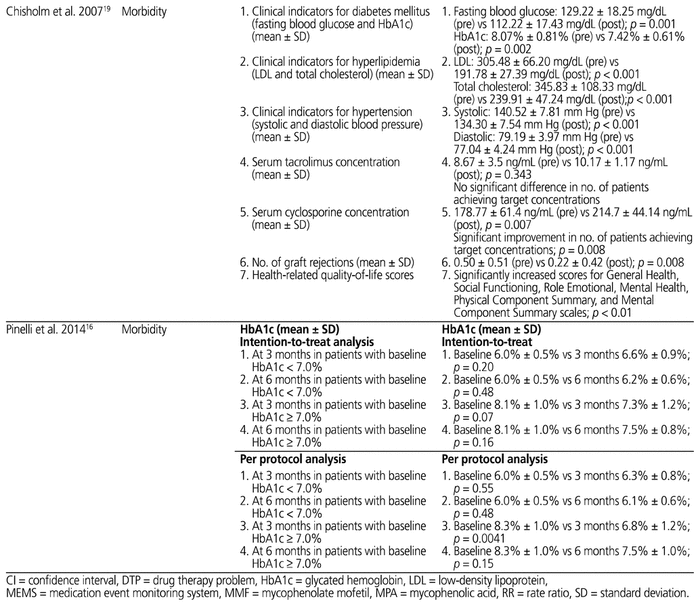
The pivotal pharmacist activities in the setting of solid organ transplant included patient education and counselling (n = 9 studies), reviewing and optimizing drug therapy (n = 7), and medication reconciliation or medical history (n = 5). Improvements were reported in the following areas: medication adherence (n = 6 studies), morbidity (n = 4), cost (n = 2), and medication errors (n = 2).
Pharmaceutical interventions were sufficiently described to understand the role of pharmacists. The average DEPICT score was 8.4 (standard deviation 1.4, minimum 6, maximum 11) (Table 3). The pharmaceutical interventions that were less frequently reported included the timing of the intervention, the support resources provided by pharmacists, and the pharmacist’s autonomy to perform some specific tasks.
Table 3 Rating of Pharmaceutical Interventions with DEPICT Tool10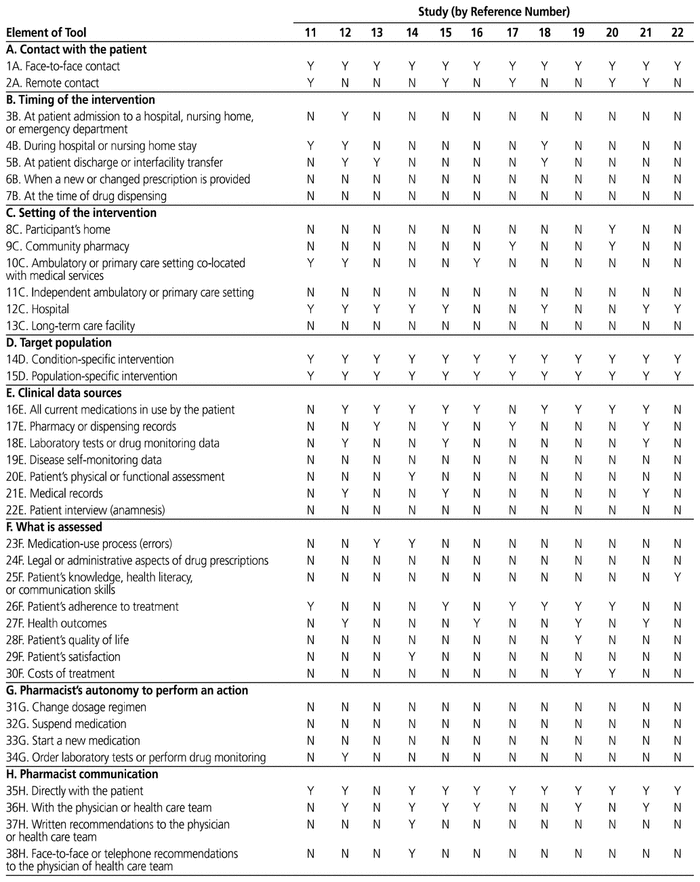
Risk of bias is reported here as described by the authors of each article (Table 1). Many studies lacked a control group and had a small sample size. Three of the included studies were carried out by the same multidisciplinary renal transplant team at the Medical College of Georgia Hospital and Clinics.15,19,21 A fourth study had the same first author as these 3 studies (Marie A Chisholm-Burns, formerly Marie A Chisholm), but was conducted within a different organization.17
Our detailed literature search identified few studies describing the inclusion of clinical pharmacists as members of multidisciplinary teams in the organ transplant setting. In these studies, transplant pharmacists were involved in medication reconciliation, drug therapy evaluation and monitoring, patient education, and problem-solving. All of the studies included in our review suggested that transplant pharmacists could improve the management and medication adherence of patients and consequently could have a positive impact on patients’ morbidity, medication errors, and costs. However, each of the studies was conducted in a single centre, and it might be difficult to show significant evidence of a pharmacist’s impact in small, focused patient populations like these.
The number of studies that met our inclusion criteria (n = 12) was low compared with studies examining the roles of pharmacists in other settings (e.g., cancer, hypertension, and asthma).23 In fact, the involvement of clinical pharmacists in transplant medicine is recent. The American Society of Health-System Pharmacists now offers a pharmacy residency in solid organ transplant,24 but no European recommendations have been formulated regarding the role of the clinical pharmacist in transplantation. Lack of knowledge and/or experience in designing and administering such services, as well as difficulty in procuring funding and reimbursement for services, can limit the implementation of clinical pharmacy services in particular settings and locations.25
The included studies reported a large range of pharmacist activities in solid organ transplant for both hospitalized and ambulatory patients, as described in Table 1.
Alloway and others5 highlighted the following basic activities of the transplant pharmacist: dedicating time for the care of transplant recipient; attending daily rounds to evaluate pharmacotherapy; coordinating development and implementation of drug therapy protocols; providing medication reconciliation, medication therapy management, and discharge counselling; providing education to members of the transplant team; facilitating cost and pharmacotherapy optimization to maximize patient outcomes; providing transplant medication education to patients; leading and assisting with clinical and pharmacoeconomic research; and providing 24/7 pharmacotherapeutic support. This list strongly concurs with the interventions summarized in Table 1 of this review, except for research. Indeed, the most frequently reported activities in studies included in our review were patient education and counselling, reviewing and optimizing drug therapy, and medication reconciliation or medical history.
Patient education and teaching sessions aimed to educate patients about all aspects of their medications and the risks of nonadherence, and to answer questions. Handing out information sheets and providing support by phone or e-mail were activities performed by pharmacists in many of the studies.
Nonadherence to the immunosuppressive regimen after transplant is a major issue than can lead to serious outcomes, such as transplant rejection or even death. Clinical pharmacists can improve patient adherence to medications.15 In a unique approach, Chisholm-Burns and others17 used a behavioural contract and trimestral meetings to maximize patient adherence.
Reviewing and optimizing drug therapy helps in identifying, resolving and preventing drug-related problems. Musgrave and others13 reported a “significant” decrease of medication errors per patient at discharge because of pharmacist interventions. Chisholm and others15,19,21 also reported that pharmacist recommendations helped nephrologists to optimize prescriptions for transplant recipients.
Few of the included studies reported medication reconciliation. Nevertheless, this has been shown to be an essential component in optimizing the quality of prescriptions, preventing drug-related problems, and significantly reducing readmission rates in the emergency department.26 In the study by Maldonado and others,12 pharmacists proposed a discharge plan to the patients, in addition to performing medication reconciliation. Harrison and others14 described collaboration with community pharmacists; such collaborations reflect the importance of continuity of care between the transplant team and community practitioners to ensure an optimal prognosis. The development of telepharmacy tools may help with post-transplant home care.
Improvements in medication adherence, morbidity, costs, and medication errors were reported in the selected studies, but these outcomes were not linked to specific pharmacist activities.
There were clear benefits in terms of patient adherence to immunosuppressive treatments.11,15,17–20 Chisholm and others19 reported a significant reduction in transplant rejections from 1 year pre-enrollment to 1 year post-enrollment (p = 0.008). Klein and others18 found fewer rejection episodes in the intervention group, although the difference was not significant (small sample size). Three studies showed an increase in achievement of target serum concentrations of oral immunosuppressants.15,18,19
Significant positive outcomes were found in terms of comorbidities such as diabetes mellitus, hyperlipidemia, and hypertension, but the results were inconsistent for morbidity outcomes.16,19,21
In the study by Tschida and others,20 implementation of a transplant pharmacy program resulted in a significantly lower mean total cost per patient ($24 315 versus $27 891, 13% decrease; p = 0.03), which the authors attributed mainly to a significantly lower mean transplant-related medical cost ($5960 versus $8486, 30% decrease; p = 0.04).
Musgrave and others13 described the avoidance of discharge medication errors through pharmacist intervention, a decrease in discharge medication errors per patient persisting until the first follow-up appointment, and a greatly improved percentage of discharges with no medication errors. Harrison and others14 reported a decrease in the mean number of drug therapy problems identified per visit.
Patients’ knowledge of medications was appraised in only one study.22 The benefits for short-term information retention were significant, but the study did not examine long-term retention. Given that patient motivation and care intensity often diminish with time, long-term persistence of pharmacist-induced outcomes needs to be evaluated.
In studies designed to evaluate the roles and impacts of health care professionals, it is very important to have a clear and complete description of the intervention. Associating an intervention with specific outcomes is especially difficult where multidisciplinary teams are involved. According to the DEPICT tool,10 the descriptions of the interventions in the included studies were generally of good quality. Nonetheless, more complete descriptions should be provided in future studies, especially regarding the timing of the intervention and pharmacists’ autonomy.
As for most pharmacy practice research studies, the studies included in this review had small sample sizes, some had no control group (n = 3), and the interventions were insufficiently described to be fully reproducible. Usual sources of bias were reported, including performance bias and contamination bias. In clinical practice within a hospital, it is usually difficult to eliminate these 2 types of bias.
Transplant recipients are treated with multiple drugs, including medications with a narrow therapeutic index. It was therefore surprising to find only a limited number of articles describing pharmacists’ roles and outcomes in this area. This systematic review highlights the need to structure teaching and internships in this discipline and to further document the practice of pharmacists in transplant medicine. Professional specialty networks may certainly contribute to better training, organization, and documentation. For instance, the American Society for Transplantation has a transplant pharmacy community of practice.27 In addition, the American College of Clinical Pharmacy has an immunology/transplantation practice and research network.28 The Board of Pharmacy Specialties received a petition to recognize solid organ transplantation pharmacy as a new specialty; the Board’s public comment period on this petition closed on May 15, 2018.29
In Canada, the Canadian Society of Transplantation has a pharmacist group whose mission is to “provide leadership and a collaborative forum for the advancement of pharmacist clinical practice in transplantation and pharmacist-led research and education”.30 The Canadian Society of Hospital Pharmacists has a transplant Pharmacy Specialty Network that promotes “practice excellence and the enhancement of patient-centred pharmacy practice through information sharing, educational events, and the facilitation of research for pharmacists who are interested in the area of transplant pharmacy practice (solid organ and hematopoietic stem cell transplant)”.31
There is currently no published literature about transplant-specific training offered in pharmacy, in Canada or elsewhere. Such training may vary substantially among regions and programs, which may explain the paucity of data as well as the wide variety of roles described in the literature.
The systematic literature search was conducted in only 4 databases, and all articles published in a language other than English or French were excluded. As a result, some eligible studies may have gone undetected. Although descriptive results lack statistical proof of significance, they may carry compelling information that could prove useful in establishing a more accurate image of the roles and impacts of the pharmacist. However, for practical reasons (notably the difficulty of screening for quality), they were omitted from this review. Eight studies involved kidney transplant recipients exclusively, and the 4 remaining studies were spread among recipients of abdominal, liver, lung, and unspecified transplants. Most anti-rejection medications are lifelong treatments, yet the temporal horizon was limited to a year or less in virtually all of the studies. It is unknown whether pharmacist interventions have lasting effects, especially in the case of temporary activities. It would be interesting to explore which interventions were the most time-effective.
Currently available evidence suggests that pharmacists can improve patient outcomes in solid organ transplant settings. Adherence, morbidity, costs, and medication errors were identified as the main outcomes that were improved by pharmaceutical interventions. Transplant programs need to invest more in this resource.
1 Annual statistics on organ replacement in Canada: dialysis, transplantation and donation, 2007 to 2016. Ottawa (ON): Canadian Institute for Health Information; 2017 [cited 2018 May 17]. Available from: https://www.cihi.ca/sites/default/files/document/corr_ar-snapshot-en.pdf
2 Denhaerynck K, Dobbels F, Cleemput I, Desmyttere A, Schäfer-Keller P, Schaub S, et al. Prevalence, consequences, and determinants of nonadherence in adult renal transplant patients: a literature review. Transpl Int. 2005; 18(10):1121–33.
3 Pinsky BW, Takemoto SK, Lentine KL, Burroughs TE, Schnitzler MA, Salvalaggio PR. Transplant outcomes and economic costs associated with patient noncompliance to immunosuppression. Am J Transplant. 2009; 9(11):2597–606.
4 Mitchell JF. Pharmacist involvement as a member of a renal transplant team. Am J Hosp Pharm. 1976;33(1):55–8.
5 Alloway RR, Dupuis R, Gabardi S, Kaiser TE, Taber DJ, Tichy EM, et al. Evolution of the role of the transplant pharmacist on the multidisciplinary transplant team. Am J Transplant. 2011;11(8):1576–83.
6 Singhal PK, Raisch DW, Gupchup GV. The impact of pharmaceutical services in community and ambulatory care setting: evidence and recommendations for future research. Ann Pharmacother. 1999;33(12):1336–55.
7 Melchiors AC, Correr CJ, Venson R, Pontarolo R. An analysis of quality of systematic reviews on pharmacist health interventions. Int J Clin Pharm. 2012;34(1):32–42.
8 Charrois TL, Durec T, Tsuyuki RT. Systematic reviews of pharmacy practice research: methodologic issues in searching, evaluating, interpreting, and disseminating results. Ann Pharmacother. 2009;43(1):118–22.
9 Moher D, Liberati A, Tetzlaff J, Altman DG. PRISMA 2009 checklist. PRISMA Group; 2009 [cited 2016 Sep 20]. Available from: http://prisma-statement.org/documents/PRISMA%202009%20checklist.pdf
10 Correr CJ, Melchiors AC, de Souza TT, Rotta I, Salgado TM, Fernandez-Llimos F. A tool to characterize the components of pharmacist interventions in clinical pharmacy services: the DEPICT project. Ann Pharmacother. 2013;47(7–8):946–52.
11 Joost R, Dörje F, Schwitulla J, Eckardt KU, Hugo C. Intensified pharmaceutical care is improving immunosuppressive medication adherence in kidney transplant recipients during the first post-transplant year: a quasi-experimental study. Nephrol Dial Transplant. 2014;29(8):1597–607.
12 Maldonado AQ, Weeks DL, Bitterman AN, McCleary JA, Seiger TC, Carson RW, et al. Changing transplant recipient education and inpatient transplant pharmacy practices: a single-center perspective. Am J Health Syst Pharm. 2013;70(10):900–4.
13 Musgrave CR, Pilch NA, Taber DJ, Meadows HB, McGillicuddy JW, Chavin KD, et al. Improving transplant patient safety through pharmacist discharge medication reconciliation. Am J Transplant. 2013;13(3):796–801.
14 Harrison JJ, Wang J, Cervenko J, Jackson L, Munyal D, Hamandi B, et al. Pilot study of a pharmaceutical care intervention in an outpatient lung transplant clinic. Clin Transplant. 2012;26(2):E149–57.
15 Chisholm MA, Mulloy LL, Jagadeesan M, DiPiro JT Impact of clinical pharmacy services on renal transplant patients’ compliance with immunosuppressive medications. Clin Transplant. 2001;15(5):330–6.
16 Pinelli NR, Clark LM, Carrington AC, Carrington JL, Malinzak L, Patel A. Pharmacist managed diabetes and cardiovascular risk reduction clinic in kidney transplant recipients: bridging the gap in care transition. Diabetes Res Clin Pract. 2014;106(3):e64–7.
17 Chisholm-Burns MA, Spivey CA, Graff Zivin J, Lee JK, Sredzinski E, Tolley EA. Improving outcomes of renal transplant recipients with behavioral adherence contracts: a randomized controlled trial. Am J Transplant. 2013;13(9):2364–73.
18 Klein A, Otto G, Krämer I. Impact of a pharmaceutical care program on liver transplant patients’ compliance with immunosuppressive medication: a prospective, randomized, controlled trial using electronic monitoring. Transplantation. 2009;87(6):839–47.
19 Chisholm MA, Spivey CA, Mulloy LL. Effects of a medication assistance program with medication therapy management on the health of renal transplant recipients. Am J Health Syst Pharm. 2007;64(14):1506–12.
20 Tschida S, Aslam S, Khan TT, Sahli B, Shrank WH, Lal LS. Managing specialty medication services through a specialty pharmacy program: the case of oral renal transplant immunosuppressant medications. J Manag Care Pharm. 2013;19(1):26–41.
21 Chisholm MA, Mulloy LL, Jagadeesan M, Martin BC, DiPiro JT. Effect of clinical pharmacy services on the blood pressure of African-American renal transplant patients. Ethn Dis. 2002;12(3):392–7.
22 Partovi N, Chan W, Nimmo CR. Evaluation of a patient education program for solid organ transplant patients. Can J Hosp Pharm. 1995;48(2):72–8.
23 Impact pharmacie [website]. Montréal (QC) : Centre hospitalier universitaire Sainte-Justine, Unité de recherche en pratique pharmaceutique; [cited 2018 Oct 4], Available from: http://impactpharmacie.org/index.php?lang=01
24 Required educational outcomes, goals, and objectives for Postgraduate Year Two (PGY2) pharmacy residencies in solid organ transplant. Bethesda (MD): American Society of Health-System Pharmacists; [cited 2018 May 17]. Available from: https://www.ashp.org/-/media/assets/professional-development/residendes/docs/pgy2-solid-organ-transplant-pharmacy.ashx?la=en&hash=52CA32882E85E9A431FFD9C2B34ACA09DB3F0A39
25 Chisholm MA. A renal transplantation advanced pharmacy practice experience. Am J Pharm Educ. 2006;15;70(1):3.

26 Renaudin P, Boyer L, Esteve MA, Bertault-Peres P, Auquier P, Honore S. Do pharmacist-led medication reviews in hospitals help reduce hospital readmissions? A systematic review and meta-analysis. Br J Clin Pharmacol 2016;82(6):1660–73.

27 Transplant pharmacy community of practice (TxPharm COP). Mt Laurel (NJ): American Society for Transplantation; [cited 2018 May 25]. Available from: https://www.myast.org/communities-practice/txpharmcop
28 Practice and research networks: Immunology/transplantation PRN. Lenexa (KS): American College of Clinical Pharmacy; [cited 2016 Dec 14]. Available from: https://www.accp.com/about/prns.aspx
29 Board of Pharmacy Specialties receives petition to recognize solid organ transplantation pharmacy as a specialty. Washington (DC): Board of Pharmacy Specialties; 2018 Apr 4 [cited 2018 May 15]. Available from: https://www.bpsweb.org/2018/04/04/board-of-pharmacy-specialties-receives-petition-to-recognize-solid-organ-transplantation-pharmacy-as-a-specialty/
30 Pharmacist group. Ottawa (ON): Canadian Society of Transplantation; [cited 2018 May 24]. Available from: http://www.cst-transplant.ca/pharmacist-group.html
31 PSN communities: Transplant. Ottawa (ON): Canadian Society of Hospital Pharmacists; [cited 2018 May 17]. Available from: https://www.cshp.ca/psn-communities
Competing interests: None declared. ( Return to Text )
Address correspondence to: Jean-François Bussières, Pharmacy Department, Centre hospitalier universitaire Sainte-Justine, 3175, chemin de la Côte Sainte-Catherine, Montréal QC H3T 1C5. e-mail:jf.bussieres@ssss.gouv.qc.ca
Funding: None received. ( Return to Text )
Canadian Journal of Hospital Pharmacy, VOLUME 71, NUMBER 5, September-October 2018