Hironori Tanaka, Rei Tanaka, Takeshi Kamoshida, Shigeki Morimoto, Junya Sato, Hiroshi Ishikawa, Tetsu Sato, Tetsumi Sato, Akiyoshi Saitoh, Daisuke Yamada, Mina Kondo, Kenji Takahashi, Chieko Takahashi, and Michihiro ShinoTo cite: Tanaka H, Tanaka R, Kamoshida T, Morimoto S, Sato J, Ishikawa H, et al. Incidence of delirium during the initiation phase of morphine and hydromorphone therapy in cancer patients: a retrospective comparative study. Can J Hosp Pharm. 2025;78(1):e3515. doi: 10.4212/cjhp.3515
ABSTRACT
Background
Opioids are known to induce delirium, and the incidence of delirium induced by individual opioids has been investigated. However, only a limited number of studies have examined the incidence of delirium induced by oral hydromorphone.
Objective
To investigate whether differences exist in the incidence of delirium associated with oral morphine and oral hydromorphone during the initiation phase of treatment.
Methods
The participants were opioid-naive inpatients with cancer who started oral morphine or oral hydromorphone at Shizuoka Cancer Center (in Shizuoka, Japan) between June 2017 and November 2020. The incidence of delirium in the first week of opioid use was compared between the 2 groups.
Results
A total of 90 patients met the inclusion criteria, 27 who received oral hydromorphone and 63 who received oral morphine. The incidence rate of delirium in the oral hydromorphone group tended to be higher (19%, 5/27) than in the oral morphine group (8%, 5/63), although the difference was not statistically significant (odds ratio 0.4, 95% confidence interval, 0.1–1.4, p = 0.16 by the Fisher exact test). Propensity score matching was used to control for differences in patient background as confounders in the development of delirium, following which the incidence rate of delirium remained higher, but not significantly so, in the oral hydromorphone group (11%, 2/19) than in the oral morphine group (5%, 1/19) (odds ratio 0.5, 95% confidence interval 0.04–5.7, p > 0.99 by the Fisher exact test).
Conclusions
There was no statistically significant difference in the incidence of delirium between those who received morphine and those who received hydromorphone, which suggests that for opioid-naive inpatients with cancer, oral hydromorphone can be used in a manner similar to that for oral morphine.
Keywords: delirium, oral opioids, morphine, hydromorphone
RÉSUMÉ
Contexte
Les opioïdes sont connus pour induire un délire, et l’incidence du délire induit par des opioïdes individuels a fait l’objet d’études. Cependant, seul un nombre limité de celles-ci s’est penché sur l’incidence du délire induit par l’hydromorphone orale.
Objectif
Étudier s’il existe des différences dans l’incidence du délire associé à la morphine orale et à l’hydromorphone orale pendant la phase d’initiation du traitement.
Méthodologie
La population à l’étude se composait de patients hospitalisés naïfs aux opioïdes atteints de cancer; ils ont commencé à prendre de la morphine orale ou de l’hydromorphone orale au Shizuoka Cancer Center (à Shizuoka, au Japon) entre juin 2017 et novembre 2020. L’incidence du délire au cours de la première semaine de consommation d’opioïdes a été comparée entre les 2 groupes.
Résultats
Au total, 90 patients répondaient aux critères d’inclusion : 27 avaient reçu de l’hydromorphone et 63 de la morphine. Le taux d’incidence du délire dans le groupe « hydromorphone orale » avait tendance à être plus élevé (19 %, 5/27) que dans le groupe « morphine orale » (8 %, 5/63), bien que la différence ne soit pas statistiquement significative (rapport de cotes [RC] 0,4, intervalle de confiance [IC] à 95 %, 0,1–1,4, p = 0,16 par le test exact de Fisher). L’appariement par score de propension a été utilisé pour contrôler les différences dans les antécédents des patients comme facteurs de confusion dans le développement du délire, après quoi le taux d’incidence du délire est resté plus élevé, mais pas de manière significative, dans le groupe « hydromorphone orale » (11 %, 2/19) que dans le groupe « morphine orale » (5 %, 1/19) (RC 0,5, IC à 95 % 0,04–5,7, p > 0,99 par le test exact de Fisher).
Conclusions
Aucune différence statistiquement significative dans l’incidence du délire entre les personnes ayant reçu de la morphine et celles ayant reçu de l’hydromorphone n’a été observée. Cela laisse supposer que pour les patients hospitalisés naïfs aux opioïdes atteints de cancer, l’hydromorphone orale peut être utilisée d’une manière similaire à l’utilisation de la morphine orale.
Mots-clés: délire, opioïdes oraux, morphine, hydromorphone
Delirium is a major problem in palliative care,1 and its prevention and care are of great importance. Factors associated with the development of delirium include old age, smoking, alcohol use, comorbidities such as dementia, and medications.2 Drug-induced delirium can be prevented by selecting drugs with the lowest possible risk. Various drugs can induce delirium, including opioids, benzodiazepines, steroids, H2 receptor antagonists, anticonvulsants, antihistamines, and anticholinergics. Opioids constitute one of the most common groups of medications associated with delirium.1 Between 2012 and 2014, we investigated the incidence of delirium induced by opioids given by injection in opioid-naive patients with cancer pain and found that the incidence was significantly lower with fentanyl citrate than with morphine hydrochloride.3 More recently, we investigated the incidence of delirium induced by extended-release opioids given orally and found no significant differences among morphine sulfate, oxycodone hydrochloride, and tapentadol hydrochloride.4
In Japan, hydromorphone first became available for oral administration in 2017. It is available in a 24-hour extended-release formulation that can be administered once daily, putting minimal burden on patients. If administered according to an appropriate conversion ratio, morphine and hydromorphone have comparable analgesic effects.5–7 Given the similarities in structure and affinity for μ, δ, and κ receptors between hydromorphone and morphine, 8–10 the incidence of delirium is not expected to differ markedly between them.
In a North American clinical study, hydromorphone was reported to have a lower risk of delirium than several other opioids (including morphine).11 That study focused on postoperative delirium in patients who underwent total joint arthroplasty, but it is also necessary to investigate the risk of delirium in patients with cancer, who are likely to use opioids, and in non-postoperative patients. The incidence of delirium associated with hydromorphone has not been fully studied because it has been on the market for only a short time, particularly in Japan. It is important to compare clinical data on the incidence of delirium between hydromorphone and other opioids to verify whether the results fit with those expected from basic science.
Here, we compared the incidence of delirium induced by oral morphine and by oral hydromorphone in opioid-naive patients with cancer.
The participants in this retrospective study were opioid-naive inpatients with cancer who started oral morphine or oral hydromorphone for relief of pain or respiratory distress at Shizuoka Cancer Center from June 2017 to November 2020. Opioid naivety was defined as having never received prescribed opioids at Shizuoka Cancer Center or any other medical institution, based on the patient’s drug history in the electronic medical record. The incidence of delirium in the first week of opioid treatment was assessed with the Intensive Care Delirium Screening Checklist (ICDSC).12 Two of the researchers (H.T. and R.T.) retrospectively extracted the ICDSC scores entered in patients’ electronic health records by treating doctors, nurses, or pharmacists. Patients with an ICDSC score of 3 or below at the time of opioid initiation were included in the study, and the occurrence of delirium was defined as an ICDSC score of at least 4. As such, patients with an ICDSC score of 4 or higher at the time of opioid initiation were excluded. If there were no ICDSC scores in the medical record, the score was deemed to be 0. Postoperative patients and patients with infections were also excluded, to eliminate potential effects of surgery and infection on delirium. The definition of infection included both active infection at the time of opioid initiation and the development of infection at any point during opioid therapy.
The following patient characteristics, which may be confounders in the development of delirium, were retrospectively extracted from the medication history, medical history, and blood test data in the most up-to-date electronic medical records documented before any opioid administration: morphine equivalent dose (where the morphine–hydromorphone ratio is 5:1)5–7; age; sex; baseline performance status (PS); presence or absence of primary brain tumour or brain metastases, central nervous system disease, dementia, past alcohol use, renal failure, or hepatic failure; and cancer type. Renal failure was defined as an increase in serum creatinine of grade 1 or greater, and hepatic failure as an increase in blood aspartate aminotransferase or alanine aminotransferase of grade 1 or greater, according to the Common Terminology Criteria for Adverse Events, version 5.0 (https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm#ctc_50). Also investigated was each patient’s concomitant use, within 1 week before and 1 week after starting opioid therapy, of the following drugs that may be associated with the occurrence of delirium: benzodiazepines, anticholinergics, antidopaminergics, antihistamines, H2 receptor antagonists, and steroids. The attending physician selected which opioid to use based on consideration of the patient’s particular situation.
Propensity score matching was performed to control for differences between the groups in terms of confounders in the development of delirium (p < 0.05). In this report, Analysis 1 refers to the full analysis of data before propensity score matching, and Analysis 2 refers to the analysis after adjustment for patient characteristics by propensity score matching.13 The incidence of delirium was compared between the 2 drug groups with the Fisher exact test. Patient characteristics, excluding cancer type, and concomitant use of drugs associated with the occurrence of delirium were also compared between the groups using the Fisher exact test. The cut-off for age as a confounder was set at 65 years, based on the definition of “elderly” used by several organizations, including the World Health Organization14; also, in a previous study of delirium, Halladay and others15 used 65 years as one of their age cut-offs. The cut-off dose was set as morphine equivalent of 90 mg/day, based on a study by Gaudreau and others.16 In performing the Fisher exact test, we corrected for n = 0 with a correction value of +0.5. The statistical analyses were performed using EZR software, version 1.55 (Saitama Medical Center, Jichi Medical University), with significance set at p < 0.05 for all analyses. Matched-pair analysis was used to extract the matched pairs for Analysis 2. The analysis conditions were as follows: PropensityScore.GLM.1, case to control ratio = 1:1, and caliper width = 0.2.
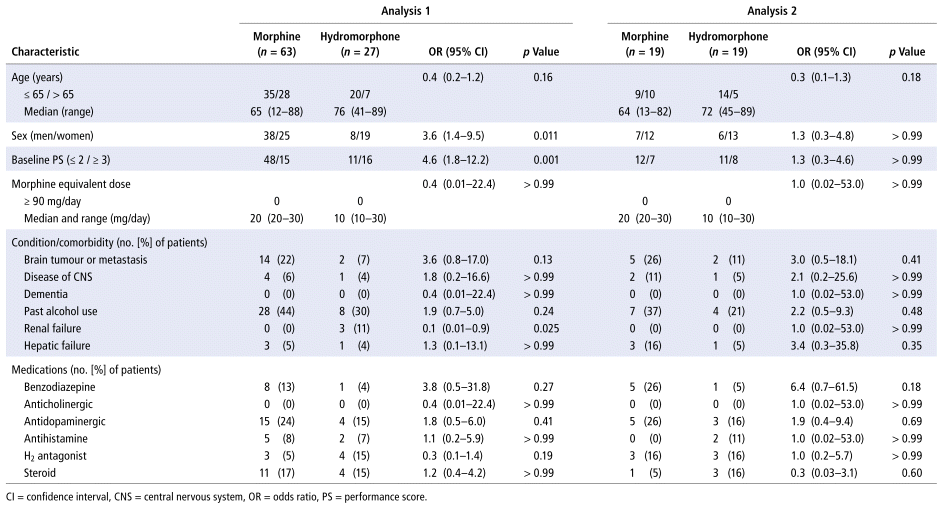
Of the 90 patients who met the inclusion criteria, 63 received oral morphine and 27 received oral hydromorphone. Table 1 shows a comparison between the groups in terms of patients’ baseline characteristics. In Analysis 1 (all data), there were no significant differences between the groups in terms of age or the presence/absence of primary brain tumours or brain metastases, central nervous system disease, dementia, past alcohol use, or hepatic failure. However, there was a significant difference in sex ratio between the oral morphine group and the oral hydromorphone group. In addition, baseline PS differed significantly, as did the presence/absence of renal failure.
TABLE 1 Patient Characteristics as Confounders in Development of Delirium before (Analysis 1) and after (Analysis 2) Propensity Score Matching

In Analysis 2, after propensity score matching for patient characteristics with significant differences in Analysis 1 (i.e., sex, baseline PS, and presence/absence of renal failure), no significant between-group differences were found for any of the 3 items.
With regard to concomitant drug use, both Analysis 1 and Analysis 2 showed no significant differences between the groups in the use of benzodiazepines, anticholinergics, antidopaminergics, antihistamines, H2 receptor antagonists, or steroids (Table 1).
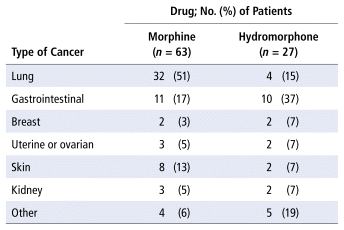
Table 2 shows the number of patients in each group according to cancer type. Oral morphine was most commonly prescribed for patients with lung cancer (n = 32, 51%) and oral hydromorphone for those with gastrointestinal cancer (n = 10, 37%).
TABLE 2 Administration of Oral Morphine or Oral Hydromorphone by Patient’s Type of Cancer
The incidence of delirium in the 2 groups is shown in Figure 1. In Analysis 1, the incidence rate of delirium tended to be higher, but not significantly so, in the oral hydromorphone group than in the oral morphine group (19% vs 8%; odds ratio [OR] 0.4, 95% confidence interval [CI] 0.1–1.4, p = 0.16). In Analysis 2, after propensity score matching, the incidence rate of delirium remained higher, but again not significantly so, in the oral hydromorphone group than in the oral morphine group (11% vs 5%; OR 0.5, 95% CI 0.04–5.7, p > 0.99).
|
| ||
|
FIGURE 1 Incidence of delirium induced by oral morphine and oral hydromorphone, before and after propensity score matching. In both analyses, there was no significant difference between the groups. | ||
The results of both Analysis 1 and Analysis 2, before and after propensity score matching, showed no significant difference in the incidence of delirium between the oral morphine and oral hydromorphone groups. This finding agrees with our previous study, in which we found nonsignificant differences in the incidence of delirium induced by oral opioids. In that earlier study, the incidence of delirium in patients taking oral morphine, oxycodone, or tapentadol was 4.8%, 6.9%, and 6.7%, respectively.4 Those past results are similar to the 8% and 5% incidence of delirium with morphine before and after matching, respectively, in the current study. Oral hydromorphone appears to have a risk of delirium similar to that of oral oxycodone or oral tapentadol. Also, given that the risk of delirium increases when the dose exceeds a morphine equivalent dose of 90 mg,16 oral hydromorphone in the dose range of 2 to 6 mg, as examined in this study, had only a minor effect on delirium. Taken together, these findings indicate that any of these oral opioids is a possible choice based on the occurrence of delirium in opioid-naive patients. Because the purpose of this study was to determine which oral drug was better for opioid initiation, there were no cases in which opioids were administered at a morphine equivalent dose of 90 mg or more. An important topic for future study would be to examine whether morphine, oxycodone, tapentadol, or hydromorphone is more suitable when switching opioids in patients already using a particular opioid at higher doses.
Analysis 1 showed a difference in the sex ratio between the oral morphine and oral hydromorphone groups. Morphine is the drug of choice for treating dyspnea in Japan.17 Many of the patients in the oral morphine group had lung cancer, and the epidemiology of lung cancer may have led to the larger proportion of men in the oral morphine group.18 The present study also showed significant differences in baseline PS and the presence/absence of renal failure between the groups, probably because morphine is not recommended for patients with renal failure.7,19 As such, hydromorphone was used for those patients.
Nandi and others11 investigated risk factors for postoperative delirium in patients who underwent total joint arthroplasty in North America. In contrast, we investigated the risk factors for non-postoperative delirium in a population of Japanese patients with cancer. Furthermore, in previous studies, multiple opioids including morphine were compared with hydromorphone, but our study compared only morphine and hydromorphone. Although the number of cases in our study was small, confounding according to the type of opioid was reduced. Our results might show different trends from those previously reported, depending on patients’ ethnic background, the targeted disease, and the type of delirium. The structure of hydromorphone is similar to that of morphine, with the addition of a keto group at the 6 position and hydrogen at the 7 and 8 positions. Also, there is similarity in terms of affinity for the μ, δ, and κ opioid receptors, suggesting that the incidence of delirium caused by each drug would be similar.8–10
One of the limitations of this study was our inability to compare blood levels of electrolytes (sodium, potassium, calcium)20 between the groups because for many patients, these levels were not measured at the start of opioid therapy; as such, their potential influence on the results cannot be ruled out. Also, differences in how hospital staff implemented nonpharmacological measures, such as interventions to address room lighting, malnutrition, and sleep deprivation,21 might also have influenced the results. Another limitation, arising from the retrospective nature of the study, is that opioid selection was at the discretion of the attending physician, which may have introduced bias. For purposes of our analyses, we assumed that appropriate doses of opioids were administered, but a more detailed prospective comparative study, including analgesic effects, is needed. A further limitation is that the 2 researchers (H.T. and R.T.) who checked and audited medical records for the relevant period were also involved in the analysis itself. The descriptions of delirium in the medical records depended on assessments performed by and the relevant skills of the attending doctor, nurse, and pharmacist (e.g., how often assessments are done and when). In addition, since it was not possible to clearly distinguish between the chemotherapy and best supportive care phases of each patient’s treatment, future prospective studies should gather clinical data with consistent patient staging.
There was no statistically significant difference in the incidence of delirium between those who received morphine and those who received hydromorphone. Although ours was a comparative study involving a relatively small number of cases, these findings suggest that either of these opioids can be selected for such inpatients. Given the retrospective nature of this study, the influence of confounding factors could not be ruled out, and future prospective studies are warranted.
1 Tuma R, DeAngelis LM. Altered mental status in patients with cancer. Arch Neurol. 2000;57(12):1727–31.
Crossref PubMed
2 Gleason OC. Delirium. Am Fam Physician. 2003;67(5):1027–34.
PubMed
3 Tanaka R, Ishikawa H, Sato T, Shino M, Matsumoto T, Mori K, et al. Incidence of delirium among patients having cancer injected with different opioids for the first time. Am J Hosp Palliat Care. 2017;34(6): 572–6.
Crossref
4 Sugiyama Y, Tanaka R, Sato T, Sato T, Saitoh A, Yamada D, et al. Incidence of delirium with different oral opioids in previously opioid-naive patients. Am J Hosp Palliat Care. 2022;39(10):1145–51.
Crossref PubMed PMC
5 Inoue S, Saito Y, Tsuneto S, Aruga E, Ogata T, Uemori M. A double-blind, randomized comparative study to investigate the morphine to hydromorphone conversion ratio in Japanese cancer patients. Jpn J Clin Oncol. 2018;48(5):442–9.
Crossref PubMed PMC
6 Hanks GW, Conno F, Cherny N, Hanna M, Kalso E, McQuay HJ, et al.; Expert Working Group of the Research Network of the European Association for Palliative Care. Morphine and alternative opioids in cancer pain: the EAPC recommendations. Br J Cancer. 2001;84(5):587–93.
Crossref PubMed PMC
7 Japanese Society for Palliative Medicine, Palliative Medicine Guidelines General Committee. [Clinical guidelines for pharmacological treatment of cancer pain (2020 version)]. Kanehara & Co, Ltd; 2020. In Japanese.
8 Martin WR, Eades CG, Thompson JA, Huppler RE, Gilbert PE. The effects of morphine- and nalorphine-like drugs in the nondependent and morphine-dependent chronic spinal dog. J Pharmacol Exp Ther. 1976;197(3):517–32.
PubMed
9 Wentland MP, Lou R, Lu Q, Bu Y, Denhardt C, Jin J, et al. Syntheses of novel high affinity ligands for opioid receptors. Bioorg Med Chem Lett. 2009;19(8):2289–94.
Crossref PubMed PMC
10 Volpe DA, McMahon Tobin GA, Mellon RD, Katki AG, Parker RJ, Colatsky T, et al. Uniform assessment and ranking of opioid μ receptor binding constants for selected opioid drugs. Regul Toxicol Pharmacol. 2011;59(3):385–90.
Crossref PubMed
11 Nandi S, Harvey WF, Saillant J, Kazakin A, Talmo C, Bono J. Pharmacologic risk factors for post-operative delirium in total joint arthroplasty patients: a case-control study. J Arthroplasty. 2014;29(2):268–71.
Crossref
12 Bergeron N, Dubois MJ, Dumont M, Dial S, Skrobik Y. Intensive Care Delirium Screening Checklist: evaluation of a new screening tool. Intensive Care Med. 2001;27(5):859–64.
Crossref PubMed
13 Sato J, Tanaka R. A retrospective comparison of haloperidol and hydroxyzine combination therapy with haloperidol alone in the treatment of overactive delirium. Support Care Cancer. 2022;30(6):4889–96.
Crossref PubMed
14 Singh S, Bajorek B. Defining ‘elderly’ in clinical practice guidelines for pharmacotherapy. Pharm Pract (Granada). 2014;12(4):489.
Crossref
15 Halladay CW, Sillner AY, Rudolph JL. Performance of electronic prediction rules for prevalent delirium at hospital admission. JAMA Netw Open. 2018;1(4):e181405.
Crossref
16 Gaudreau JD, Gagnon P, Roy MA, Harel F, Tremblay A. Opioid medications and longitudinal risk of delirium in hospitalized cancer patients. Cancer. 2007;109(11):2365–73.
Crossref PubMed
17 Japanese Society for Palliative Medicine, Palliative Medicine Committee. [Clinical guidelines for respiratory symptoms in cancer patients (2016 version)]. Kanehara & Co, Ltd; 2016. In Japanese.
18 Torre LA, Siegel RL, Jemal A. Lung cancer statistics. Adv Exp Med Biol. 2016;893:119.
19 Mawatari H, Shinjo T, Morita T, Kohara H, Yomiya K. Revision of pharmacological treatment recommendations for cancer pain: clinical guidelines from the Japanese Society of Palliative Medicine. J Palliat Med. 2022;25(7):1095–114.
Crossref PubMed
20 Korevaar JC, van Munster BC, de Rooij SE. Risk factors for delirium in acutely admitted elderly patients: a prospective cohort study. BMC Geriatr. 2005;5:6.
Crossref PubMed PMC
21 Abraha I, Rimland JM, Trotta F, Pierini V, Cruz-Jentoft A, Soiza R, et al. Non-pharmacological interventions to prevent or treat delirium in older patients: clinical practice recommendations the SENATOR-ONTOP series. J Nutr Health Aging. 2016;20(9):927–36.
Crossref PubMed
Address correspondence to: Dr Rei Tanaka, Department of Pharmacy, Shizuoka Cancer Center, 1007 Shimonagakubo, Nagaizumi-cho, Sunto-gun, Shizuoka-ken 411-8777, Japan, email: 32114152@alumni.tus.ac.jp
Competing interests: None declared.
Funding: None received.
Submitted: June 26, 2023
Accepted: July 5, 2024
Published: January 15, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 1, 2025