Emily Blacklaws, Kieran Shah, and Sarah N StablerTo cite: Blacklaws E, Shah K, Stabler SN. Glycemic management in patients with COVID-19 admitted to the intensive care unit: evaluation of glycemic control and drug therapy. Can J Hosp Pharm. 2024;77(4):e3553. doi: 10.4212/cjhp.3553
ABSTRACT
Background
Emerging evidence describes the high incidence and strong impact of hyperglycemia on the outcomes of critically ill patients with a diagnosis of COVID-19. Given resource limitations during the COVID-19 pandemic, clinicians moved away from using continuous IV infusions of insulin to manage hyperglycemia.
Objective
To evaluate glycemic control in critically ill patients receiving various medication regimens to manage their hyperglycemia.
Methods
This retrospective cohort study involved 120 mechanically ventilated adult patients (> 18 years) with COVID-19 who were admitted to the intensive care unit (ICU) between February 2020 and December 2021. The following data were collected for the first 14 days of the ICU admission: blood glucose values (up to 4 times daily), hypoglycemia events, and antihyperglycemic medication regimens.
Results
The use of IV insulin infusions maintained glucose measurements within the target range of 4 to 10 mmol/L more often than any other medication regimen, with 60% of measured values falling within the target range. The use of a sliding-scale insulin regimen maintained 52% of glucose measurements within the target range. Oral hypoglycemic agents performed relatively poorly, with only 12% to 29% of glucose measurements within range. The coadministration of corticosteroids led to worse glycemic control across all medication regimens.
Conclusions
This study confirmed that ICUs should continue using the standard protocol of IV insulin infusion to achieve recommended blood glucose targets in critically ill patients with COVID-19, particularly those receiving corticosteroids.
KEYWORDS: critical care, intensive care, diabetes mellitus, glycemic control, COVID-19
RÉSUMÉ
Contexte
Des données probantes émergentes font état de l’incidence élevée et des fortes répercussions de l’hyperglycémie sur les résultats des patients gravement malades ayant reçu un diagnostic de COVID-19. Vu les ressources restreintes pendant la pandémie de COVID-19, les cliniciens se sont éloignés de l’utilisation des perfusions continues d’insuline par IV pour gérer l’hyperglycémie.
Objectif
Évaluer le contrôle glycémique chez les patients gravement malades qui reçoivent divers régimes médicamenteux pour gérer leur hyperglycémie.
Méthodologie
Cette étude de cohorte rétrospective portait sur 120 patients adultes (> 18 ans) atteints de la COVID-19 ventilés mécaniquement ayant été admis à une unité de soins intensifs entre février 2020 et décembre 2021. Les données suivantes ont été recueillies pendant les 14 premiers jours de l’admission en USI : valeurs glycémiques (jusqu’à 4 fois par jour), événements d’hypoglycémie et régimes de médicaments antihyperglycémiants.
Résultats
L’utilisation de perfusions d’insuline par intraveineuse permettait de maintenir les mesures de glucose dans la plage cible de 4 à 10 mmol/L plus souvent que tout autre schéma thérapeutique, avec 60 % des mesures se situant dans la plage cible. L’utilisation d’un schéma thérapeutique à insuline à échelle mobile a permis de maintenir 52 % des mesures de glucose dans la plage cible. Les résultats des hypoglycémiants oraux étaient relativement mauvais, avec seulement 12 % à 29 % des mesures de glucose se situant dans la plage cible. L’administration concomitante de corticostéroïdes a entraîné un moins bon contrôle glycémique dans tous les schémas thérapeutiques.
Conclusions
Cette étude a confirmé que les unités de soins intensifs devraient continuer à utiliser le protocole standard de perfusion d’insuline par IV pour atteindre les objectifs de glycémie recommandés chez les patients gravement malades atteints de la COVID-19, en particulier ceux recevant des corticostéroïdes.
Mots-clés: soins critiques, soins intensifs, diabète, contrôle glycémique, COVID-19
In early 2020, the spread of SARS-CoV-2 and the emergence of the COVID-19 pandemic led to intensive care units (ICUs) around the world coping with a surge of critically ill patients.1 Out of necessity, multiple, rapid changes were made to standard evidence-based practice patterns to manage scarce resources and mitigate risk of exposure among the staff caring for these patients. At our institution, one of these changes involved the clustering of nursing care interventions, to preserve the use of short-supply personal protective equipment (PPE) and to reduce the risk of viral exposure for staff while providing care and while doffing PPE. One of the significant therapeutic impacts of this change in practice was the shift from using the standard protocol for IV insulin infusion to using alternative therapies such as sliding-scale insulin with or without basal insulin, oral hypoglycemic agents, or a combination of these strategies. This approach allowed for glucose to be checked every 4 hours, rather than every 1 to 2 hours. Subsequently, during the multiple surges of patients admitted to the Surrey Memorial Hospital ICU, glycemic control was observed to be consistently suboptimal, and maintaining optimal control remained an ongoing therapeutic challenge. As we emerged out of the pandemic and critical care admissions returned to more typical seasonal levels, PPE was no longer in short supply, and nurse-to-patient ratios normalized, routine standards of care also began to return to baseline. However, one of the challenges we experienced was a lack of acceptance by prescribers and nurses to return to using IV insulin infusion as standard therapy for managing hyperglycemia in critically ill patients whether or not they had COVID-19.
Outside of the COVID-19 pandemic, multiple studies have shown an association between hyperglycemia and increased morbidity and mortality in mixed critical care populations.2–4 Although tight glycemic control has not been proven to improve outcomes in this patient population and the optimal blood glucose target in the critical care population remains controversial, guidelines recommend maintaining blood glucose below 10 mmol/L while avoiding hypoglycemia (defined as < 4 mmol/L).2–4 The lower level of the recommended target range is typically between 7 and 8 mmol/L to reduce the risk of hypoglycemia episodes, given the lack of benefit and risk of harm with intensive glycemic control in critical care patients.2,3 Due to both patient- and disease-related factors in critical care patients, whose condition is often unstable, the standard treatment regimen for hyperglycemia is continuous IV infusion of regular insulin.
Early in the pandemic, diabetes mellitus was identified as a significant risk factor for morbidity and mortality related to COVID-19, which added to the complexity of managing hyperglycemia.1,5 This phenomenon was also seen in previous coronavirus outbreaks, specifically Middle Eastern respiratory syndrome and severe acute respiratory syndrome coronavirus 1.6 Although the link between diabetes and severity of illness in patients with SARS-CoV-2 infection is not yet fully understood, it is hypothesized that hyperglycemia impairs the host immune response through impaired phagocytosis and chemotaxis, decreased interleukin production, and reduced T-cell and neutrophil response.6,7 The inflammatory storm associated with COVID-19 can release cytokines, resulting in complications that include increasing insulin resistance and acute (stress) hyperglycemia.8 SARS-CoV-2 can also infect and replicate in cells of the pancreas, resulting in decreased insulin-secretory granules in β-cells and impaired glucose-stimulated insulin secretion, which can lead to de novo development of diabetes.8 There is also evolving research suggesting that the virus may cause angiotensin converting enzyme 2–dependent damage to pancreatic islet cells and exocrine tissue, exacerbating hyperglycemia and its associated effect on the immune response.6,7 Additionally, after publication of the RECOVERY trial,9 dexamethasone became the standard treatment for patients with COVID-19 admitted to the ICU, with its adverse effect of hyperglycemia adding yet another layer of complexity.
Although the true relationship between hyperglycemia and SARS-CoV-2 infection in this patient population is unknown—that is, does the virus cause worsening glycemic control or does poor glycemic control impair the host’s immune response to the virus, or rather do patients experience a combination of these 2 effects?—emerging evidence shows that hyperglycemia during hospitalization for SARS-CoV-2 infection is associated with an increase in length of stay and mortality.5,10–13
Since the beginning of the pandemic, several retrospective cohort studies have demonstrated an association between hyperglycemia and mortality.5,10–13 In their propensity-matched analysis of 500 patients, Zhu and others5 found an adjusted hazard ratio (HR) for mortality of 0.13 (95% confidence interval [CI] 0.04–0.44, p < 0.001), as well as a lower risk of acute respiratory distress syndrome, cardiovascular injury, acute kidney injury, shock, and disseminated intravascular coagulation in patients whose blood glucose was maintained between 3.9 and 10 mmol/L, relative to those whose glucose ranged from 3.9 to greater than 10 mmol/L. Bode and others,10 in their retrospective review involving 1122 patients, also found an increased risk of death among patients with type 2 diabetes who had hyperglycemia relative to those who did not have hyperglycemia (41% vs 14.8%, p < 0.001). Patients who had hyperglycemia also had an increased length of hospital stay (5.7 vs 4.3 days, p < 0.001).10 Mazori and others12 conducted a retrospective, single-centre study of 133 patients admitted to the ICU in which nondiabetic patients with early-onset hyperglycemia (> 10 mmol/L during the first 2 calendar days after ICU admission) had a higher risk of death at 14 days (HR 7.51, 95% CI 1.70–33.24) and 60 days (HR 6.97, CI 1.86–26.13) than nondiabetic patients who did not have early-onset hyperglycemia. Most recently, Krishnasamy and others13 conducted a multicentre retrospective study of 1645 patients hospitalized with SARS-CoV-2 pneumonia in which patients with elevated glycated hemoglobin A1c (HbA1c) (> 6.5%) and/or hyperglycemia on admission (> 11.1 mmol/L) were at increased risk of acute in-hospital cardiovascular events (odds ratio [OR] 1.73, 95% CI 1.07–2.80), ICU admission (OR 1.61, 95% CI 1.10–2.34), and death (OR 1.77, 95% CI 1.02–3.07) relative to patients with or without type 2 diabetes who did not have hyperglycemia on admission.13 As such, current evidence suggests that hyperglycemia is likely associated with poor outcomes in critically ill patients with COVID-19, irrespective of a prior history of diabetes.
As we move into a new era of SARS-CoV-2 being part of the usual constellation of respiratory viruses that can lead to critical illness, and given emerging evidence of the impact of hyperglycemia on the morbidity and mortality of patients with SARS-CoV-2 infection, this study was undertaken to systematically understand the level of glycemic control achieved with different antihyperglycemic regimens. Subgroup analyses were also conducted to further describe glycemic control in patients for whom antihyperglycemic therapy was clinically indicated, as well as in patients who were and were not receiving corticosteroids for the management of SARS-CoV-2 infection.
We conducted a retrospective cohort study with a prespecified convenience sample of 120 consecutive mechanically ventilated adult patients (> 18 years of age) admitted to the Surrey Memorial Hospital ICU with a positive result on reverse transcription polymerase chain reaction testing for SARS-CoV-2 between February 1, 2020, and December 31, 2021. Patients receiving concurrent treatment for diabetic ketoacidosis were excluded. Data were collected from each patient’s electronic medical record (Meditech software system) from day 1 to day 14 of the ICU admission (or until ICU discharge, if length of stay in the ICU was less than 14 days). Blood glucose values were documented for 4 time points within each 24-hour period (at 0600, 1200, 1800, 2359). Where no blood glucose measurement was documented for a specified time point, the value obtained closest to that time was documented; where there were 2 blood glucose measurements documented at times equidistant from the specified time, the value from the earlier time point was documented. All hypoglycemia events (blood glucose < 4 mmol/L) were recorded, regardless of time of occurrence, and multiple events could be counted within a 24-hour period.
Blood glucose measurements were collected from the bedside point-of-care testing records completed by critical care nurses, laboratory (chemistry) results, medication administration records (MARs), and blood glucose/insulin flow sheets (nursing notes). The measurements were then grouped into 4 categories: 4–10 mmol/L (within target range), 10–12.5 mmol/L, 12.6–15 mmol/L, and greater than 15 mmol/L. Information about the medication regimen was collected from the MAR, and the regimens were grouped into 9 categories. Patients could be on different regimens at different times during their critical care stay; therefore, the baseline characteristics and clinical outcomes were described for the full cohort, not subdivided according to regimen. The use of glucocorticoid medications (dexamethasone, methylprednisolone, hydrocortisone, prednisone, and prednisolone) was also documented from ICU day 1 to day 14 (or until ICU discharge). Additional baseline demographic data collected were age, gender, medical diagnoses, APACHE II score on admission to critical care, need for and duration of mechanical ventilation, length of stay in the ICU and in hospital, and in-hospital death.
Data were collected and recorded by one investigator (E.B) using a standardized data collection form. Ten percent of the charts from which data were collected were checked by the co-investigators (S.N.S. and K.S.) to ensure inter-rater reliability.
Descriptive statistical methods were used to analyze the baseline demographic data, blood glucose measurements, hypoglycemia events, hospital and ICU length of stay, and in-hospital death for the entire cohort. Results are reported as medians and interquartile ranges (IQRs) or means and standard deviations for continuous variables and as percentages for categorical variables.
A subgroup analysis was performed of patients for whom antihyperglycemic therapy was clinically indicated. Patients were excluded from the subgroup analysis if they did not require antihyperglycemic medication during their ICU stay or received only sliding-scale subcutaneous (SC) insulin therapy with no blood glucose readings above 10 mmol/L at any time. In this subgroup analysis, a χ2 test of independence was performed to examine the relation between the medication regimen used (IV insulin–based, SC sliding-scale insulin, basal SC insulin with or without SC bolus or sliding-scale insulin, or oral medication regimens with or without insulin) and the level of glycemic control (4–10 mmol/L [within target range], 10–12.5 mmol/L, 12.6–15 mmol/L, and > 15 mmol/L) for patients with an indication for ongoing insulin therapy.
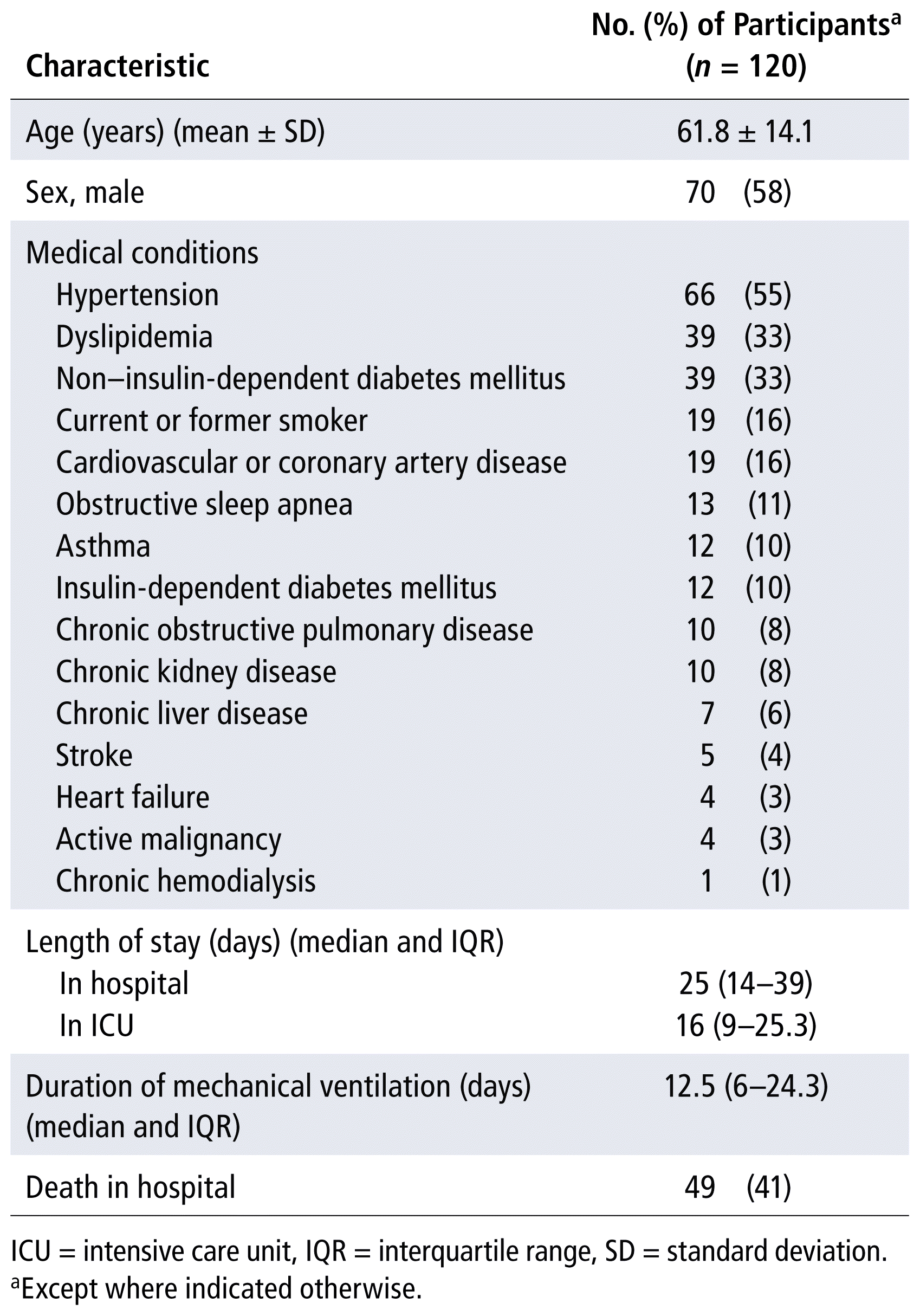
The baseline characteristics of the cohort (n = 120) are reported in Table 1. Of note, 43% of participants had diabetes mellitus (and 10% were dependent on insulin). The median length of stay in the ICU was 16 (IQR 9–25.3) days, and the median duration of mechanical ventilation was 12.5 (IQR 6–24.3) days.
TABLE 1 Characteristics and Outcomes of Study Participants

In the overall cohort, 45% (n = 2116/4673) of glucose measurements fell within the target range of 4–10 mmol/L. The 2 regimens incorporating IV insulin infusion maintained glucose values within the target range more often than any other regimen (59% and 60%, respectively, of measured values were between 4 and 10 mmol/L) (Table 2), whereas regimens incorporating oral hypoglycemic medications (combined with sliding-scale insulin with or without basal insulin) had only 12% and 29% of values, respectively, falling within the target range (Table 2). Additional data stratified by medication regimen and level of blood glucose control are outlined in Table 2.
TABLE 2 Blood Glucose Measurements, Stratified by Medication Regimena
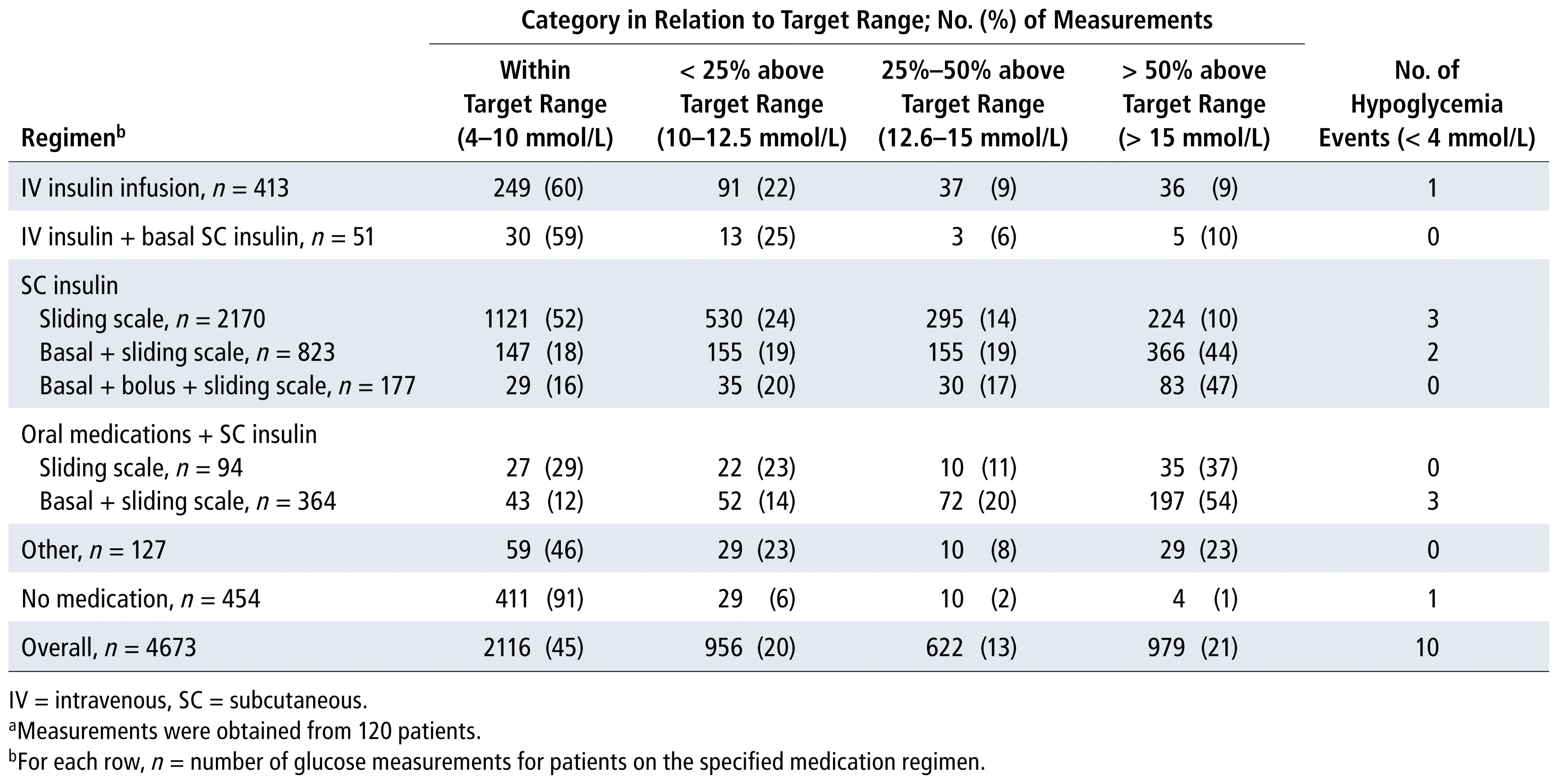
In total, 979 (21%) of the blood glucose measurements were greater than 15 mmol/L (Table 2). Of these, only 36 (less than 1% of all measurements) occurred in patients receiving IV insulin compared with 646 (14%) in patients receiving basal SC insulin-based regimens (with or without SC bolus insulin, sliding-scale insulin, or oral antihyperglycemic medications).
In the subgroup of 73 patients for whom antihyperglycemic therapy was clinically indicated, 32% (n = 917/2872) of glucose measurements fell within the target range of 4 to 10 mmol/L, again with 59% and 60% of readings, respectively, falling within the target range among patients receiving either of the 2 IV insulin infusion regimens (Table 3). In this subgroup, the proportion of patients who had any reading greater than 15 mmol/L was lower in the groups receiving IV insulin infusion (7% and 10%, respectively) than in the groups for all other regimens (20%–54%).
TABLE 3 Blood Glucose Measurements in Subgroup of Patients with Indication for Antihyperglycemic Therapy, Stratified by Medication Regimena
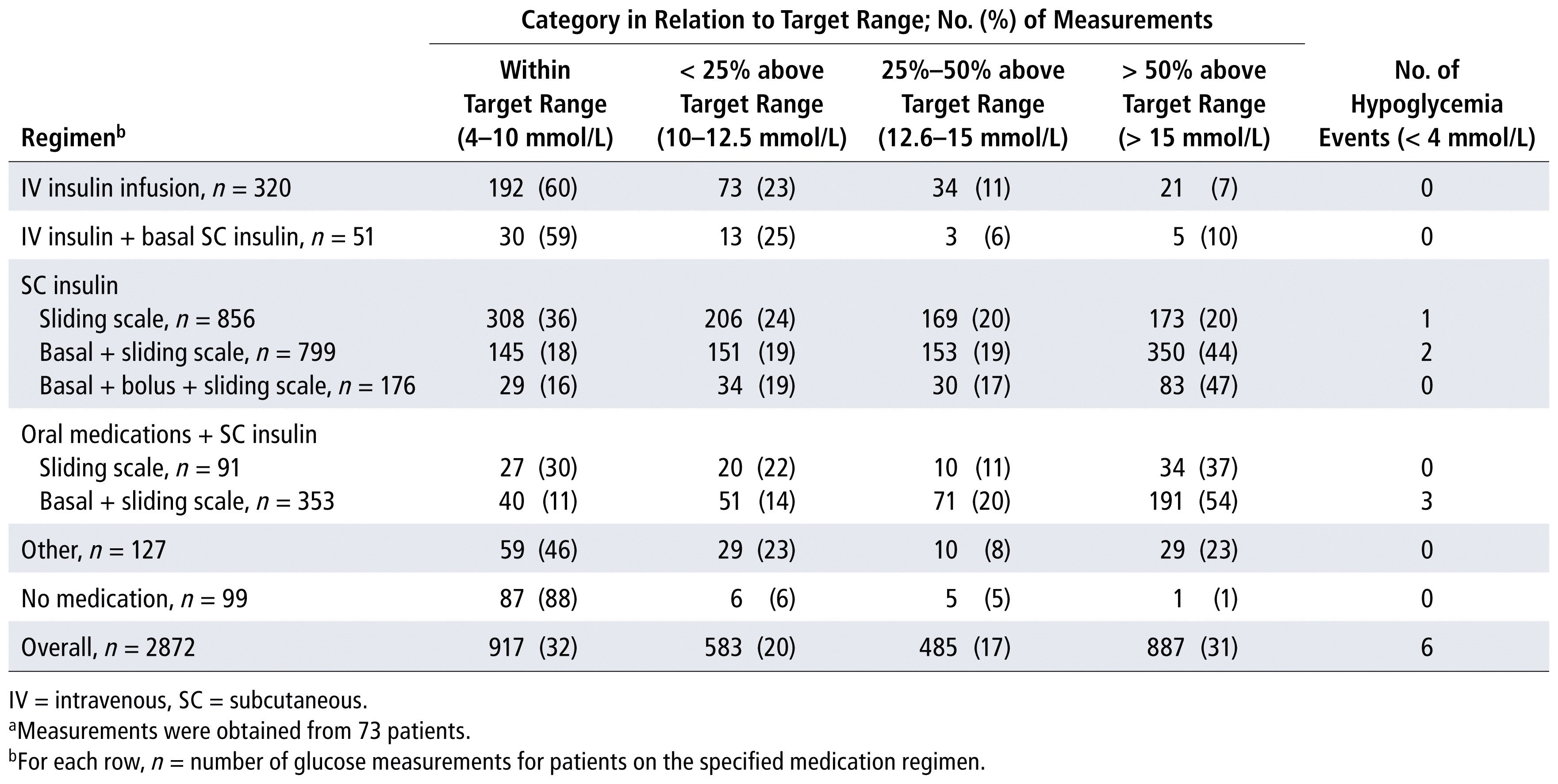
The χ2 test to examine the relationship between glycemic control and the various medication regimens in the subgroup of patients with a clinical indication for regimens versus SC basal insulin–based regimens with SC sliding-scale insulin, with or without short-acting SC bolus insulin, χ2 = 286.4 (df = 3, p < 0.001); for IV insulin based–regimens versus oral-based medication regimens with SC sliding-scale insulin, with or without SC basal insulin, χ2 = 254.3 (df = 3, p < 0.001). When the oral therapy– based medication regimens with SC sliding-scale insulin (with or without SC basal insulin) were compared with SC basal insulin–based regimens with SC sliding-scale insulin (with or without short-acting SC bolus insulin), the association between the medication regimen and level of glycemic control was not significant: χ2 = 5.5 (df = 3, p = 0.14).
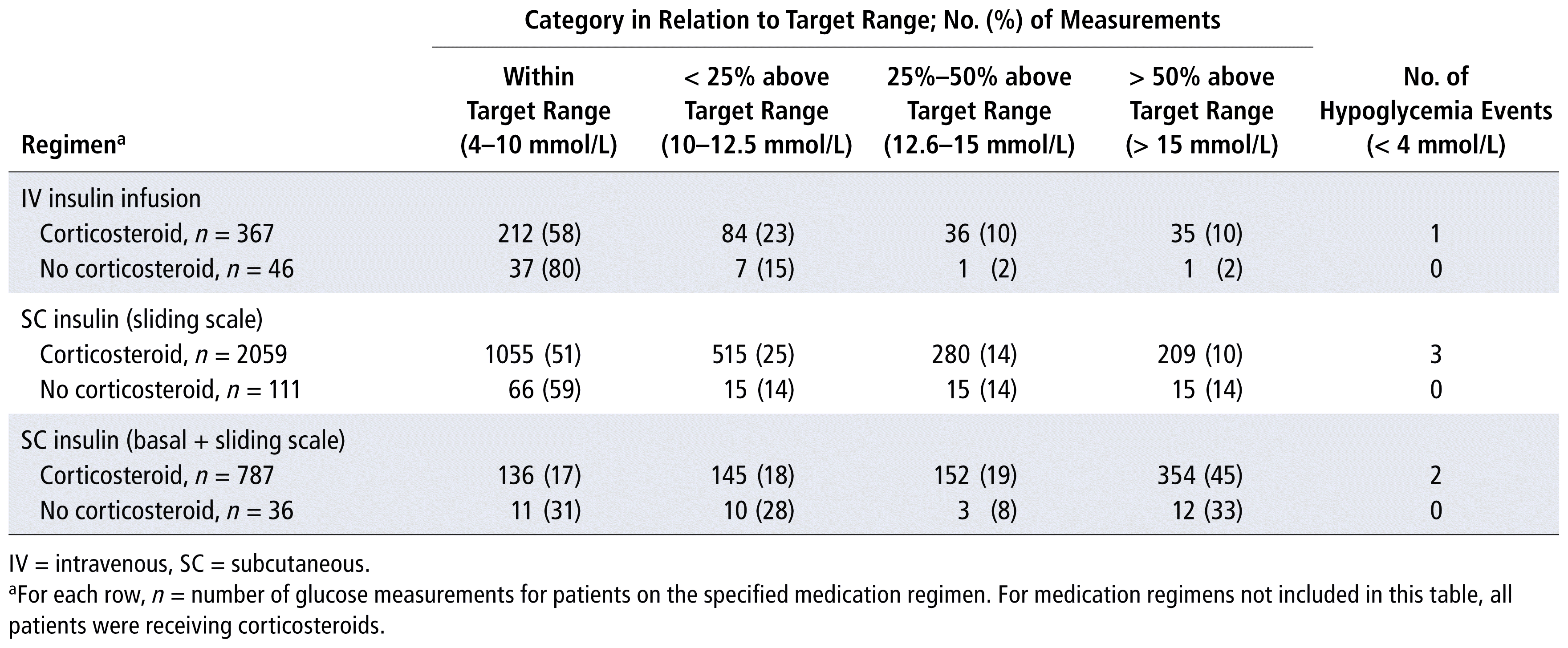
When glycemic control was compared between patients who did and did not receive corticosteroid therapy, it was evident that glucose was less well controlled among patients who were receiving concomitant glucocorticoid therapy (Table 4). Among patients receiving IV insulin infusion without corticosteroids, 80% (n = 37/46) of measured glucose values were within the target range, whereas in the presence of corticosteroids, this proportion dropped to 58% (n = 212/367). The same trend was observed across other regimens (Table 4).
TABLE 4 Blood Glucose Measurements in Presence or Absence of Corticosteroid Therapy, Stratified by Medication Regimen
Overall, 8 (7%) of the patients experienced at least 1 hypoglycemia event, defined as blood glucose less than 4 mmol/L. A total of 10 hypoglycemia events occurred during this study, the majority when patients had no enteral or parenteral nutrition or had poor or intermittent nutritional intake. Only 1 hypoglycemia event occurred while a patient was receiving goal-rate enteral nutrition. Of the 8 patients who experienced a hypoglycemia event, 6 died before discharge from hospital.
In this study, IV insulin–containing regimens reliably controlled blood glucose and managed hyperglycemia more consistently than alternative insulin therapies in patients infected with SARS-CoV-2. After exclusion of patients who had no indication for insulin-based therapies (i.e., those who required no antihyperglycemic therapies or received SC sliding-scale insulin with no blood glucose readings above 10 mmol/L), the results were even more pronounced, with IV insulin–based therapies significantly outperforming the alternative regimens. Across all regimens, better glycemic control was maintained when corticosteroids were not administered. The incidence of hypoglycemia events was low, with the majority occurring when non-IV insulin–based alternative therapies were used and in patients who were not receiving adequate nutrition.
To our knowledge, we are the first to report findings of a comparison between IV insulin–containing regimens and alternative antihyperglycemic therapies in critically ill patients with COVID-19.
Our results are consistent with those of studies comparing these regimens in critically ill patients without COVID-19 pneumonia. Tran and others14 demonstrated that ICU patients treated with IV insulin regimens had better blood glucose control than those treated with SC insulin regimens (63% vs 45.7%). They also found that hypoglycemia events were more common among patients receiving SC regimens than among those receiving IV insulin.14 Furthermore, IV insulin has been traditionally used and studied in large randomized controlled trials investigating ideal blood glucose targets in critically ill patients.2,15,16
Diabetes and uncontrolled hyperglycemia have been recognized as risk factors for poor outcomes in patients with COVID-19, including severe illness and death.17–19 In addition, a large population-based cohort study showed that SARS-CoV-2 infection was associated with a higher risk of diabetes among patients not known to have a previous diagnosis of this condition.20 While there is clearly an interplay between infection with SARS-CoV-2 and glycemic control, the impact of tight versus liberal glycemic targets on COVID-19–related morbidity and mortality remains unknown.
We found no studies that used alternative antihyperglycemic therapies, such as SC insulin and/or oral antihyperglycemic regimens, for management of hyperglycemia in patients with COVID-19. However, some centres have investigated continuous glucose monitoring as a means of minimizing the need for point-of-care testing, nursing and staff resources, PPE, and exposure to infection while maintaining the use of IV insulin.21
Given the retrospective and observational nature of this study, we are unable to draw conclusions about a possible association or causation between glycemic control and clinical outcomes. While we were able to demonstrate that alternative treatment strategies were suboptimal for surrogate outcomes (moderate and severe hyperglycemia and hypoglycemia), it is unknown if these patients experienced worse clinical outcomes than they otherwise would have if they had received IV insulin and thereby experienced better glycemic control. We were unable to compare clinical outcomes across regimens, as there was substantial crossover in the medication regimens used by individual patients.
Another limitation of this study is our inability to generalize to current practice, because certain variants of SARS-CoV-2 have lower disease severity, many patients and health care providers are now vaccinated, and better resource supplies now exist. However, there remains a large proportion of patients admitted to hospitals with COVID-19 pneumonia22 and non-IV insulin strategies are still used to minimize staff exposure to the virus because of momentum gained from this strategy early in the pandemic. Furthermore, should a future pandemic arise, our study highlights that IV insulin can remain the standard of care for management of hyperglycemia in critically ill patients. Instead, alternative methods should be explored to minimize exposure and preserve PPE.
In critically ill patients, both tight glycemic control (and the associated increase in risk of hypoglycemia) and moderate-to-severe hyperglycemia may be associated with an increased risk of death.2 There is also growing evidence to suggest that wide variation in blood glucose levels may have detrimental effects on patient outcomes.23 However, the optimal target for each individual patient remains unknown and may differ according to whether the patient is experiencing acute or chronic hyperglycemia. Although this study evaluated only patients who tested positive for SARS-CoV-2, the results do improve our understanding of the impact that various medication regimens have on glycemic control and provides evidence supporting return to the usual standards of care for patients presenting to the ICU with COVID-19. In addition, even though we cannot generalize the results of this study to critically ill patients without COVID-19, our findings give a more systematic understanding of the effect that this seemingly benign change in the route of medication administration had on patients. This enhanced understanding will help to inform therapeutic decision-making as we face future pandemics and other situations involving resource limitations (in terms of both staffing and equipment).
COVID-19 has caused several challenges in caring for critically ill patients, including the maintenance of glycemic control. Although we found that many different strategies were employed to manage blood glucose in our study sample, IV insulin infusion provided the best glycemic control for these patients. Understanding the impact of the rapid changes we made while managing care for patients infected with a novel virus will allow us to be better prepared for managing the therapeutic challenges we will face in future pandemics. Future studies should include prospective and randomized trials assessing clinical outcomes in patients with and without COVID-19 whose hyperglycemia is managed with IV insulin relative to other treatment alternatives, as well as evaluating alternatives to point-of-care testing (e.g., continuous glucose monitoring) to decrease requirements for nurses’ time, PPE needs, and exposure of health care workers to infectious pathogens.
1 Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–9.
Crossref PubMed PMC
2 NICE-SUGAR Study Investigators; Finfer S, Chittock DR, Su SYS, Blair D, Foster D, Dhingra V, et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283–97.
Crossref PubMed
3 Griesdale DEG, de Souza RJ, van Dam RM, Heyland DK, Cook DJ, Malhotra A, et al. Intensive insulin therapy and mortality among critically ill patients: a meta-analysis including NICE-SUGAR study data. CMAJ. 2009;180(8):821–7.
Crossref PubMed PMC
4 Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock 2021. Crit Care Med. 2021; 49(11):e1063–e1143.
Crossref PubMed
5 Zhu L, She ZG, Cheng X, Qin JJ, Zhang XJ, Cai J, et al. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;31(6):1068–77.e3.
Crossref PubMed PMC
6 Erener S. Diabetes, infection risk and COVID-19. Mol Metab. 2020; 39:101044.
Crossref PubMed PMC
7 Somasundaram NP, Ranathunga I, Ratnasamy V, Wijewickrama PSA, Dissanayake HA, Yogendranathan N, et al. The impact of SARS-Cov-2 virus infection on the endocrine system. J Endocr Soc. 2020;4(8):bvaa082.
Crossref PubMed PMC
8 Kethireddy R, Gandhi D, Kichloo A, Patel L. Challenges in hyperglycemia management in critically ill patients with COVID-19. World J Crit Care Med. 2022;11(4):219–27.
Crossref PubMed PMC
9 Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, etal. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021; 384(8):693–704.
Crossref
10 Bode B, Garrett V, Messler J, McFarland R, Crowe J, Booth R, et al. Glycemic characteristics and clinical outcomes of COVID-19 patients hospitalized in the United States. J Diabetes Sci Technol. 2020; 14(4):813–21.
Crossref PubMed PMC
11 Sardu C, D’Onofrio N, Balestrieri ML, Barbieri M, Rizzo MR, Messina V, et al. Outcomes in patients with hyperglycemia affected by COVID-19: can we do more on glycemic control? Diabetes Care. 2020; 43(7):1408–15.
Crossref PubMed PMC
12 Mazori AY, Bass IR, Chan L, Mathews KS, Altman DR, Saha A, et al. Hyperglycemia is associated with increased mortality in critically ill patients with COVID-19. Endocr Pract. 2021;27(2):95–100.
Crossref PubMed PMC
13 Krishnasamy S, Sheikh D, Ali T, Clemons V, Furmanek S, Mohamed Fawzy Abdelhaleem AA, et al. Impact of hyperglycemia on cardiovascular events and clinical outcomes in patients hospitalized with COVID-19 pneumonia. Endocr Pract. 2022;28(8):780–6.
Crossref PubMed PMC
14 Tran KK, Kibert JL 2nd, Telford ED, Franck AJ. Intravenous insulin infusion protocol compared with subcutaneous insulin for the management of hyperglycemia in critically ill adults. Ann Pharmacother. 2019; 53(9):894–8.
Crossref PubMed
15 Van den Berghe G, Wilmer A, Hermans G, Meersseman W, Wouters PJ, Milants I, et al. Intensive insulin therapy in the medical ICU. N Engl J Med. 2006;354(5):449–61.
Crossref PubMed
16 Van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345(19):1359–67.
Crossref
17 Guo W, Li M, Dong Y, Zhou H, Zhang Z, Tian C, et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020;36(7):e3319.
Crossref PubMed PMC
18 Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–62.
Crossref PubMed PMC
19 Rawshani A, Kjölhede EA, Rawshani A, Sattar N, Eeg-Olofsson K, Adiels M, et al. Severe COVID-19 in people with type 1 and type 2 diabetes in Sweden: a nationwide retrospective cohort study. Lancet Reg Health Eur. 2021;4:100105.
Crossref PubMed PMC
20 Naveed Z, Velásquez García HA, Wong S, Wilton J, McKee G, Mahmood B, et al. Association of COVID-19 infection with incident diabetes. JAMA Netw Open. 2023;6(4):e238866.
Crossref PubMed PMC
21 Faulds ER, Boutsicaris A, Sumner L, Jones L, McNett M, Smetana KS, et al. Use of continuous glucose monitor in critically ill COVID-19 patients requiring insulin infusion: an observational study. J Clin Endocrinol Metab. 2021;106(10):e4007–e4016.
Crossref PubMed
22 BC Centre for Disease Control. Respiratory virus data. Provincial Health Services Authority; 2024 [cited 2024 Mar 6]. Available from: http://www.bccdc.ca/health-professionals/data-reports/respiratory-virus-data#Reports
23 Eslami S, Taherzadeh Z, Schultz MJ, Abu-Hanna A. Glucose variability measures and their effect on mortality: a systematic review. Intensive Care Med. 2011;37(4):583–93.
Crossref PubMed PMC
Address correspondence to: Dr Sarah N Stabler, Surrey Memorial Hospital, 13750 96 Avenue, Surrey BC V3V 1Z2, email: sarah.stabler@fraserhealth.ca
Competing interests: For work not directly related to the study reported here, Kieran Shah received payment for teaching from the Columbia Paramedic Academy and has served in a non-remunerated role with the CSHP Advocacy Committee; and Sarah Stabler has received payment from the University of British Columbia for presentation of lectures. No other competing interests were declared.
Funding: None received.
Submitted: October 24, 2023
Accepted: May 30, 2024
Published: October 9, 2024
© 2024 Canadian Society of Hospital Pharmacists | Société canadienne des pharmaciens d’hôpitaux
Canadian Journal of Hospital Pharmacy, VOLUME 77, NUMBER 4, 2024