Chinonyerem O Iheanacho and Valentine U OdiliTo cite: Iheanacho CO, Odili VU. Adverse drug reactions and predictors of medication adherence in patients with prostate cancer. Can J Hosp Pharm. 2024;77(4):e3567. doi: 10.4212/cjhp.3567
ABSTRACT
Background
Adherence to therapy with prostate cancer medicines is critical for delaying the progression of disease and enhancing health outcomes.
Objectives
To determine patients’ medication adherence, the predictors of adherence, and the frequency and types of adverse drug reactions (ADRs) in persons with prostate cancer.
Methods
A serial entry-point cross-sectional study of patients with prostate cancer was conducted in 3 cancer hospitals in Nigeria over a 12-month period (January 7, 2022, to January 3, 2023). Data on medication adherence were self-reported by patients, and data on ADRs were obtained from hospital records. Descriptive and inferential statistical analyses were performed, and p less than 0.05 was considered statistically significant.
Results
Of the 133 study participants, most 112 (84.2%) reported high medication adherence. The cost of drugs was the most frequently reported potential barrier to adherence (n = 63, 47.4%). Adherence was significantly dependent on family history of cancer (df = 3, F = 4.557, p = 0.005) and health-related quality of life (HRQOL) (ß = 0.275, T = 2.170, p = 0.032) but not illness perception (ß = 0.046, T = 0.360, p = 0.72). Adverse events were observed in 36 participants (27.1%) and were deemed to be “possible ADRs” (n = 19, 53%) or “probable ADRs” (n = 17, 47%); all were nonpreventable and expected (100%), and most (n = 31, 86%) were within the level 1 category of severity. Loss of erection and low libido was the most frequently reported ADR (n = 14, 39%).
Conclusions
In this study, medication adherence was high, with cost being a potential barrier to adherence. Family history of cancer and HRQOL significantly predicted medication adherence. The medications were well tolerated, and observed ADRs had minor severity. Policies targeting the reduction of cost-related factors for prostate cancer medications are essential.
KEYWORDS: Nigeria, medication adherence, adverse drug reaction, illness perception, quality of life, prostate cancer
RÉSUMÉ
Contexte
L’observance thérapeutique quant à la prise de médicaments contre le cancer de la prostate est essentielle pour retarder la progression de la maladie et améliorer les résultats en matière de santé.
Objectifs
Déterminer l’observance thérapeutique des patients, les facteurs prédictifs de l’observance ainsi que la fréquence et les types d’effets indésirables du médicament chez les personnes atteintes d’un cancer de la prostate.
Méthodologie
Une étude transversale en série a été menée auprès de patients atteints d’un cancer de la prostate dans trois hôpitaux spécialisés dans le traitement du cancer au Nigéria sur une période de 12 mois (du 7 janvier 2022 au 3 janvier 2023). Les patients ont eux-mêmes consigné les données relatives à l’observance thérapeutique et les données concernant les effets indésirables ont été obtenues à partir des dossiers de l’hôpital. Des analyses statistiques descriptives et inférentielles ont été réalisées, où une valeur de p inférieure à 0,05 était considérée comme significative d’un point de vue statistique.
Résultats
Des 133 participants à l’étude, la plupart (112, 84,2 %) ont déclaré une forte observance thérapeutique. Le coût des médicaments était l’obstacle potentiel à l’observance le plus fréquemment invoqué (n = 63, 47,4 %). L’observance dépendait significativement des antécédents familiaux de cancer (df = 3, F = 4,557, p = 0,005) et de la qualité de vie liée à la santé (QVLS) (ß = 0,275, T = 2,170, p = 0,032), mais pas de la perception de la maladie (ß = 0,046, T = 0,360, p = 0,72). Des événements indésirables ont été observés chez 36 participants (27,1 %) et ont été considérés comme des « effets indésirables du médicament possibles » (n = 19, 53 %) ou des « effets indésirables du médicament probables » (n = 17, 47 %); tous étaient inévitables et attendus (100 %), et la plupart (n = 31, 86 %) relevaient de la catégorie de gravité de niveau 1. La perte d’érection et la faible libido étaient les effets indésirables les plus fréquemment signalés (n = 14, 39 %).
Conclusions
Dans cette étude, l’adhésion au régime médicamenteux était élevée, le coût étant un obstacle potentiel à l’observance. Les antécédents familiaux de cancer et la QVLS prédisaient de manière significative l’observance thérapeutique. Les médicaments étaient bien tolérés et les effets indésirables du médicament observés n’étaient pas fort graves. Il est essentiel de mettre en place des politiques visant à réduire les facteurs liés au coût des médicaments contre le cancer de la prostate.
Mots-clés: Nigéria, observance thérapeutique, réaction indésirable aux médicaments, perception de la maladie, qualité de vie, cancer de la prostate
Prostate cancer is a leading type of cancer in males in several regions of the world and an important cause of death, particularly among men of African descent.1 It is the most frequently diagnosed cancer among Nigerian males, accounting for 12.3% of all new cases of cancer in the country and 29.8% of all new cancers in males in 2020.2 Also in 2020, a prostate cancer–specific mortality of 10.8% and a cumulative risk of 2.44% were recorded among Nigerian males, with a 5-year prevalence of 21.27% per 100 000.2 As a hormone-responsive disease, prostate cancer is mostly managed through androgen deprivation therapies, which comprise both oral and parenteral medications that reduce testosterone to castration levels in affected men.3 Adherence to prostate cancer therapy is a critical requirement for delay of disease progression, effective palliative care, and enhancement of health outcomes.4 Therefore, knowledge of medication adherence and its potential barriers is essential for optimization of drug therapy. At the same time, various factors, such as illness perception, socioeconomic circumstances, complexity of medicines, and comorbidities, have been previously identified to have a significant impact on medication adherence.5,6 One previous study reported a high rate of medication nonadherence among patients with prostate cancer.4 Medication side effects and potential adverse drug reactions (ADRs) are also important factors influencing medication adherence.
Globally, ADRs are a major cause of morbidity, hospital admission, and mortality. They represent a clinically significant burden with high incidence and prevalence. A previous study reported that 1 in 9 emergency hospital visits were the result of an ADR.7 Several ADRs have been reported among cancer patients,8,9 and the extent of their occurrence may have an impact on quality of life among those affected. ADRs may be mild, moderate, or severe, and various forms have been previously reported among persons with prostate cancer in various regions of the globe.8,10–12 It is essential to recognize ADRs and to establish a causal relationship between the drug and the adverse events. Therefore, active monitoring of drug therapy is an important tool in timely detection, assessment, and early management of ADRs in patients undergoing management of prostate cancer.13 Furthermore, adequate understanding of ADRs and their pattern of occurrence in various geographic regions would enhance patient care.
Medication adherence and ADRs have received limited research attention and have not yet been fully explored in persons with prostate cancer, yet such knowledge is essential to guide intervention strategies. Therefore, the findings from this study are anticipated to contribute to baseline studies regarding interventions and treatment plans. Although the study was conducted in Africa’s most populous nation, Nigeria, its findings may be extrapolated to other clinical settings, particularly other low- and medium-income countries. Notably, poor medication adherence and ADRs have been associated with significantly prolonged hospital stays in Nigeria and increased cost of care for patients.14 Meanwhile, there has been limited study in Nigeria on hospital-based assessment of ADRs in persons with prostate cancer and the responsible medications. Certain ADRs are preventable, and as such, collection and assessment of these data are essential for health care planning, budgeting, policy formation, and development of treatment protocols to enable appropriate, timely, and optimal patient care.7 In this study, we assessed patients’ reported medication adherence, predictors of and barriers to medication adherence, occurrence of ADRs, and the pattern of ADRs in persons with a diagnosis of prostate cancer.
This serial entry-point, cross-sectional study involved patients with prostate cancer recruited from the 3 cancer reference hospitals in Cross River State, Nigeria: Asi Ukpo Hospital, University of Calabar Teaching Hospital, and the Nigerian Navy Reference Hospital.
The study population comprised persons with prostate cancer who were treated at the included health care facilities. All patients with this diagnosis who visited the health care facilities between January 7, 2022, and January 3, 2023, were eligible for the study.
Patients who had been on drug therapy for at least 1 day were included in the evaluation of ADRs, whereas patients who had been receiving drug therapy for at least 3 months were included in the assessment of medication adherence. Patients who declined consent and those who were too weak to speak were excluded.
A medication adherence questionnaire was developed through a structured qualitative session, which took the form of a focus group discussion involving persons with prostate cancer. The newly developed survey items generated from this discussion were submitted to experts for review and administered to selected members of the population of interest for debriefing. This approach was used to evaluate the quality of responses and to determine if the questions developed would generate the required information about medication adherence.
Assessment of face and content validity of the medication adherence data collection tool was performed by 3 clinical pharmacists. The content of the tool was assessed for validity by pretesting, which involved 15 selected respondents who were not included in the study. The results of this pretest showed adequate comprehension of the questions by the participants. The study tool was also tested for reliability using the Cronbach α,15 for which results are interpreted as follows: if α is less than 0.5, reliability is unacceptable; if α lies between 0.5 and less than 0.6, reliability is poor; if α lies between 0.6 and less than 0.7, reliability is questionable; if α lies between 0.7 and less than 0.8, reliability is acceptable; if α lies between 0.8 and less than 0.9, reliability is good; and if α is 0.9 or above, reliability is excellent.
Medication adherence, the extent to which a patient follows the medication regimen, was assessed by patients’ self-reporting through the self-completed questionnaire, which was anchored on a Yes/No response scale, except for the section on sociodemographic characteristics. The questionnaire was divided into 2 sections; in the first section, we collected information about patients’ sociodemographic characteristics, and in the second section we gathered data on adherence to prescribed medications (7 questions) and barriers to adherence (5 questions).
ADRs were determined following an assessment of all patients who had been receiving drug therapy for at least 1 day. Data for this determination were obtained from interactions with patients and reports in the patients’ hospital records. Appropriate proforma, developed in accordance with the hospitals’ ADR reporting forms, were used to collect data on details of drugs used for therapy, routes of administration, time of initial use of the drugs, and any pre-existing medical conditions.
Data collection was performed between January 7, 2022, and January 3, 2023. The data were obtained, during patients’ visits to the cancer centre or urology clinics in the health care facilities, through interviews and a review of corresponding hospital records. The study tools were administered in a separate office after each patient’s consultation with the physician.
The study end points were participants’ medication adherence, predictors of medication adherence, barriers to medication adherence, and the pattern of ADRs among participants. Data were cleaned and entered into SPSS software, version 25 (IBM), for descriptive and inferential analyses. Categorical data were presented as frequencies and percentages. Adherence scores were summed, with higher scores connoting higher adherence. Predictors of adherence were determined by inferential statistics. The survey questions on medication adherence were negatively worded, with each “yes” response scored as 0, and each “no” response scored as 1. The total scores ranged from 0 to 7 and were grouped into 3 levels of adherence: low (score < 4), medium (score 4 or 5), and high (score ≥ 6). The effects of selected clinical and sociodemographic characteristics on medication adherence were determined by univariate analysis of variance (F tests of equality of means), where F represents the F-ratio for the sample statistic in the analysis of variance (ANOVA). Multiple linear regression was used to determine the effect on medication adherence of illness perception and health-related quality of life (HRQOL), factors that were investigated in previous studies by our team (the first of these was published earlier in 2024,16 and the second [Iheanacho CO, Odili VU, Ekeocha RC. Health-related quality of life in prostate cancer] is currently under consideration for publication). ANOVA was performed to show the overall significance of the regression model. In our 2 earlier studies, illness perception was assessed by the previously validated brief illness perception questionnaire,17 while HRQOL was assessed by the functional assessment of cancer therapy – prostate (FACT-P) questionnaire.18
We also analyzed the observed pattern of ADRs. We first identified the drugs suspected to be involved in any adverse effects and then analyzed the potential causal association using the modified Naranjo scale.19 The preventability of ADRs and their severity were evaluated with the modified Schumock and Thornton scale20 and the modified Hartwig severity assessment scale,10 respectively. Severity describes the extent of influences exerted by the ADR on the patient’s daily activities. The Summaries of Product Characteristics (SmPCs) approved during the medicines’ marketing authorization were used to evaluate the expectedness of observed ADRs. Any ADR listed in the SmPCs was considered to be expected and vice versa.
In the context of this study, drug therapies refer to drug products that are prescribed for the management of prostate cancer in the study participants. Drugs for comorbidities refer to medicinal products used by the study participants to treat other disease conditions.
The study was conducted in accordance with the Helsinki declaration. Ethics approval was obtained from the Health and Research Ethics Committee of the University of Calabar Teaching Hospital (reference no. UCTH/HREC/33/582). Individual written informed consent was also obtained from each participant before the study.
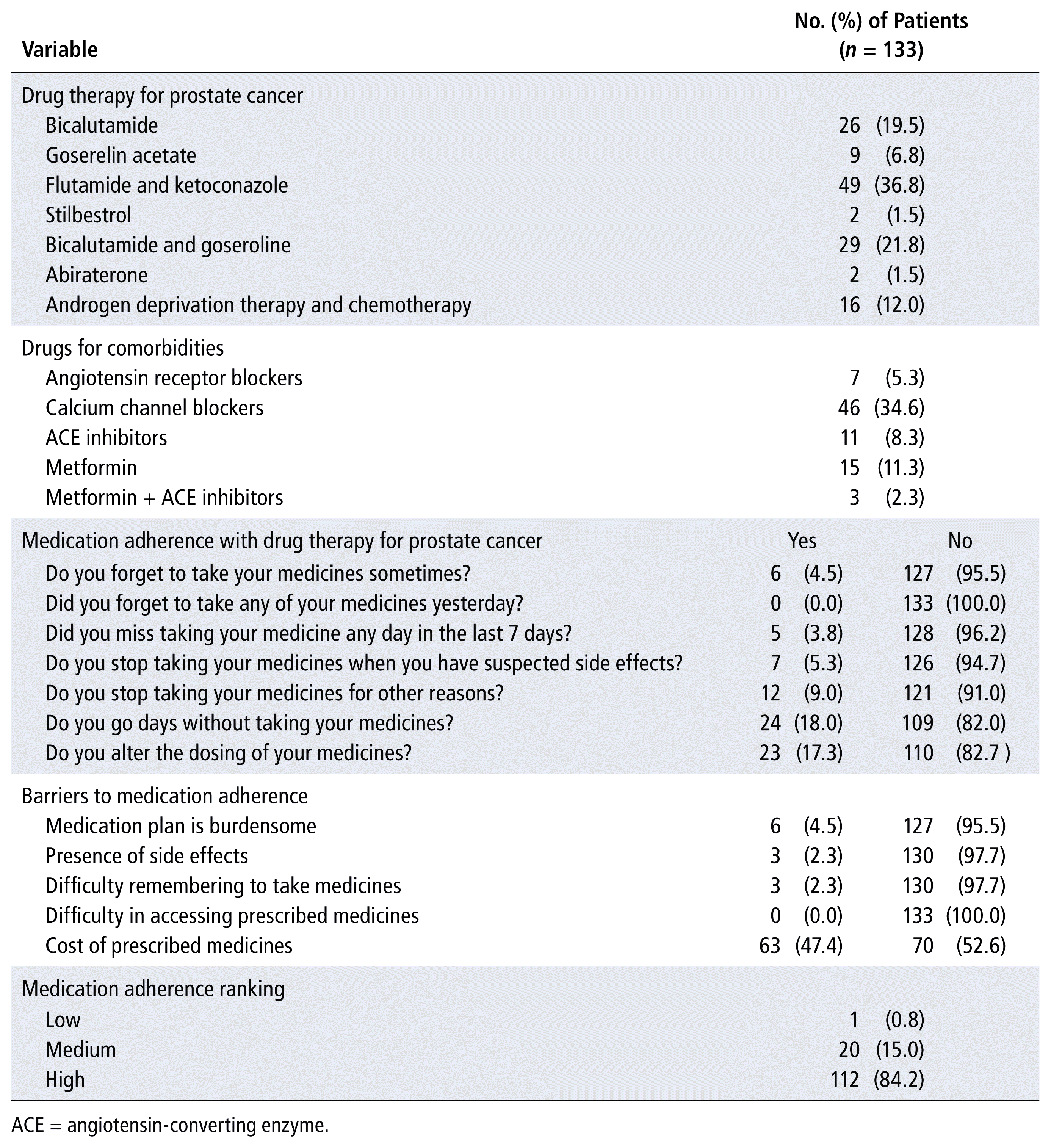
The medication adherence data collection tool had a Cronbach α of 0.72, indicating acceptable reliability. This study is a follow-up of previous studies of HRQOL (Iheanacho CO, Odili VU, Ekeocha RC. Health-related quality of life in prostate cancer. Manuscript in preparation) and the impact of a pharmacist-led intervention on illness perception among patients with prostate cancer16; the demographic characteristics of study participants are presented in those previous reports. Table 1 shows the distribution of patients’ drug therapies for prostate cancer and various comorbidities.
TABLE 1 Medications and Medication Adherence

All eligible patients (n = 133) provided consent and participated in the study. Table 1 shows that most patients (n = 112, 84.2%) had high medication adherence. Among the observed barriers to adherence, cost of drugs was reported by almost half of the participants (n = 63, 47.4%).
A family history of cancer had a significant effect on medication adherence (df = 3, F = 4.557, p = 0.005). Therefore, medication adherence was significantly dependent on the patient’s family history of cancer (Table 2).
TABLE 2 Effect of Patient Characteristics on Medication Adherence
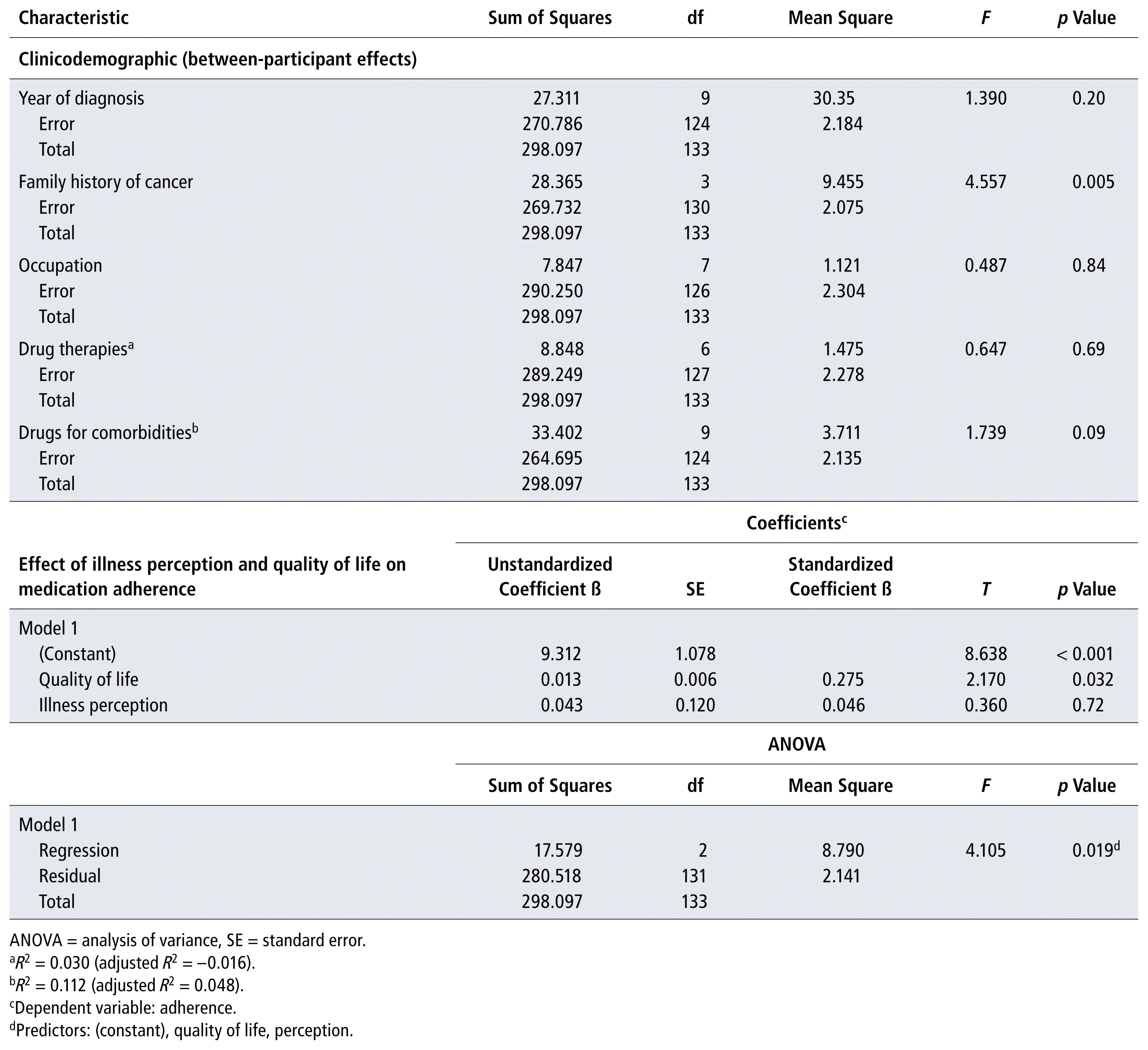
Although HRQOL significantly influenced adherence (β = 0.275, T = 2.170, p = 0.032), illness perception had an insignificant contribution (β = 0.046, T = 0.360, p = 0.72). The regression model was significant (F = 4.105, p = 0.019) and thus can be used for prediction. The same model showed that both HRQOL and illness perception were positively related to medication adherence.
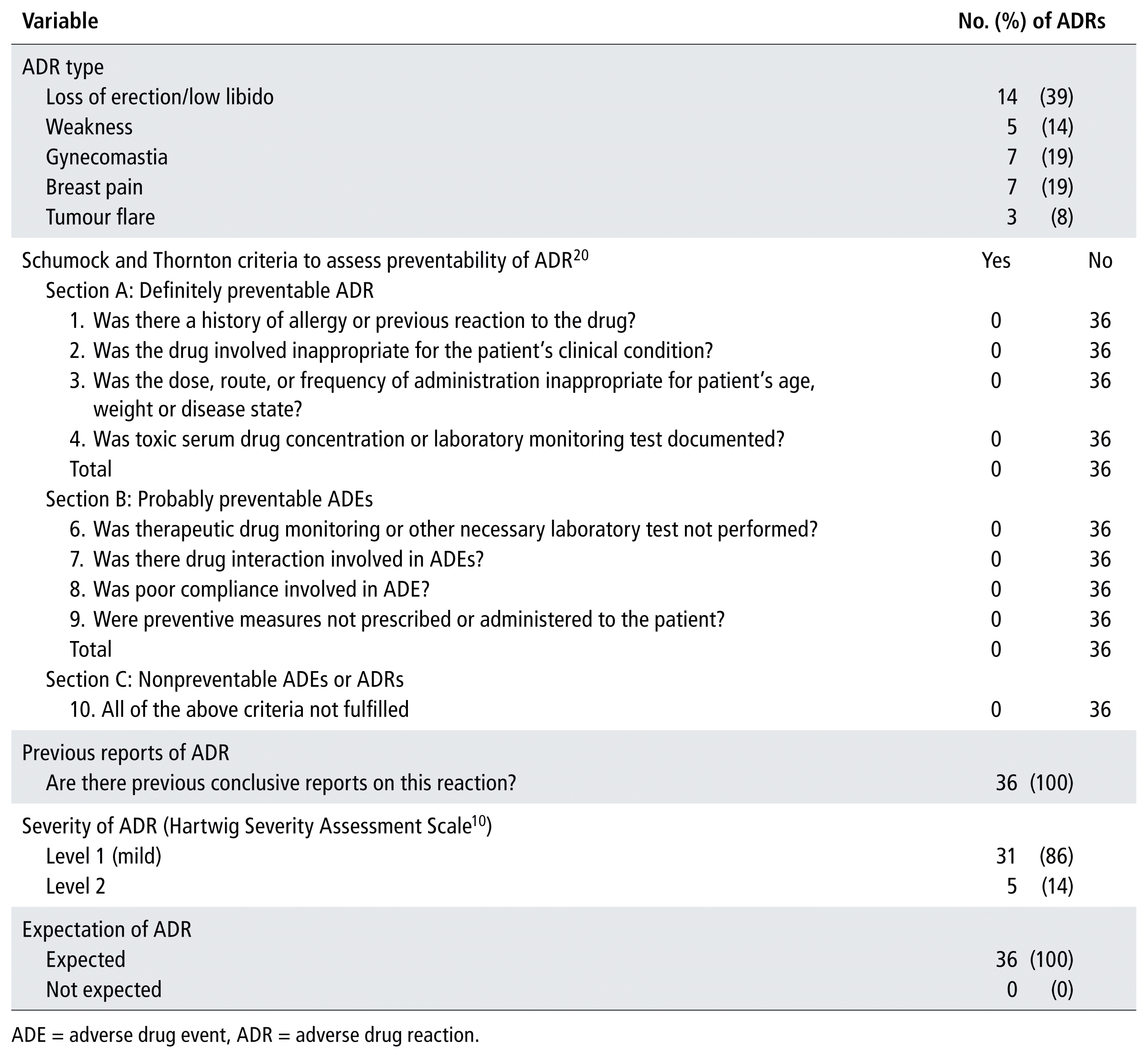
A total of 36 participants experienced an adverse event. The modified Naranjo algorithm scale for the probability of an ADR showed that ADRs probably occurred in 17 (47%) of the patients and possibly occurred in 19 (53%).
All of the observed ADRs were deemed to be nonpreventable according to the Schumock and Thornton criteria for assessment of ADR preventability.20 Overall, 31 (86%) of the observed ADRs had level 1 severity, as assessed by the Hartwig severity assessment scale (which has a total of 7 levels of severity).10 In addition, all of the ADRs were expected (Table 3).
TABLE 3 Patterns of Observed ADRs (n = 36)
High medication adherence was observed in the majority of participants, with the cost of medicines being the most frequently observed potential barrier. ADRs were reported by less than a third of participants, and all were nonpreventable. However, the majority of reported ADRs were mild, with more than half within the “possible” category.
The finding of high medication adherence among most patients was consistent with the findings of Rescigno and others in Italy,21 Iacorossi and others in Italy,22 and Diel and others23 in Germany and Central and Eastern Europe. However, Pilon and others4 reported a high rate of medication nonadherence, which affected almost half of patients with advanced prostate cancer within the first 6 months of initiation of therapy. These differing results may be related to differences in study design, given that Pilon and others4 reported medication adherence as proportion of days over a 6-month follow-up period. In a recent review of several studies, high percentages of nonadherence were also reported for both oral and parenteral medications, with higher rates observed among older persons.24 The high medication adherence observed in the current study may be associated with the availability of relevant drugs at discounted rates from the Nigerian government during the study period, indicating that access to drugs may enhance adherence.
However, cost of drugs remained a potential barrier, reported by almost half of the study participants. A related factor, low income, was a reported risk for nonadherence among patients with prostate cancer in a previous study.25 Rescigno and others21 reported forgetfulness as a potential barrier, and other recognized barriers to medication adherence in patients with prostate cancer include patient-specific factors such as suboptimal adherence-related education, comorbid mental disorders, living alone without a care partner, and high symptom burden.24 Chronic stress was also associated with nonadherence to medications.25 Androgen deprivation therapies increase survival among persons with prostate cancer, so it is important to establish good adherence by gaining insight into the potential obstacles. To improve outcomes, health care providers and policy-makers should also engage in targeted interventions to reduce the potential effect of cost of medicines on adherence.
Among factors that predicted medication adherence, patients’ family history of prostate cancer and HRQOL were significant. Meanwhile, the age of patients was a predictor of medication adherence in previous studies, with those of middle age having the best adherence.26,27 Disease prognosis and treatment regimen predicted medication adherence in a previous study of persons with breast cancer.27 These findings imply that medication adherence could be predicted by demographic, disease-specific, or medication-related variables. In addition, findings from the current study portray the need for targeted medication adherence education and prostate cancer awareness. Unlike illness perception, HRQOL predicted patients’ medication adherence in this study. This corroborates a study by de Carvalho Viana and others,28 who found that HRQOL had a direct impact on adherence to therapy among patients with prostate cancer. In a study of persons with diabetes, illness perception—particularly the domains of personal control and disease comprehension—was significantly correlated with medication adherence.29 These findings suggest that HRQOL and several domains of illness perception may significantly affect medication adherence among patients with prostate cancer. In the context of prostate cancer, HRQOL comprises the functional, social, emotional, physical, and additional concerns subscales; therefore, effective measures targeted toward improving these HRQOL domains should be explored and offered by health care providers. Effective counselling on the role of family history in subsequent cancer development should also be explored by health care professionals.
Findings from the Naranjo causality scale showed that the observed ADRs were mostly within the “possible” and “probable” categories. These results are consistent with findings from another study conducted in Nigeria involving patients with various types of cancer and a study in Nepal.8,30 Taken together, they show that ADRs of differing levels of severity can be expected during cancer therapy; hence, close monitoring of patients is vital to overall management.
All of the ADRs that occurred were expected and nonpreventable, and most had level 1 severity, as assessed by the Hartwig severity assessment scale.10 Level 1 ADRs are mild, requiring no change in therapy with the suspected drug. In contrast, most ADRs in previous studies in Nigeria and Nepal, which involved persons with various cancer types, were classified as moderate.8,30 This between-study difference in the severity of observed ADRs is likely related to the differences in cancer types studied: the previous studies included all cancer types and their associated chemotherapies, whereas our study focused on prostate cancer. Therefore, pharmacodynamic and pharmacokinetic differences between the prostate cancer drugs and those used in other types of cancer are likely responsible for the differences in severity of ADRs. Overall, our findings suggest that the prostate cancer drugs were well tolerated by the study participants.
It is important to note that loss of erection and low libido were the most frequently occurring ADRs among our study participants. Consistent with this observation, Soni and others11 identified feeling less masculine as the most disturbing symptom of hormonal treatment among study participants in Nigeria. Aguiar and others9 reported that the most common adverse reactions to treatments for prostate cancer in Brazil were reduced libido, erectile dysfunction, hyperglycemia, fatigue, and gynecomastia. Similarly, Downing and others12 reported that sexual dysfunction was common among patients with prostate cancer. Therefore, interventions such as sexual rehabilitation and appropriate measures to mitigate the effects of androgen deprivation therapies are required.
A limitation of this study is the non-use of a standardized adherence tool for assessment of medication adherence. The results for medication adherence were also based on self-reported assessment by the participants, and are thus subject to the risks associated with self-reporting and recall bias. The study was also conducted in a facility setting (rather than in the community), which may limit the generalizability of findings; however, recruitment from multiple facilities may reduce this limitation. The inclusion of all prostate cancer reference hospitals in the Nigerian state where the study was conducted also limited potential selection bias.
High medication adherence was observed in most patients, and family history of cancer was a significant predictor of medication adherence. Among the potential barriers to medication adherence that were assessed, cost of medications was determined to be a potential barrier. Therefore, policies to reduce the cost of prostate cancer medications are essential. HRQOL was also significantly related to medication adherence. The medications were well tolerated, and ADRs were minor in severity. Data from this study also revealed some predictors of medication adherence, which could be used in programs designed to enhance medication adherence. Although the observed ADRs were few, the study presented those most frequently reported by patients. This information will contribute to the design of ADR management strategies for patients with prostate cancer.
1 Rawla P. Epidemiology of prostate cancer. World J Oncol 2019;10(2): 63–89.
Crossref PubMed PMC
2 International Agency for Research and Cancer; Global Cancer Observatory. Nigeria [fact sheet]. World Health Organization; 2021 [cited 2024 Sep 3]. Available from: https://gco.iarc.who.int/media/globocan/factsheets/populations/566-nigeria-fact-sheet.pdf
3 Chen FZ, Zhao XK. Prostate cancer: current treatment and prevention strategies. Iran Red Crescent Med J. 2013;15(4):279–84.
Crossref PubMed PMC
4 Pilon D, Lamori J, Rossi C, Ghelerter I, Ke X, Lafeuille MH, et al. Medication adherence among prostate cancer patients using advanced oral therapies [abstract]. J Clin Oncol. 2021;39(6 Suppl):44.
Crossref
5 Eze UIH, Akhumi TF, Iheanacho CO, Saka SA. Drug therapy and medication adherence in type 2 diabetes in a care facility: a cross-sectional survey. Explor Res Clin Soc Pharm. 2022;8:100200.
6 Atinga RA, Yarney L, Gavu NM. Factors influencing long-term medication non-adherence among diabetes and hypertensive patients in Ghana: a qualitative investigation. PLoS One. 2018;13(3):e0193995.
Crossref PubMed PMC
7 Zed PJ, Abu-Lan RB, Balen RM, Loewen PS, Hohl CM, Brubacher JR, et al. Incidence, severity and preventability of medication-related visits to the emergency department: a prospective study. CMAJ. 2008;178(12):1563–9.
Crossref PubMed PMC
8 Musa A, Abubakar D, Caleb AO. Pattern of adverse drug reactions of anticancer drugs in oncology unit of a tertiary care teaching hospital in south west, Nigeria. Afr J Pharm Res Dev. 2016;8(2):100–7.
9 Aguiar PM, Mori ALPM, de Lima MGF, Rossi MSPN, Nicoletti MA, Martins KOF, et al. The effects of pharmacist interventions on health outcomes in patients with advanced prostate cancer in Brazil. Braz J Pharm Sci. 2022;58:e19273.
Crossref
10 Hartwig SC, Siegel J, Schneider PJ. Preventability and severity assessment in reporting adverse drug reactions. Am J Hosp Pharm. 1992; 49(9):2229–32.
PubMed
11 Soni JS, Oparah AC, Osejie F, Osifo IM, Yusuf SO, Aghahowa MA. Quality of life of patients treated for prostate cancer in a Nigerian hospital. J Sci Pract Pharm. 2017;4(1):169–76.
12 Downing A, Wright P, Hounsome L, Selby P, Wilding S, Watson E, et al. Quality of life in men living with advanced and localised prostate cancer in the UK: a population-based study. Lancet Oncol. 2019;20(3):436–47.
Crossref PubMed
13 Walang JB, Laishram PD, Brahma DK, Sarkar C, Lahon J, Nongkynrih BS. Adverse drug reactions due to cancer chemotherapy in a tertiary care teaching hospital. Ther Adv Drug Saf. 2017;8(2):61–6.
Crossref
14 Fasipe OJ, Akhideno PE, Owhin OS. The observed effect of adverse drug reactions on the length of hospital stay among medical inpatients in a Nigerian university teaching hospital. Toxicol Res Appl. 2019;3. doi:10.1177/2397847319850451
Crossref
15 Cronbach LJ. Coefficient alpha and the internal structure of tests. Psychometrika. 1951;16:297–334.
Crossref
16 Iheanacho CO, Odili VU. Impact of a pharmacist-led intervention on prostate cancer illness perception. Am J Health Syst Pharm. 2024 Jul 15: zxae193. doi:10.1093/ajhp/zxae193. Online ahead of print.
Crossref PubMed
17 Broadbent E, Petrie KJ, Main J, Weinman J. The brief illness perception questionnaire. J Psychosom Res. 2006;60(6):631–7.
Crossref PubMed
18 FACT-P. Functional assessment of cancer therapy – prostate [website]. FACIT Group; [cited 2024 Aug 4]. Available from: https://www.facit.org/measures/fact-p
19 Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239–45.
Crossref PubMed
20 Schumock G, Thornton J. Focusing on the preventability of adverse drug reactions. Hosp Pharm. 1992;27(6):538.
PubMed
21 Rescigno P, Maruzzo M, Rebuzzi SE, Murianni V, Cinausero M, Lipari H, et al. Adherence to oral treatments in older patients with advanced prostate cancer, the ADHERE study: a prospective trial of the Meet-URO Network. Oncologist. 2022;27(12):e949–e956.
Crossref PubMed PMC
22 Iacorossi L, Gambalunga F, De Domenico R, Serra V, Marzo C, Carlini P. Qualitative study of patients with metastatic prostate cancer to adherence of hormone therapy. Eur J Oncol Nurs. 2019;38:8–12.
Crossref PubMed
23 Diel IJ, Greil R, Janssen J, Kluike CW, Behera B, Abbasi A, et al. Medication adherence with denosumab in patients with bone metastases from solid tumors treated in routine clinical settings: a retrospective study. Support Care Cancer. 2022;30(11):9267–78.
Crossref PubMed PMC
24 Higano CS, Hafron J. Adherence with oral anticancer therapies: clinical trial vs real-world experiences with a focus on prostate cancer. J Urol. 2023;209(3):485–93.
Crossref
25 Oates GR, Juarez LD, Hansen B, Kiefe CI, Shikany JM. Social risk factors for medication nonadherence: findings from the CARDIA Study. Am J Health Behav. 2020;44(2):232–43.
Crossref PubMed PMC
26 Neugut AI, Zhong X, Wright JD, Accordino M, Yang J, Hershman DL. Nonadherence to medications for chronic conditions and nonadherence to adjuvant hormonal therapy in women with breast cancer. JAMA Oncol. 2016;2(10):1326–32.
Crossref PubMed
27 Dragvoll I, Bofin AM, Søiland H, Taraldsen G, Engstrøm JM. Predictors of adherence and the role of primary non-adherence in antihormonal treatment of breast cancer. BMC Cancer. 2022;22:1247.
Crossref PubMed PMC
28 de Carvalho Viana LR, Pimenta CJL, Ferreira GRS, dos Santos Oliveira J, Ferreria da Costa T, de Freitas Macedo Costa KN. Health-related quality of life and therapeutic adherence in breast and prostate cancer. Texto Contexto Enferm (Text Context Nurs). 2021;30: e20200217.
Crossref
29 Bilondi SS, Noghabi AD, Aalami H. The relationship between illness perception and medication adherence in patients with diabetes mellitus type II: illness perception and medication adherence. J Prev Med Hyg. 2022;62(4):E966–E971.
PubMed PMC
30 Tamang R, Bharati L, Khatiwada AP, Ozaki A, Shrestha S. Pattern of adverse drug reactions associated with the use of anticancer drugs in an oncology-based hospital of Nepal. JMA J. 2022;5(4):416–26.
Crossref PubMed PMC
Address correspondence to: Dr Chinonyerem O Iheanacho, Department of Clinical Pharmacy and Public Health, Faculty of Pharmacy, University of Calabar, Calabar, 540271, Cross River State, Nigeria, email: nonye2m@yahoo.com
Competing interests: None declared.
Funding: This study was supported by funding from Nigeria’s Tertiary Education Trust Fund (TETFund).
Submitted: November 28, 2023
Accepted: March 28, 2024
Published: October 9, 2024
© 2024 Canadian Society of Hospital Pharmacists | Société canadienne des pharmaciens d’hôpitaux
Canadian Journal of Hospital Pharmacy, VOLUME 77, NUMBER 4, 2024