
Rocky Lam, Jacqueline Myers, Carolyn Gray, and Alexander WongTo cite: Lam R, Myers J, Gray C, Wong A. Overcoming challenges during breastfeeding to prevent perinatal HIV transmission in Canada: a case series. Can J Hosp Pharm. 2024;77(4):e3572. doi: 10.4212/cjhp.3572
In high-income settings, exclusive use of formula remains the recommended method of feeding for infants born to women* living with HIV (WLWH). Various groups, including the American Academy of Pediatrics, endorse this practice because of the potential risk of vertical transmission through breast milk.1–3 Guidance from the World Health Organization differs and recommends breastfeeding in resource-limited settings where safe replacement feeding is not readily accessible.4 The latest recommendations from the Canadian Pediatric & Perinatal HIV/AIDS Research Group (CPARG)1 and the US Department of Health and Human Services (DHHS)2 reflect changing practice to support WLWH who choose to breastfeed, as evidence suggests minimal risk of HIV transmission (less than 1%) when maternal adherence to antiretroviral therapy (ART) is optimal and viral load is suppressed.5 Case reports describing breastfeeding in high-income settings have provided limited information detailing the real-world challenges of implementation and the considerations required to facilitate successful change in practice. To inform a shared decision-making approach to infant feeding management, this study presents a case series involving WLWH who breastfed their infants at a single site in Regina, Saskatchewan.
The medical records of 2 multipara WLWH and their 3 infants, breastfed between September 2021 and March 2023, were retrospectively reviewed. Information gathered encompassed the motivations for breastfeeding and the particular feeding strategy in each case, as well as maternal and infant characteristics and outcomes.
The study was approved by the Saskatchewan Health Authority Ethics Committee. Written consent for publication was obtained from all patients or guardians of minors.
In each case, once the mother expressed a desire to breastfeed the infant, collaborative discussions were undertaken. These discussions involved pediatric and adult infectious diseases specialists, pharmacists, lactation consultants, social workers, and dietitians, as well as nurse educators and managers. The goal was to formulate a comprehensive infant feeding strategy to support breastfeeding in WLWH, while addressing any associated stigma or concerns regarding this practice.
In accordance with recommendations from the pediatric infectious diseases specialist or specialists involved in each case, the infants were to be exclusively breastfed and were to receive combination ART (termed infant prophylaxis) consisting of nevirapine, lamivudine, and zidovudine while breastfeeding and for 1 month after cessation. Mixed feeding (with a combination of breast milk and formula) was to be avoided due to a theoretical increased risk of postnatal transmission. Donor human milk (DHM) was available as needed in all cases described here, with coverage of costs supported through the institution’s HIV program.
Infant monitoring included once-monthly HIV viral load testing up to 1 month after cessation of breastfeeding, along with complete blood count, differential blood count, liver panel, and renal panel at 2 months of age while receiving ART. Maternal monitoring included once-monthly HIV viral load testing while breastfeeding, with plans to cease breastfeeding if detectable viremia was identified. Before delivery, the health care teams of all involved units were informed of the infant feeding strategy through documentation in the maternal health record, email correspondence, and fax transmission. Application of the infant feeding strategy is outlined in Table 1, with maternal and infant characteristics described in Tables 2 and 3, respectively.
TABLE 1 Infant Feeding Strategy and Care Received for WLWH

TABLE 2 Maternal Baseline Characteristics

TABLE 3 Infant Characteristics
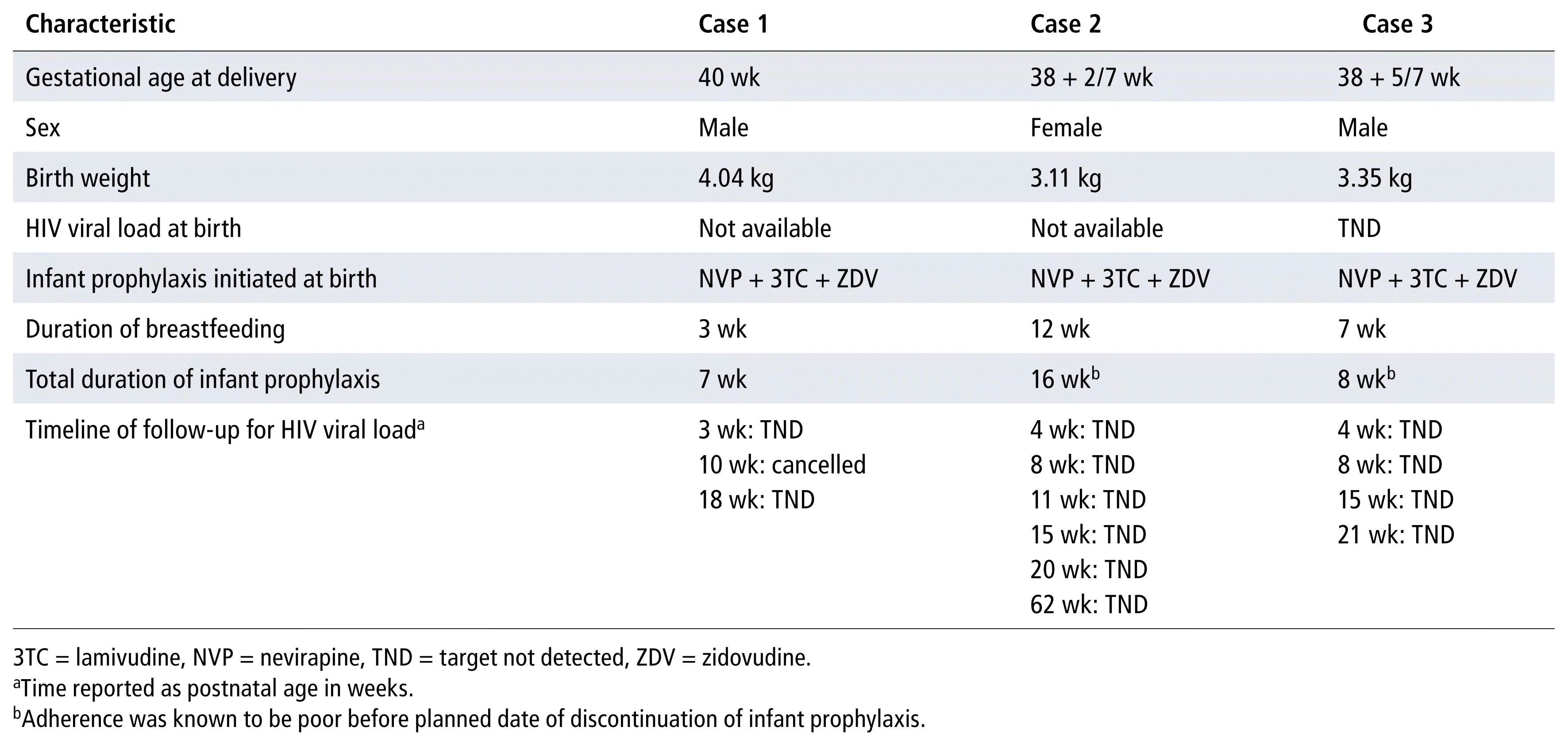
The first case involved an infant born at 40 weeks’ gestation to a 30-year-old mother of First Nations descent. Her past medical history included opioid use disorder (although she was stable on opioid agonist therapy at the time of delivery) and severe postpartum depression (PPD) with her first child, whom she did not breastfeed. She expressed interest in breastfeeding the second child to decrease the potential risk of PPD. Through a shared decision-making process, education was provided, including counselling by a lactation consultant for antenatal milk expression to begin at 36 weeks’ gestation. Following delivery, the infant was given formula during a period of unexpected separation from the mother (caused by a need for additional monitoring of the infant). The mother was informed of the error, and a shared decision was made to continue with the initial feeding strategy.
DHM was initiated due to lactation insufficiency while the mother and infant were in hospital, and the dietitian coordinated outpatient supply. Despite pharmacotherapy with domperidone for 3 weeks after discharge, lactation insufficiency continued, and a shared decision was made to discontinue breastfeeding. The infant was transitioned to formula, and infant prophylaxis continued for 1 month. Monitoring of HIV viral load at 1 month after cessation of breastfeeding and at 4 months of age confirmed that the infant remained HIV-negative. The mother did not experience PPD.
The second and third cases relate to 2 pregnancies involving the same mother of First Nations descent with a past medical history of stimulant use disorder, anxiety, depression, and PPD; the previous experience of PPD had resulted in a return to stimulant use. Although there was known difficulty with adherence to ART, the mother’s HIV viral load remained consistently suppressed before conception and throughout both pregnancies. Her expressed motivations to breastfeed included a desire to bond with her infants and a desire to ameliorate the risk of PPD and return to substance use. She also reported that she had breastfed her prior infant, although this history had not previously been disclosed to the health care team.
In case 2 (the first of this mother’s 2 pregnancies of interest), initial lactation insufficiency resulted in the use of DHM for 3 days, which was discontinued after stimulation of milk production by breast pump. Infant neutropenia, attributed to zidovudine, was identified at 9 weeks. Although a shared decision was made to continue the infant’s medications, the mother independently discontinued infant prophylaxis and began mixed feeding because she had concerns about adverse effects from infant medications. When this situation was identified 1 week later, infant prophylaxis and breastfeeding were resumed. Ongoing adherence difficulties with maternal and infant ART persisted, leading to a shared decision to transition to exclusive formula feeding. Infant prophylaxis was continued for 4 weeks after last disclosure of breastfeeding, but adherence was uncertain. The mother did not experience PPD or return to substance use, and the infant remained HIV-negative.
In case 3 (the mother’s subsequent pregnancy of interest), DHM was initiated in hospital to address the mother’s concerns regarding her milk supply. The mother was referred to a lactation consultant, who encouraged her to breastfeed, and DHM was discontinued before discharge. One month after the delivery, the mother disclosed supplemental use of DHM obtained through a private online source; she cited mental health concerns as a barrier to adherence to infant prophylaxis and exclusive breastfeeding. Nevirapine monotherapy was offered as a simplified option, but the mother elected to continue triple therapy with ongoing attempts to breastfeed. Shortly thereafter, the mother reported ongoing use of unregulated DHM, with no administration of the infant’s prophylactic medications. A shared decision was made to discontinue infant prophylaxis and all forms of breast milk, with transition to exclusive formula feeding. The infant remained HIV-negative, and the mother was referred to primary care for management of depression. The mother later disclosed to the health care team that none of the infant prophylaxis medications had been administered after discharge.
This report describes implementation and outcomes of breastfeeding for 2 WLWH and their infants at a single site in Regina, Saskatchewan. Several published cases from high-income countries (Canada, the United States, and Germany) have reported various approaches to clinical management for both mother and infant.6–9 What differentiates the current report from those earlier publications is the description of measures taken to facilitate breastfeeding, the complications experienced throughout the breastfeeding period, and the inclusion of cases involving poor adherence to ART and infant prophylaxis.
In high-income settings, there is growing interest in breastfeeding among WLWH.10,11 The reasons for wanting to breastfeed include immunologic or nutritional benefits, infant bonding, cultural expectations, and fear of HIV disclosure.7,8,12 The CPARG guidelines1 acknowledge the psychological impacts when a person’s experience of breastfeeding is limited or constrained and the subsequent feelings of grief or sadness that may arise. The decision to support these women in their choice to breastfeed respected patient desires and promoted harm reduction, given the women’s previous experiences of PPD and return to substance use, as well as a history of breastfeeding without disclosure to the health care team. To our knowledge, this report is the first to describe the risk of return to substance use and PPD as reasons for breastfeeding among WLWH.
These cases preceded the publication of the CPARG and DHHS guidelines.1,2 Successful implementation of breastfeeding for WLWH requires diligent communication and collaboration between the health care team and patient. A survey of medical professionals working with WLWH in the United States revealed that only 10% of the 100 respondents had an institutional protocol to support breastfeeding, but nearly half of respondents described having cared for or provided counselling to WLWH interested in or actively breastfeeding.13 These findings illustrate the variability in messaging and care that WLWH may receive when seeking guidance about infant feeding. To reduce the risk of deviating from the breastfeeding strategy, documentation and accessibility of this information upon transitions of care are important.
The cases reported here demonstrate the importance of supporting WLWH in making informed decisions about infant feeding without judgment or bias, as well as the value of strong trusting relationships between each patient and their practitioners. Such ongoing relationships are valuable to ensure that patients feel secure in disclosing their personal situations and the challenges they have encountered during breastfeeding. The various complications described in these cases highlight the importance of educating health care team members and parents well in advance of delivery, and the need for comprehensive follow-up to support WLWH who choose to breastfeed. Although initial education about breastfeeding and risk of HIV transmission is imperative for all involved, ongoing reinforcement of knowledge is needed to ensure clarity and understanding of concepts and to avoid misinterpretation and clinical complications.
Recent guidelines in Canada and the United States have acknowledged the need to support breastfeeding in WLWH and have recognized the importance of multidisciplinary engagement and collaboration. The cases presented here highlight various strategies that can be used to optimize successful infant feeding, while also detailing clinical complications that can occur in real-world practice. When developing flexible breastfeeding management plans for WLWH, health care providers should prioritize the establishment of a trusting patient–practitioner relationship and should apply interdisciplinary expertise accordingly. Ongoing efforts are needed to standardize clinical protocols, to promote greater awareness and adoption of current guidelines, and to educate members of the health care team about the evolving approaches in breastfeeding management for WLWH.
1 Khan S, Tsang KK, Brophy J, Kakkar F, Kennedy VL, Boucoiran I, et al. Canadian Pediatric & Perinatal HIV/AIDS Research Group consensus recommendations for infant feeding in the HIV context. J Assoc Med Microbiol Infect Dis Can. 2023;8(1):7–17.
PubMed PMC
2 Panel on Treatment of HIV During Pregnancy and Prevention of Perinatal Transmission. Recommendations for the use of antiretroviral drugs during pregnancy and interventions to reduce perinatal HIV transmission in the United States. Department of Health and Human Services (US); updated 2022 Mar 17 [cited 2023 Aug 21]. Available from: https://clinicalinfo.hiv.gov/en/guidelines/perinatal
3 Meek JY, Noble L; American Academy of Pediatrics Section on Breastfeeding. Policy statement: Breastfeeding and the use of human milk. Pediatrics. 2022;150(1):e2022057988.
Crossref PubMed
4 Guideline: updates on HIV and infant feeding: the duration of breastfeeding, and support from health services to improve feeding practices among mothers living with HIV. World Health Organization; 2016 [cited 2023 Oct 20]. 68 pp. Available from: https://iris.who.int/handle/10665/246260
5 Bispo S, Chikhungu L, Rollins N, Siegfried N, Newell ML. Postnatal HIV transmission in breastfed infants of HIV-infected women on ART: a systematic review and meta-analysis. J Int AIDS Soc. 2017;20(1):21251.
Crossref PubMed PMC
6 Weiss F, von Both U, Rack-Hoch A, Sollinger F, Eberle J, Mahner S, et al. Brief report: HIV-positive and breastfeeding in high-income settings: 5-year experience from a perinatal center in Germany. J Acquir Immune Defic Syndr. 2022;91(4):364–7.
Crossref PubMed
7 Yusuf HE, Knott-Grasso MA, Anderson J, Livingston A, Rosenblum N, Sturdivant H, et al. Experience and outcomes of breastfed infants of women living with HIV in the United States: findings from a single-center breastfeeding support initiative. J Pediatr Infect Dis Soc. 2022; 11(1):24–7.
Crossref
8 Nashid N, Khan S, Loutfy M, MacGillivray J, Yudin MH, Campbell DM, et al. Breastfeeding by women living with human immunodeficiency virus in a resource-rich setting: a case series of maternal and infant management and outcomes. J Pediatr Infect Dis Soc. 2020;9(2):228–31.
Crossref
9 Levison J, McKinney J, Duque A, Hawkins J, Bowden EVH, Dorland J, et al. Breastfeeding among people with human immunodeficiency virus in North America: a multisite study. Clin Infect Dis. 2023;77(10): 1416–22.
Crossref PubMed PMC
10 Bukkems VE, Finkenflügel RNN, Grintjes K, Marneef M, De Haan M, Mielitz I, et al. Exploring the breastfeeding desires and decision-making of women living with HIV in the Netherlands: implications for perinatal HIV management in developed countries. Breastfeed Med. 2023;18(5):356–61.
Crossref PubMed PMC
11 Haberl L, Audebert F, Feiterna-Sperling C, Gillor D, Jakubowski P, Jonsson-Oldenbüttel C, et al. Not recommended, but done: breastfeeding with HIV in Germany. AIDS Patient Care STDs. 2021;35(2): 33–8.
Crossref PubMed
12 Boucoiran I, Kaida A, Blakeley C, Skerritt L, Khan S, Bakombo MD, et al. Practices, support and stigma related to infant feeding and postpartum engagement in care among women living with HIV in Canada. AIDS Care. 2023;35(12):1971–81.
Crossref PubMed
13 Lai A, Young ES, Kohrman H, Chateau G, Cohan D, Pollock L, et al. Tilting the scale: current provider perspectives and practices on breastfeeding with HIV in the United States. AIDS Patient Care STDs. 2023;37(2):84–94.
Crossref PubMed PMC
*Within this article, the term “woman” is applied to any person who gestates a fetus. This terminology reflects current guidelines and may not be inclusive of all gender identities. Alternative terms such as “chestfeeding” or “body feeding” may be preferred to “breastfeeding”.
Address correspondence to: Dr Rocky Lam, Regina General Hospital, Department of Pharmacy Services, Saskatchewan Health Authority, Regina area, 1440 14th Avenue, Regina SK S4P 0W5, email: rocky.lam@saskhealthauthority.ca
Competing interests: For activities not related to the cases reported here, Carolyn Gray has served in a volunteer role with the Saskatchewan Branch of the CSHP; and Alexander Wong has received grants and research funds from Merck, Gilead Sciences, and ViiV Healthcare (paid to the institution); consulting fees from Gilead Sciences and ViiV Healthcare; and honoraria from Merck, Gilead Sciences, and ViiV Healthcare. No other competing interests were declared.
Funding: None received.
Submitted: December 9, 2023
Accepted: June 13, 2024
Published: November 13, 2024
© 2024 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 77, NUMBER 4, 2024