
FIGURE 1 Serum ferritin levels in relation to dose of deferoxamine (DFO).
Huisun Lau, Eddie Huang, and Caitlin OlatunbosunTo cite: Lau H, Huang E, Olatunbosun C. Continuous IV deferoxamine for chronic iron overload in patients undergoing hemodialysis by home parenteral therapy: a case report and literature review. Can J Hosp Pharm. 2024;77(4):e3577. doi: 10.4212/cjhp.3577
Chronic iron overload is a complication that affects patients with various conditions, including hematological conditions such as thalassemia, which often necessitate regular blood transfusions. Frequent transfusion may result in increased breakdown of red blood cells, leading to accumulation of iron in the body. In severe cases, excess iron can affect various organs, resulting in cirrhosis, heart failure, or endocrinological diseases. The diagnosis of chronic iron overload is based on laboratory and imaging findings, including serum ferritin, hepatic iron concentration, and cardiac iron concentration. In general, treatment is considered when there is evidence of end organ damage and when serum ferritin levels reach 800 to 1000 μg/L (normal range 12–300 μg/L).1
Iron chelation therapy, involving agents such as deferasirox, deferoxamine (DFO), and deferiprone, is the mainstay treatment for chronic iron overload. These agents work by creating complexes with free circulating elemental iron, which are then excreted renally.2 Oral agents are the preferred first-line treatment due to their ease of administration. Alternatively, IV administration of DFO offers advantages in cases of severe iron overload, as this route of administration allows the drug to bypass first-pass metabolism; furthermore, this approach has the most long-term evidence. For transfusion-dependent thalassemia, the optimal chelation regimen is individualized according to the patient’s clinical response.1 This condition may be treated with oral or IV monotherapy or a combination of the 2 modes of administration. The goals of treatment for iron chelation therapy include preventing end organ damage and targeting serum ferritin levels between 500 and 1499 μg/L.1
The usual dosage of DFO for chronic iron overload is 40 to 50 mg/kg/day (maximum 60 mg/kg/day) given intravenously for 5 to 7 days per week.1 In patients with decreased renal function (creatinine clearance 10–50 mL/min), dose reduction by 25% to 50% may be appropriate.2 There is limited evidence regarding the safe and effective dose of DFO for patients with creatinine clearance below 10 mL/min and those undergoing hemodialysis; as such, use of this agent is to be avoided in these patient populations.2 High doses of and prolonged treatment with DFO may result in adverse effects, such as increased serum transaminases, opportunistic infection (e.g., Yersinia spp.), neuropathy, myalgia, visual changes, and hearing loss. When infused over an extended duration (8–12 hours or up to 24 hours), DFO may achieve a steadier chelation effect throughout the day, with reduced risk of neurological adverse effects.1
Additional support for patients is required for such extended infusions of DFO. The Home Parenteral Therapy (HPT) Program at the University of Alberta Hospital is an outpatient program that allows patients to administer IV medications at home. Patients are given medications and supplies and receive education from staff on how to safely administer the medications in the comfort of their own homes. Sterile compounding is performed by program staff according to the National Association of Pharmacy Regulatory Authorities (NAPRA) standards for beyond-use dates (BUDs) to maintain drug sterility.3
Here, we describe a patient with iron overload and chronic kidney disease (CKD) who was undergoing hemodialysis and iron chelation therapy by continuous IV infusion through HPT. This report highlights the safe and effective administration of DFO in end-stage kidney disease requiring hemodialysis, as well as the role of the HPT pharmacist in facilitating safe and effective home treatment.
A 67-year-old woman with a complex medical history significant for chronic iron overload secondary to transfusion-dependent α-thalassemia, stage V CKD, and heart failure was referred (in August 2022) as an outpatient to HPT at the University of Alberta Hospital for initiation of IV DFO chelation therapy.* The patient had been transfusion-dependent for 4 years (since 2018), with iron overload being identified 9 months after the transfusions were started (serum ferritin 1580 μg/L; hepatic iron concentrations in the pathological range). She began receiving chelation therapy with deferiprone 1500 mg PO 3 times daily in June 2019, with minimal response. She was then referred to HPT for initiation of a second chelation agent, specifically DFO by IV administration. At the time of referral, her serum ferritin was 3094 μg/L, serum creatinine 313 μmol/L, estimated glomerular filtration rate (eGFR) 13 mL/min/1.73 m2, and creatinine clearance 14 mL/min.
DFO was initiated at a dose of 2000 mg IV daily in 250 mL normal saline (NS; 0.9% sodium chloride) as a continuous infusion over 24 hours (33 mg/kg/day), at a rate of 12.5 mL/h for ongoing chelation therapy through HPT. For this patient, the standard dose of 40 to 50 mg/kg/day was adjusted downward because of impaired renal function. The patient remained connected continuously to a home IV pump (CADD-Solis VIP ambulatory infusion pump, Smiths Medical ASD, Inc) through a peripherally inserted central catheter (PICC), and the patient’s daughter was taught to perform daily bag changes. After 1 month of dual therapy, serum ferritin levels remained above 3000 μg/L; repeat imaging showed significant improvement in hepatic iron concentrations, although they were still markedly abnormal. Over the same period, the patient’s serum creatinine increased by 35% above baseline, to 422 μmol/L (eGFR 9 mL/min/1.73 m2) with creatinine clearance of 9 mL/min. The DFO dose was reduced to 1500 mg IV daily in 250 mL NS as a continuous infusion over 24 hours (25.5 mg/kg/day) at a rate of 12.3 mL/h to accommodate the decline in renal function.
The patient experienced worsening of both heart failure and CKD and was admitted to hospital 1 month later (in October 2022) for ongoing volume overload despite maximized diuresis. After consultation with the nephrology department, intermittent hemodialysis was started, 3 times weekly over 4 hours; once the patient was discharged, 2 weeks later, the hemodialysis was performed at an outpatient dialysis clinic. Review of blister-packed medications revealed that the patient was not taking the previously prescribed deferiprone regularly; therefore, iron chelation therapy was continued with DFO as IV monotherapy. Based on a case series by Lee and others,4 DFO was given as 500 mg in 100 mL NS IV twice weekly after dialysis, as a continuous 12-hour overnight infusion at a rate of 8.3 mL/h. This extended continuous infusion, made possible through support from HPT, was preferred over post-dialysis bolus infusion of DFO to decrease the risk of neurological adverse effects. The patient had blood work and virtual follow-up from one of the HPT pharmacists (including H.L. and E.H.) every 2 weeks to monitor serum ferritin levels and adverse effects, including patient-reported visual and auditory changes, hematological changes (e.g., aplastic anemia, thrombocytopenia), increased serum transaminases, and signs of infection.
After 3 months of twice-weekly administration of DFO, the patient’s serum ferritin levels continued to rise (to 4990 μg/L), and it was decided to increase the DFO regimen to 500 mg IV 3 times weekly after dialysis, with no changes to the infusion rate or time. After 6 months of 3 times weekly administration, the serum ferritin decreased by 65%, to 1760 μg/L (Figure 1). The patient tolerated DFO throughout this course of treatment, with no neurological changes, and serum transaminases remained within normal limits. Following a readmission to address a PICC line infection, the DFO regimen was changed to 500-mg IV bolus doses during the last hour of dialysis, both for simplicity and to avoid the need for insertion of another PICC line. At this point, the patient was discharged from HPT.
|
| ||
|
FIGURE 1 Serum ferritin levels in relation to dose of deferoxamine (DFO). | ||
The patient in this case experienced severe iron overload warranting DFO therapy. The literature was searched for guidance on safe and effective dosing of this drug for treating iron overload in patients undergoing hemodialysis. Searches were conducted in the MEDLINE (Ovid) and Embase databases with the search terms “deferoxamine” and “renal insufficiency or kidney failure or hemodialysis” and “iron or iron overload” (with exploded terms). Nine relevant studies were found.4–12 Seven were case reports or small studies in which DFO was used in hemodialysis4,5,8–12; the others were small studies examining other aspects of hemodialysis in patients receiving DFO.6,7 None of the studies involved patients with renal impairment receiving DFO through HPT or an ambulatory setting.
The dose of DFO for patients undergoing hemodialysis, as described in the literature, has varied widely (Table 1), ranging from 500 mg to 2 g in total and from 1 mg/kg per week to 40 mg/kg per dialysis session. The frequency has also varied, from weekly to 3 times weekly. In most studies, DFO was given at the end of the dialysis session, over a period of 30 minutes to 3 hours. None of the dosing strategies employed continuous IV infusion.
TABLE 1 Literature Review of DFO Doses for Iron Overload in Patients Undergoing Hemodialysis
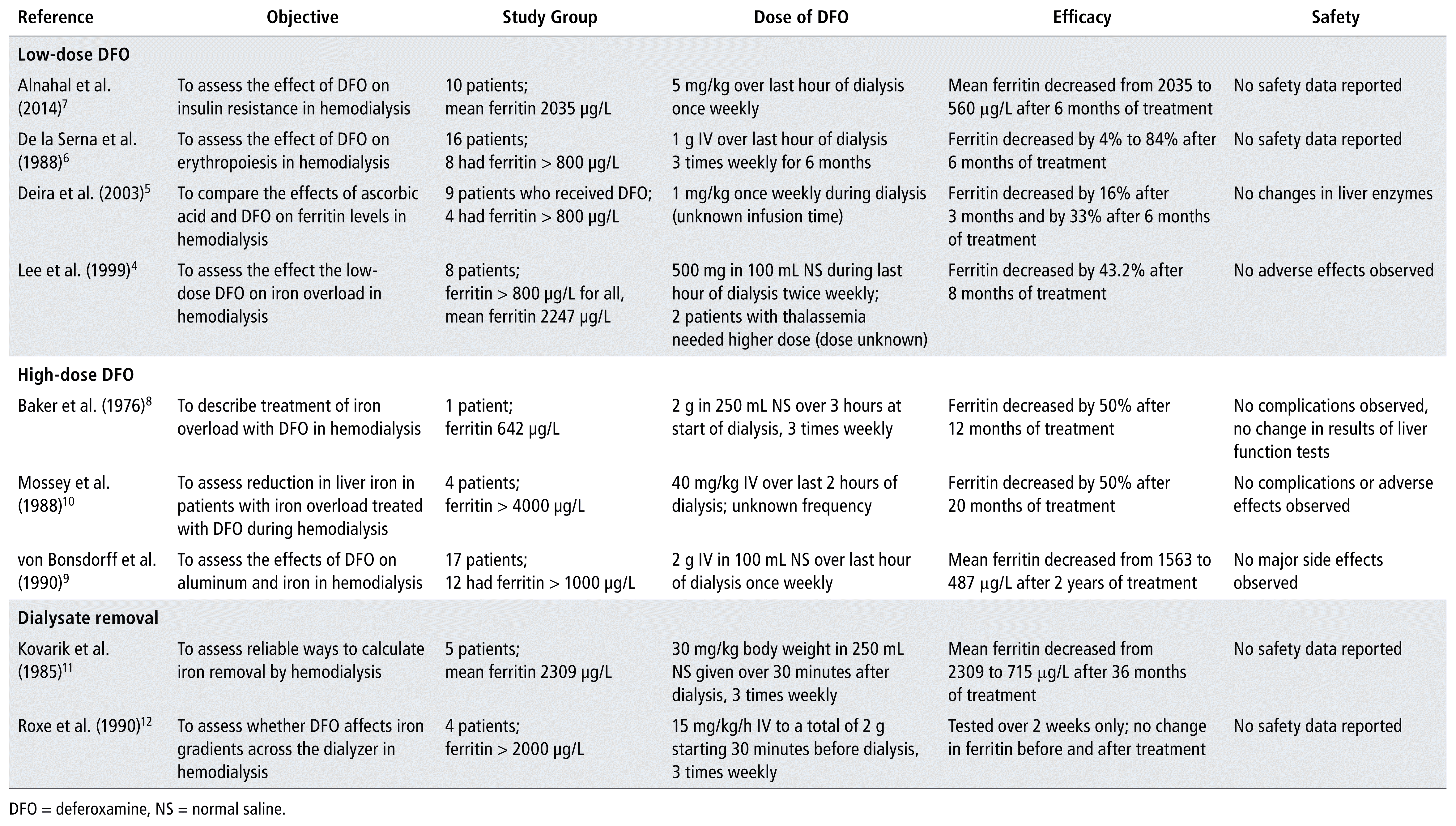
Low-dose DFO was used in 1 case series and 3 small studies.4–7 The case series4 described 8 patients who were undergoing hemodialysis accompanied by treatment with DFO for hemochromatosis, at a dose of 500 mg IV twice weekly, which resulted in a 43.2% reduction in ferritin levels after 8 months. For a 70-kg person, this total dose would translate to a weight-based dose of approximately 7 mg/kg. No adverse effects were reported for the duration of treatment. 4 Deira and others5 compared ascorbic acid with low-dose DFO to reduce iron stores through hemodialysis. Seven patients in the DFO group finished the study, but only 4 of them had ferritin levels above 800 μg/L at baseline. Although the low-dose DFO group showed improvements in serum ferritin levels, the ascorbic acid and control groups showed declines of similar proportions.5 Two other studies investigated erythropoiesis and insulin resistance, respectively, in patients undergoing hemodialysis and treatment with low-dose DFO. In one study, patients experienced a wide range of improvement in serum ferritin, with decreases ranging from 4% to 84%,6 whereas the other study summarized changes in serum ferritin with an average that indicated clinically significant improvement.7
Two case reports and 1 small study used higher doses of DFO.8–10 Both Baker and others8 and von Bonsdorff and others9 administered 2 g IV during each dialysis session (approximately 30 mg/kg for a 70-kg person), one research team attributing the dose decision to calculations on how much iron was being removed by the dialysate,8 whereas Mossey and others10 used 40 mg/kg per dose. All of these studies reported decreases in serum ferritin and no adverse effects, but the frequency of administration was highly variable, from weekly to 3 times weekly.8–10
Two small studies sought to calculate the removal of iron across the dialysate.11,12 The evidence for such removal was conflicting, but both studies used substantial doses of DFO,11,12 and one also followed the outcome in terms of serum ferritin,12 thus adding to our composite data.
The available evidence is limited by the study methods of case reports and small studies and the lack of reported details that would allow reproducibility, including administration details, success in attaining clinical targets, and adverse effects. We found no studies that compared low and high doses of DFO, or any study or case series showing dose adjustment according to clinical response. There is some evidence to suggest that heterogeneity in response to DFO may be accounted for by genetic variability rather than serum ferritin or DFO dose, as researchers have found that certain alleles associated with hemochromatosis are aligned with treatment response.13 Guidance on the doses appropriate for this population is severely lacking. For the patient described in the current report, a low-dose approach was chosen in an attempt to achieve an adequate response while avoiding adverse effects in a frail older adult. The initial dose was then adjusted based on clinical response. Our experience with this case and our review of the literature support the concept that administration of DFO 500 mg IV 3 times weekly after dialysis can be effective and safe in patients with chronic iron overload.
Several challenges specific to HPT had to be considered for the administration of DFO 500 mg IV 2 or 3 times weekly after dialysis as a continuous overnight (12-hour) infusion, including meeting NAPRA standards for BUD, infusion rate, and administration logistics.
The BUD refers to the storage time and temperature requirements that will ensure appropriate stability and sterility of the product before its administration.3 For compounded sterile products, such as DFO, the BUD is based on the risk of microbial contamination when the product is prepared. DFO is considered a medium-risk sterile preparation3; therefore, the maximum BUD that can be assigned is 9 days with refrigeration. When DFO is prepared at a concentration of 5 to 73 mg/mL in NS in elastomeric infusion pumps, it retains adequate concentration with less than 10% loss.14 Therefore, DFO 500 mg was prepared in 100-mL NS bags for a concentration of 5 mg/mL to provide a BUD of 9 days in the fridge, in adherence with NAPRA standards.
We also had to consider the infusion rate to ensure that the medication was administered continuously, at a steady rate, without increasing the risk of line complications. With continuous infusion of DFO 500 mg over 24 hours, the infusion rate is less than 5 mL/h. This low rate of infusion is often associated with a higher risk of PICC line occlusions over time, based on the experience of our HPT program and physician practice. Additionally, because DFO is largely (90%) unbound and is excreted in the urine, a portion (approximately 13%–27%) of the medication may be dialyzed out during hemodialysis.15 Because of constraints on the infusion rate, logistical coordination for the timing of doses in relation to dialysis (to prevent DFO from dialyzing out), and the outpatient support needed for HPT, DFO for this patient was administered over 12 hours at a rate of 8.3 mL/h.
We have reported a case showing the safe and effective administration of DFO through HPT for a patient undergoing hemodialysis. This case and our literature review highlight that both the doses and efficacy of DFO can be highly variable; as such, individualizing therapy according to response should be considered. Low-dose DFO can achieve therapeutic response if given 3 times weekly after dialysis. Continuous IV infusion can be employed to mitigate adverse effects, and DFO can be safely administered in an ambulatory setting with the support of an HPT team.
1 Transfusional iron overload. In: Dynamed. EBSCO Information Services; 2023 [cited 2023 Dec 7]. Accessed through institutional subscription.
2 Deferoxamine. In: Lexidrug [database]. UpToDate Inc; 2023 [cited 2023 Dec 7]. Available from: https://online.lexi.com. Subscription required to access content.
3 Model standards for pharmacy compounding of non-hazardous sterile preparations. National Association of Pharmacy Regulatory Authorities; 2015 Nov [revised 2016 Nov]. Available from: https://napra.ca/wp-content/uploads/2022/09/NAPRA-Mdl-Stnds-Pharmacy-Compounding-Non-Hazardous-Sterile-Preparations-Nov-2016-Revised-b.pdf.
4 Lee CT, Liao SC, Hsu KT, Lam KK, Chen JB. Low dose desferrioxamine can improve erythropoiesis in iron-overload hemodialysis patients without side effects. Renal Fail. 1999;21(6):665–73.
Crossref
5 Deira J, Diego J, Martínez R, Oyarbide A, González A, Díaz H. Comparative study of intravenous ascorbic acid versus low-dose desferroxamine in patients on hemodialysis with hyperferritinemia. J Nephrol. 2003;16(5):703–9.
6 De la Serna FJ, Gilsanz F, Ruilope LM, Praga M, Rodicio JL, Alcazar JM. Improvement in the erythropoiesis of chronic haemodialysis patients with desferrioxamine. Lancet. 1988;331(8593):1009–11.
Crossref
7 Alnahal AA, Tahan M, Fathy A, Fathy T. Effect of deferoxamine therapy on insulin resistance in end-stage renal disease patients with iron overload. Saudi J Kidney Dis Transpl. 2014;25(4):808–13.
Crossref PubMed
8 Baker LRJ, Barnett MD, Brozovic B, Cattell WR, Ackrill P, McAlister J, et al. Hemosiderosis in a patient on regular hemodialysis: treatment by desferioxamine. Clin Nephrol. 1976;6(1):326–8.
PubMed
9 von Bonsdorff M, Sipilä R, Pitkänen E. Correction of haemodialysis-associated anaemia by deferoxamine. Effects on serum aluminum and iron overload. Scand J Urol Nephrol Suppl. 1990;131:49–54.
PubMed
10 Mossey RT, Wielopolski L, Bellucci AG, Wilkes BM, Chandra M. Reduction in liver iron in hemodialysis patients with transfusional iron overload by deferoxamine mesylate. Am J Kidney Dis. 1988;12(1):40–4.
Crossref PubMed
11 Kovarik J, Irschik H, Graf H, Woloszczuk W, Meisinger V, Linkesch W, et al. Iron removal by desferrioxamine in patients on chronic hemodialysis—kinetic study and long-term results. Contrib Nephrol. 1985:49:44–55.
12 Roxe DM, Krumlovsky FA, Del Greco F, Fitzsimons E. Failure of deferoxamine to improve iron overload in chronic hemodialysis patients. Int J Artif Organs. 1990;13(4):211–7.
Crossref PubMed
13 Hakim RM, Stivelman JC, Schulman G, Fosburg M, Wolfe L, Imber MJ, et al. Iron overload and mobilization in long-term hemodialysis patients. Am J Kidney Dis. 1987;10(4):293–9.
Crossref PubMed
14 Deferoxamine in NS. In: Trissel’s IV compatibility tool in UpToDate Lexidrug. Wolters Kluwer; 2023 [cited 2023 Dec 7]. Available from: https://online.lexi.com/lco/action/ivcompatibility/trissels. Subscription required to access content.
15 Deferoxamine. In: Dialyze-IHD: dialyzability of medications in patients undergoing intermittent hemodialysis. Cathy Kwok [self-published]; © 2012 [cited 2024 Mar 15]. Available from: https://www.dialyzeihd.com/index.php/anemia-medications/deferoxamine
*The patient provided verbal consent for publication of this case report.
Huisun Lau, PharmD, is with the Home Parenteral Therapy Program, Alberta Health Services, Edmonton, Alberta.Address correspondence to: Dr Huisun Lau, Kaye Edmonton Clinic – Clinical Pharmacy, 11400 University Avenue, Edmonton AB T6G 1Z1, email: huisun.lau@ahs.ca
Competing interests: None declared.
Funding: None received.
Submitted: December 21, 2023
Accepted: March 28, 2024
Published: October 9, 2024
© 2024 Canadian Society of Hospital Pharmacists | Société canadienne des pharmaciens d’hôpitaux
Canadian Journal of Hospital Pharmacy, VOLUME 77, NUMBER 4, 2024