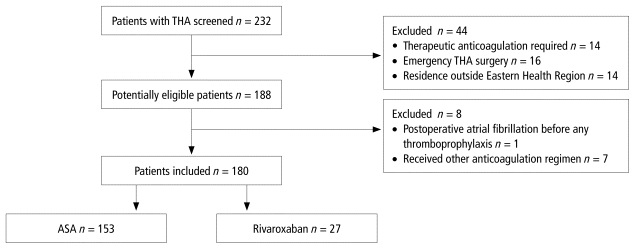
FIGURE 1 Flow diagram of study cohort. ASA = acetylsalicylic acid, THA = total hip arthroplasty.
Rebecca Norman, Jason Kielly, Kwadwo Osei Bonsu, Rufaro S Chitsike, and Stephanie YoungTo cite: Norman R, Kielly J, Bonsu KO, Chitsike RS, Young S. Comparison of acetylsalicylic acid with rivaroxaban or alternative anticoagulants for thromboprophylaxis in elective total hip arthroplasty: a retrospective cohort study. Can J Hosp Pharm. 2025;78(3):e3599. doi: 10.4212/cjhp.3599
ABSTRACT
Background
The role of acetylsalicylic acid (ASA) thromboprophylaxis following elective total hip arthroplasty (THA) remains unclear, given limited high-quality evidence and differing guideline recommendations.
Objectives
To compare thromboprophylaxis prescribing patterns for ASA and alternative anticoagulants and to determine subsequent health care utilization for patients undergoing elective THA within one Canadian health region.
Methods
This retrospective cohort study involved adult patients who underwent THA between January and June 2019, comparing those who received ASA with those who received an alternative anticoagulant. Data for drug- and patient-specific characteristics are reported as means and standard deviations for continuous variables and as percentages for categorical variables. Patient characteristics and treatments were compared using χ2 and t tests. Multivariable logistic regression was conducted to identify predictors of the choice of treatment (ASA vs rivaroxaban).
Results
Of the 180 patients who underwent THA in the study period, thromboprophylaxis consisted of ASA for 153 (85.0%) and rivaroxaban for 27 (15.0%). The most common ASA regimen was 325 mg daily (152/153, 99.3%) for a mean of 43.0 (standard deviation 6.9) days. Significant patient differences between the ASA and rivaroxaban groups included history of malignancy (6.5% vs 51.9%, p < 0.001), previous venous thromboembolism (0% vs 11.1%, p = 0.001), thrombophilia (0% vs 3.7%, p = 0.018), and chemotherapy (0% vs 11.1%, p < 0.001). Patients with a history of malignancy were more likely to receive rivaroxaban than ASA (odds ratio 31.65, 95% confidence interval 18.22 to 2.4 × 104, p < 0.001). No differences were observed for health care utilization.
Conclusions
ASA was used as thromboprophylaxis after THA for most patients within one Canadian health region. Significant differences in baseline characteristics were noted between patients who received ASA and those who received rivaroxaban.
Keywords: thromboprophylaxis, acetylsalicylic acid, rivaroxaban, hip replacement
RÉSUMÉ
Contexte
Le rôle de l’acide acétylsalicylique (AAS) dans la prophylaxie de la thrombose à la suite d’une arthroplastie totale de la hanche (ATH) demeure flou, vu la quantité restreinte de données probantes de haute qualité et les recommandations divergentes des lignes directrices.
Objectifs
Comparer les schémas de prescription de l’AAS et d’autres anticoagulants pour la prophylaxie de la thrombose; et déterminer l’utilisation subséquente des soins de santé pour les patients qui subissent une ATH au sein d’une région sanitaire canadienne.
Méthodologie
Cette étude de cohorte rétrospective portait sur des patients adultes ayant subi une ATH entre janvier et juin 2019. Elle comparait ceux ayant reçu de l’AAS à ceux ayant reçu d’autres anticoagulants. Les données concernant les caractéristiques spécifiques aux patients et aux médicaments sont présentées sous forme de moyennes et d’écarts-types pour les variables continues, et sous forme de pourcentages pour les variables catégorielles. Les caractéristiques et les traitements des patients ont été comparés à l’aide du test du χ2 et du test t. Une régression logistique multivariée a été effectuée afin de cerner les facteurs prédictifs du choix du traitement (AAS contre rivaroxaban).
Résultats
Parmi les 180 patients ayant subi une ATH au cours de la période de l’étude, la thromboprophylaxie consistait en de l’AAS pour 153 patients (85,0 %) et en du rivaroxaban pour 27 d’entre eux (15,0 %). Le schéma posologique d’AAS le plus courant était de 325 mg par jour (152/153, 99,3 %) pendant une durée moyenne de 43,0 jours (écart-type : 6,9 jours). Des différences significatives entre les groupes AAS et rivaroxaban ont été observées concernant les antécédents de malignités (6,5 % contre 51,9 %, p < 0,001), les antécédents de thromboembolie veineuse (0 % contre 11,1 %, p = 0,001), la thrombophilie (0 % contre 3,7 %, p = 0,018) et la chimiothérapie (0 % contre 11,1 %, p < 0,001). Les patients ayant des antécédents de malignités étaient plus susceptibles de recevoir du rivaroxaban que de l’AAS (rapport de cotes : 31,65; intervalle de confiance à 95 % : 18,22 à 2,4 × 104; p < 0,001). Aucune différence n’a été observée en ce qui concerne le recours aux soins de santé.
Conclusions
L’AAS a été utilisé comme thromboprophylaxie après une ATH chez la plupart des patients dans une région sanitaire canadienne. Des différences significatives dans les caractéristiques de base ont été observées entre les patients ayant reçu de l’AAS et ceux ayant reçu du rivaroxaban.
Mots-clés: thromboprophylaxie, acide acétylsalicylique, rivaroxaban, arthroplastie de la hanche
Patients who undergo total hip arthroplasty (THA) or total knee arthroplasty (TKA) are particularly susceptible to venous thromboembolism (VTE), which manifests as deep vein thrombosis or pulmonary embolism.1 This problem is due to vascular tissue injury from the procedure compounded by postsurgical immobilization.1 In these patients, major and fatal VTE is now infrequent, because the use of evidence-based thromboprophylaxis has significantly reduced the risk.1 Although a number of clinical trials have examined various thromboprophylaxis regimens in orthopedic surgeries, controversy remains in terms of appropriate agents, optimal timing, and the duration of administration.
Current guidelines and clinical guides differ in their recommendations for thromboprophylaxis after THA or TKA.1–4 The American College of Chest Physicians’ 2012 guidelines recommended several options, including acetylsalicylic acid (ASA) monotherapy, although one panel member felt strongly that ASA alone should not be included as an option.2 The American Society of Hematology has also suggested ASA or anticoagulants for thromboprophylaxis in this setting.3 Furthermore, despite the American Academy of Orthopaedic Surgeons recommending ASA as a potential pharmacologic option for VTE prophylaxis in the past, this organization’s current guidelines no longer make specific recommendations regarding pharmacotherapies for thromboprophylaxis in this setting, citing an insufficient evidence base to draw conclusions about optimal and suboptimal prophylactic regimens.4 The Extended Venous Thromboembolism Prophylaxis Comparing Rivaroxaban to Aspirin Following Total Hip and Knee Arthroplasty study (EPCAT II), a large, multicentre Canadian double-blind, randomized controlled trial (RCT), provided evidence for the use of ASA in an extended prophylaxis regimen consisting of rivaroxaban 10 mg per day for the first 5 days postoperatively followed by ASA 81 mg daily for 9 days after TKA and 30 days after THA (with the comparator group continuing to receive rivaroxaban).5 Thrombosis Canada provides a clinical guide for thromboprophylaxis after orthopedic surgery, including THA or TKA, which lists direct oral anticoagulants (DOACs), low-molecular-weight heparin (LMWH), or ASA (after 5 days of rivaroxaban) as pharmacologic options.1 US guidelines and the Thrombosis Canada clinical guides both indicate that the optimal duration for VTE prophylaxis is not known; however, they recommend prophylaxis ranging from 14 to 35 days, with longer duration to be used for patients at a greater-than-usual risk of VTE.1–4 A recent systematic review and meta-analysis concluded that ASA did not differ in its clinical effectiveness or safety profile from anticoagulants used for VTE prophylaxis after THA and TKA.6 However, the meta-analysis was based on a small number of RCTs, with the included studies having limitations of small sample sizes, variable methodologies, and inconsistent ASA and comparator regimens; as such, the authors highlighted the need for future high-quality studies in this area.6
At the time of the current study, the Eastern Regional Health Authority, also known as Eastern Health, was the largest of 4 integrated health authorities in Newfoundland and Labrador. (On April 1, 2023, the province’s 4 health authorities and the Newfoundland and Labrador Centre for Health Information merged to become a single health authority, Newfoundland and Labrador Health Services. Because the study period for the research presented here predated this merger, the previously used name “Eastern Health” is used in this article.) Eastern Health offered the full continuum of health and community services, including orthopedic surgery, and served a population of more than 300 000 (about 60% of the provincial population). Before 2009, the standard of care at Eastern Health for thromboprophylaxis after elective THA and TKA was LMWH. In May 2009, rivaroxaban became the principal thromboprophylaxis agent used after TKA and THA, based on trials showing similar efficacy and safety relative to LMWH and increased compliance and ease of administration with oral rivaroxaban. Both rivaroxaban and LMWH are included as medication options in the hospital’s standard, evidence-based VTE prophylaxis order set for patients undergoing orthopedic surgery.7–10 This order set is usually completed by the orthopedic surgeon or a delegate. ASA is not included in the standard order set as a medication option; therefore, if ASA is requested, a separate medication order must be written. In clinical practice, ASA appears to be increasingly prescribed for this patient population.11,12 Because the role of ASA for thromboprophylaxis after total joint arthroplasty remains unclear, we were interested in the frequency and potential reasons for use of ASA relative to other anticoagulants for this patient population within our organization. We surmised that this information would provide data for discussion and review of existing practices within the organization.
In this study, we sought to identify and compare prescribing patterns for ASA and alternative anticoagulants, as well as postoperative health care utilization, among patients undergoing elective THA within Eastern Health.
We conducted a retrospective cohort study of patients within Eastern Health who had undergone elective THA. The decision to focus on patients with THA reflected limited resources available and a requirement to complete the project within 1 year (the duration of a postgraduate year 1 hospital pharmacy residency program). The study was approved by the provincial Health Research Ethics Board and the Eastern Health Research Proposals Approval Committee.
All patients 19 years of age or older who had undergone elective THA at Eastern Health between January 1 and June 30, 2019, were eligible for inclusion. Patients were identified through the electronic health records of the Eastern Health Surgery Program. Patients were excluded if they received therapeutic anticoagulation for another reason, underwent emergency THA surgery or surgery for revision of THA, or resided outside the Eastern Health region (because detailed health records for patients in other regions were not available).
Each patient was assigned a unique identifier before data collection. The principal investigator (R.N.) collected the following data from the electronic hospital records using a standardized data collection form: demographic characteristics, medications and medical conditions before surgery, surgery-specific details as reported in the postoperative notes, thromboprophylaxis prescribed, and health outcomes and health care utilization up to 90 days postoperatively. The duration of thromboprophylaxis consisted of the days of therapy while an inpatient and the total number of days covered by the discharge prescription.
The primary outcomes involved comparisons of patients who received ASA and those who received alternative anticoagulants and included the percentage of patients receiving each type of medication, the specific regimens prescribed (dose and duration), and health care utilization up to 90 days postoperatively. Health care utilization consisted of postoperative hospitalization, emergency department visits, and referrals to community health services.
The secondary outcomes were the incidence of VTE and major bleeding events for patients receiving ASA and those receiving alternative anticoagulants up to 90 days postoperatively. The presence of VTE, either pulmonary embolism or proximal deep vein thrombosis, was based on review of data from electronic medical records, including radiologists’ text reports of diagnostic imaging noted as positive for VTE. Occurrence of major bleeding, defined according to the International Society on Thrombosis and Haemostasis,13 was based on review of medical notes, laboratory testing results, and radiologists’ text reports of diagnostic imaging. Two team members (the principal investigator, R.N., and a thrombosis physician/hematologist, R.S.C.) adjudicated the occurrence of VTE and major bleeding episodes.
Descriptive statistics are used to report characteristics within the groups. Continuous variables are reported as means with standard deviation (SD), and categorical data as frequencies and percentages. Proportions and means for categorical and continuous variables were compared between treatment groups using χ2 and t tests, respectively. Logistic regression was used to determine predictors of treatment choice (ASA vs rivaroxaban). The data were analyzed using SPSS software, version 26 (IBM Corporation).
A total of 232 patients who underwent THA were screened for inclusion (Figure 1). Of these, 44 were excluded (14 needed therapeutic anticoagulation, 16 had undergone emergency THA surgery, and 14 resided outside the Eastern Health region) (Figure 1). Of the 188 eligible patients, an additional 8 were subsequently excluded: 1 patient experienced postoperative atrial fibrillation before receiving thromboprophylaxis, and 7 patients received an anticoagulation regimen other than ASA or rivaroxaban, specifically a mixed regimen of LMWH and rivaroxaban or a combination of ASA with either rivaroxaban or LMWH. Although we were interested in various thromboprophylactic regimens, there was no consistency or standard within the mixed-regimen group regarding the prophylactic regimen prescribed. Furthermore, given the small sample size, statistical comparisons were impracticable. For these reasons, individuals receiving regimens other than ASA or rivaroxaban were excluded from the primary analysis.
|
| ||
|
FIGURE 1 Flow diagram of study cohort. ASA = acetylsalicylic acid, THA = total hip arthroplasty. | ||
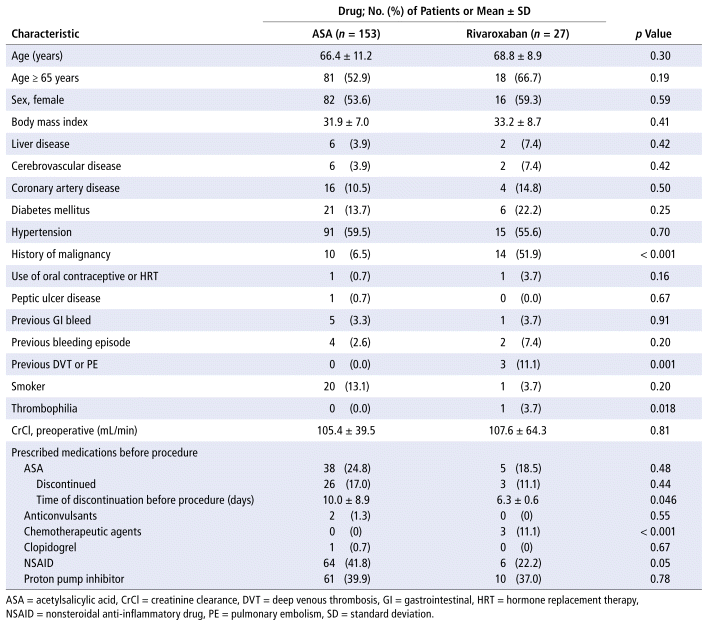
As a result, a total of 180 patients were included in the study, 153 (85%) who received ASA and 27 (15%) who received rivaroxaban (Table 1). Baseline characteristics of eligible patients were similar between the ASA and rivaroxaban groups for most variables, except history of malignancy (6.5% vs 51.9%, p < 0.001), previous VTE (0% vs 11.1%, p = 0.001), thrombophilia (0% vs 3.7%, p = 0.018), and use of chemotherapeutic agents (0% vs 11.1%, p < 0.001).
TABLE 1 Baseline Characteristics
The surgical characteristics were largely similar between the 2 treatment groups. However, some statistically significant differences were observed between patients treated with ASA and those treated with rivaroxaban. Specifically, those treated with ASA had a significantly longer duration of surgery (mean 1.2 [SD 0.3] h vs 1.0 [SD 0.3] h, p = 0.032), lower mean estimated blood loss (mean 392.2 [SD 141.0] mL vs 462.5 [SD 200.7] mL, p < 0.001), and shorter length of hospital stay (mean 3.3 [SD 2.9] days vs 4.7 [SD 3.7] days, p = 0.040) (Table 2).
Among patients in the ASA group, the most common regimen prescribed was 325 mg daily (152/153 patients, 99.3%) for a mean of 43.0 (SD 6.9) days (Table 3). The remaining patient received ASA 162 mg once daily for 4 days postoperatively, with a discharge prescription for ASA 325 mg daily.
TABLE 3 Characteristics of Thromboprophylaxis Prescribed
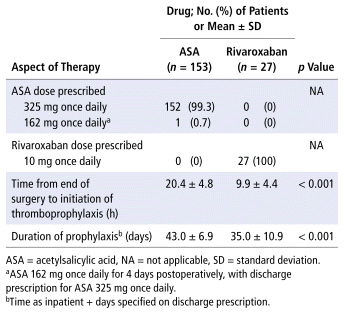
Among the patients who received rivaroxaban, the prescribed regimen was 10 mg daily for all 27 patients (100%) for a mean of 35.0 (SD 10.9) days.
Patients in the ASA group differed significantly from those in the rivaroxaban group in terms of a longer time from completion of surgery to initiation of thromboprophylaxis (mean 20.4 [SD 4.8] h vs 9.9 [SD 4.4] h, p < 0.001) and longer duration of prophylaxis (mean 43 [SD 6.9] days vs 35.0 [SD 10.9] days, p < 0.001).
There were no statistically significant differences between groups in terms of visits to the emergency department, hospitalizations, or referrals to community health services (Table 4).
TABLE 4 Health Care Utilization and Postsurgical Outcomes
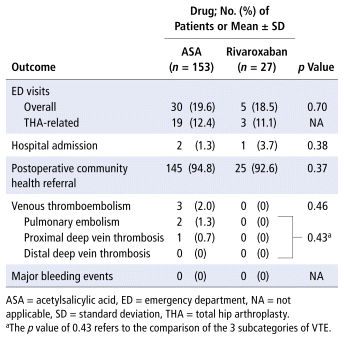
Similarly, the secondary outcomes of postsurgical VTE and major bleeding events were not significantly different between the 2 treatment groups. The overall incidence of postoperative VTE in the primary analysis population was 1.7% (3/180). These VTE events occurred in 3 (2.0%) of the 153 patients in the ASA group; no patients in the rivaroxaban group experienced postoperative VTE. There were no major bleeding events in either group up to 90 days after THA.
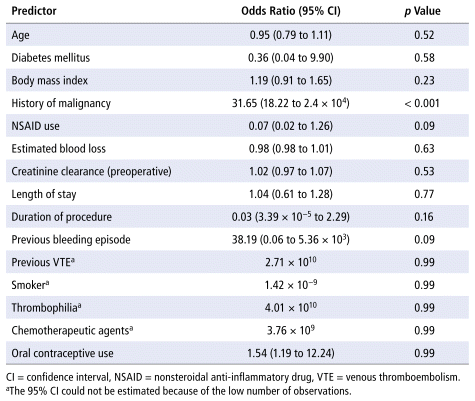
A history of malignancy was found to be a statistically significant predictor of the choice of rivaroxaban versus ASA for VTE prophylaxis (odds ratio 31.65, 95% confidence interval 18.22 to 2.4 × 104, p < 0.001) (Table 5).
TABLE 5 Multivariate Logistic Regression Analysis for Predictors of Choice of Thromboprophylaxis
In this study, we found that for VTE prophylaxis after THA, ASA monotherapy was prescribed for 85% of the eligible study population, with rivaroxaban being prescribed for the remaining 15%. The overall incidence of postoperative VTE in the primary analysis population was 1.7%.
There were significant differences in patient characteristics between the ASA and rivaroxaban groups, including history of malignancy, previous VTE, thrombophilia, and receipt of chemotherapy. These characteristics may be associated with a higher risk of VTE, thus providing a possible rationale for treatment choice by the prescribers.14 However, the clinical relevance of these differences requires further exploration. Additionally, multivariate logistic regression showed that patients with a history of malignancy were more likely to receive rivaroxaban than ASA for VTE prophylaxis after elective THA. Although other potential predictors, such as previous VTE, thrombophilia, and receipt of chemotherapy, did not yield significant results in the regression analysis, investigation with a larger sample size might illustrate a difference.
Despite the literature concerning use of ASA monotherapy for thromboprophylaxis after THA, much of the available evidence remains of limited quality, with small sample sizes, variable methodology, and inconsistencies in terms of the ASA and comparator regimens (i.e., dose and duration) assessed.6,15,16 In the available studies, patients with characteristics associated with higher risk of VTE were often underrepresented or excluded, and thus there is limited evidence to support use of ASA in these patients. The results of our study identify prescribing patterns that are consistent with this concept.
Our findings are consistent with some observational cohort studies, which have reported that ASA was effective for VTE prophylaxis and that the effectiveness and safety profile of ASA was similar to that of other commonly used anticoagulants.17,18 The findings are also supported by previous RCTs5,19 and a recent meta-analysis of more than 6000 patients,6 which concluded that ASA is as effective and safe as DOACS for VTE prophylaxis in patients who have undergone THA and TKA. In particular, the 2 RCTs found no difference between these therapies.5,19 Although our study was not sufficiently powered to determine differences in clinical outcomes between ASA and rivaroxaban for VTE prophylaxis in THA, the findings are still important in the context of the existing literature. The current evidence may plausibly support the continued use of ASA for VTE prophylaxis after THA, given that our study enrolled consecutive patients who underwent the procedure over a 6-month period; as such, our study population was unselected, and our findings represent a true reflection of outcomes in real-world patients.
Conversely, our findings do not agree with those of certain other studies. One RCT demonstrated a higher risk of VTE with ASA than with rivaroxaban,20 and a recent large observational study by Matharu and others21 suggested that DOACs were associated with a reduced risk of VTE and shorter length of stay compared with ASA. Given that our research was a small retrospective cohort study that was underpowered to detect differences in rates of thromboembolic events (which are inherently low in this patient population), further RCTs are needed to obtain a definitive answer. Nonetheless, our observational study of consecutive patients provides useful data to power subsequent trials comparing ASA with DOACs.
The overall rate of postoperative VTE in our study was lower (1.7%, 3/180) than that reported in a recent meta-analysis of 13 RCTs comparing ASA with alternative anticoagulants (7.9%, 459/5787).6 This may be the result of most trials in the meta-analysis including patients with both symptomatic and asymptomatic VTE. In contrast, our study reports data for symptomatic VTE only, which could account for the lower rate that we observed. The large EPCAT II RCT compared rivaroxaban 10 mg daily with ASA 81 mg daily, preceded by 5 days of postoperative rivaroxaban 10 mg orally daily among patients who underwent elective THA and TKA; in that study, the rate of symptomatic VTE was 0.67% (23/3424).5 It is unclear why the rate of symptomatic VTE was higher in our study. Notably, a large proportion (85%) of our patients were receiving ASA alone, whereas the EPCAT II trial had similar-sized groups in which all patients received 5 days of anticoagulation at the outset. In our study, all VTE events occurred in the ASA group. However, the difference between groups did not reach statistical significance, likely due to the small sample size and the study being underpowered to detect a significant difference in VTE rates.
The dose of ASA prescribed in this study was 325 mg daily, despite evidence that low-dose ASA (81 mg) is sufficient for platelet inhibition.22 There are other proposed mechanisms by which ASA is thought to reduce VTE risk,23 but it is unclear why this specific dose of ASA was used. The standard VTE prophylaxis order set for patients undergoing orthopedic surgery at this organization does not include ASA as an option and therefore makes no recommendation regarding a specific ASA dose. Currently, 2 ongoing trials are investigating the use of ASA monotherapy as thromboprophylaxis after elective THA and TKA. The results of these trials will help our organization to interpret the current findings and will provide future direction on clinical practice by providing clarity on the effectiveness of ASA monotherapy for VTE prevention, as well as dosing information in this setting. The Extended Venous Thromboembolism Prophylaxis Comparing Rivaroxaban and Aspirin to Aspirin Alone Following Total Hip and Knee Arthroplasty (EPCAT III) trial is a randomized, double-blind, non-inferiority study comparing ASA monotherapy 81 mg daily for 35 days after THA and 14 days after TKA with rivaroxaban 10 mg daily for 5 days followed by ASA 81 mg daily for 30 days after THA and 9 days after TKA ( ClinicalTrials.gov ID NCT04075240). The Comparative Effectiveness of Pulmonary Embolism Prevention After Hip and Knee Replacement (PEPPER) trial is an RCT comparing ASA monotherapy, rivaroxaban, and low-intensity warfarin for VTE prophylaxis in elective primary or revision THA and TKA ( ClinicalTrials.gov ID NCT02810704). In the PEPPER trial, ASA 81 mg twice a day will be compared with rivaroxaban 10 mg daily and warfarin (dosed empirically based on weight and targeting an international normalized ratio of 2.0 [range 1.7–2.2]).
A strength of our study was the enrolment of consecutive eligible patients within the study period, reflecting real-world choice of thromboprophylaxis and treatment outcomes.
This study had several limitations. Given its retrospective nature, causal inferences could not be drawn. Additionally, our study was underpowered to detect a difference in important outcomes such as VTE or major bleeding events, which have inherently low incidence rates in this patient population. The study was restricted to a single Canadian health region, with data collected over a short period, which may affect generalizability. Finally, adherence to thromboprophylaxis treatment was not evaluated. Future studies would require collection of data on treatment adherence to evaluate the true impact of treatment on patient-important outcomes.
Within one Canadian health region, ASA was used as thromboprophylaxis following elective THA in a substantial percentage of patients. Significant differences in baseline characteristics were seen between patients who received ASA and those who received rivaroxaban. Patients with a history of malignancy were more likely to receive rivaroxaban than ASA for VTE prophylaxis following elective THA. No differences were seen between groups in terms of health care utilization up to 90 days after the surgery. The clinical relevance of the observed differences should be further explored.
1 Thromboprophylaxis: orthopedic surgery [clinical guide]. Thrombosis Canada; 2020 [cited 2021 May 22]. Available from: https://thrombosiscanada.ca/clinicalguides/?search=thromboprophylaxis#
2 Falck-Ytter Y, Francis CW, Johanson NA, Curley C, Dahl OE, Schulman S, et al. Prevention of VTE in orthopedic surgery patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e278S–325S.
Crossref PubMed PMC
3 Anderson DR, Morgano GP, Bennett C, Dentali F, Francis CW, Garcia DA, et al. American Society of Hematology 2019 guidelines for management of venous thromboembolism: prevention of venous thromboembolism in surgical hospitalized patients. Blood Adv. 2019; 3(23):3898–944.
Crossref
4 Mont MA, Jacobs JJ, Boggio LN, Bozic KJ, Della Valle CJ, Goodman SB, et al. Preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. J Am Acad Orthop Surg. 2011;19(12):768–76.
Crossref PubMed
5 Anderson DR, Dunbar M, Murnaghan J, Kahn SR, Gross P, Forsythe M, et al. Aspirin or rivaroxaban for VTE prophylaxis after hip or knee arthroplasty. N Engl J Med. 2018;378(8):699–707.
Crossref PubMed
6 Matharu GS, Kunutsor SK, Judge A, Blom AW, Whitehouse MR. Clinical effectiveness and safety of aspirin for venous thromboembolism prophylaxis after total hip and knee replacement: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2020;180(3):376–84.
Crossref PubMed PMC
7 Eriksson BI, Borris LC, Friedman RJ, Haas S, Huisman MV, Kakkar AK, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N Engl J Med. 2008;358(26):2765–75.
Crossref PubMed
8 Kakkar AK, Brenner B, Dahl OE, Eriksson BL, Mouret P, Muntz J, et al. Extended duration rivaroxaban versus short-term enoxaparin for the prevention of venous thromboembolism after total hip arthroplasty: a double-blind, randomised controlled trial. Lancet. 2008;372(9632):31–9.
Crossref PubMed
9 Lassen MR, Ageno W, Borris LC, Lieberman JR, Rosencher N, Bandel TJ, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty. N Engl J Med. 2008;358(26):2776–85.
Crossref PubMed
10 Turpie AG, Lassen MR, Davidson BL, Bauer KA, Gent M, Kwong LM, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty (RECORD4): a randomised trial. Lancet. 2009;373(9676):1673–80.
Crossref PubMed
11 Bala A, Murasko MJ, Burk DR, Huddleston JI 3rd, Goodman SB, Maloney WJ, et al. Venous thromboprophylaxis after total hip arthroplasty: aspirin, warfarin, enoxaparin, or factor Xa inhibitors? Hip Int. 2020;30(5):564–71.
Crossref
12 Shah SS, Satin AM, Mullen JR, Merwin S, Goldin M, Sgaglione NA. Impact of recent guideline changes on aspirin prescribing after knee arthroplasty. J Orthop Surg Res. 2016;11(1):123.
Crossref PubMed PMC
13 Schulman S, Angerås U, Bergqvist D, Eriksson B, Lassen MR, Fisher W; Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in surgical patients. J Thromb Haemost. 2010;8(1):202–4.
Crossref
14 Heit JA, Spencer FA, White RH. The epidemiology of venous thromboembolism. J Thromb Thrombolysis. 2016;41(1):3–14.
Crossref PubMed PMC
15 Haykal T, Kheiri B, Zayed Y, Barbarawi M, Miran MS, Chahine A, et al. Aspirin for venous thromboembolism prophylaxis after hip or knee arthroplasty: an updated meta-analysis of randomized controlled trials. J Orthop. 2019;16(4):294–302.
Crossref PubMed PMC
16 Azboy I, Groff H, Goswami K, Vahedian M, Parvizi J. Low-dose aspirin is adequate for venous thromboembolism prevention following total joint arthroplasty: a systematic review. J Arthroplasty. 2020; 35(3):886–92.
Crossref
17 Bala A, Huddleston JI 3rd, Goodman SB, Maloney WJ, Amanatullah DF. Venous thromboembolism prophylaxis after TKA: aspirin, warfarin, enoxaparin, or factor Xa inhibitors? Clin Orthop Relat Res. 2017;475(9):2205–13.
Crossref PubMed PMC
18 Parvizi J, Ceylan HH, Kucukdurmaz F, Merli G, Tuncay I, Beverland D. Venous thromboembolism following hip and knee arthroplasty: the role of aspirin. J Bone Joint Surg Am. 2017;99(11):961–72.
Crossref PubMed
19 Jiang Y, Du H, Liu J, Zhou Y. Aspirin combined with mechanical measures to prevent venous thromboembolism after total knee arthroplasty: a randomized controlled trial. Chin Med J (Engl). 2014;127(12):2201–5.
Crossref PubMed
20 Zou Y, Tian S, Wang Y, Sun K. Administering aspirin, rivaroxaban and low molecular-weight heparin to prevent deep venous thrombosis after total knee arthroplasty. Blood Coagul Fibrinolysis. 2014;25(7):660–4
Crossref PubMed
21 Matharu GS, Garriga C, Whitehouse MR, Rangan A, Judge A. Is aspirin as effective as the newer direct oral anticoagulants for venous thromboembolism prophylaxis after total hip and knee arthroplasty? An analysis from the National Joint Registry for England, Wales, Northern Ireland, and the Isle of Man. J Arthroplasty. 2020;35(9):2631–2639.e6.
Crossref PubMed
22 Undas A, Brummel-Ziedins KE, Mann KG. Antithrombotic properties of aspirin and resistance to aspirin: beyond strictly antiplatelet actions. Blood. 2007;109(6):2285–92.
Crossref
23 Diep R, Garcia D. Does aspirin prevent venous thromboembolism? Hematology Am Soc Hematol Educ Program. 2020;2020(1):634–41.
Crossref PubMed PMC
Address correspondence to: Dr Stephanie Young, School of Pharmacy, Memorial University, 300 Prince Philip Drive, St John’s NL A1B 3V6, email: swyoung@mun.ca
Competing interests: For activities not directly related to the study reported here, Jason Kielly received a speaker’s honorarium from the Pharmacists Association of Newfoundland and Labrador and travel support from the National Association of Pharmacy Regulatory Authorities and the Newfoundland and Labrador Department of Health related to roles on various advisory committees; Rufaro Chitsike has received grants from Sanofi and Bayer and has participated on an advisory board for Astra Zeneca; and Stephanie Young has received grant support from Sanofi Canada and NL SUPPORT (provincial funding agency supporting patient-oriented research) and speaker’s honoraria from Thrombosis Canada. No other competing interests were declared.
Funding: None received.
Submitted: June 11, 2024
Accepted: February 3, 2025
Published: July 9, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 3, 2025