Meghan Ludlam, Isla Drummond, and Angela LoTo cite: Ludlam M, Drummond I, Lo A. Impact of short-acting spinal anesthetic on discharge from postanesthetic care unit: a retrospective analysis. Can J Hosp Pharm. 2025;78(1):e3618. doi: 10.4212/cjhp.3618
ABSTRACT
Background
Chloroprocaine has recently regained accessibility as a short-acting local anesthetic in Canada. It offers an alternative to bupivacaine, the predominant spinal anesthetic used at Vancouver Coastal Health sites in Vancouver, British Columbia.
Objective
To compare the recovery profile of surgical patients who received either chloroprocaine or bupivacaine for spinal anesthesia.
Methods
This multicentre retrospective cohort study involved adult patients who received chloroprocaine or bupivacaine as spinal anesthetic for short-duration surgery. The primary outcome was the time from admission to the postanesthesia care unit (PACU) to discharge from the PACU. Time from PACU admission to discharge readiness was also assessed, along with the perioperative variables of type of operation performed, duration of surgery, dose of anesthetic, dose of intraoperative analgesic, and number of days from operation to hospital discharge.
Results
The study involved 145 patients aged 30 to 94 years: 72 who received chloroprocaine and 73 who received bupivacaine. For the primary outcome, there was a nonsignificant shorter time from PACU admission to discharge for the chloroprocaine cohort (mean difference 36 minutes, p = 0.07). There was a significant difference favouring chloroprocaine over bupivacaine in the time from PACU admission to meeting PACU discharge criteria (mean difference 48 minutes, p < 0.001). Chloroprocaine was associated with significantly shorter surgery duration (mean difference 5 minutes, p = 0.026). Regression analysis showed a significant correlation between the anesthetic given and the time to meeting PACU discharge eligibility (p < 0.001).
Conclusion
Use of chloroprocaine was associated with earlier time to patients meeting PACU discharge criteria. These findings support the potential benefit of chloroprocaine over bupivacaine in terms of achieving earlier PACU discharge readiness, although this difference did not consistently translate into earlier actual PACU discharge.
Keywords: chloroprocaine, bupivacaine, spinal anesthesia, postanesthetic recovery
RÉSUMÉ
Contexte
La chloroprocaïne est récemment redevenue disponible en tant qu’anesthésique local à action courte au Canada. Elle peut être utilisée à la place de la bupivacaïne, l’anesthésique spinal prédominant utilisé aux sites de Vancouver Coastal Health, à Vancouver (Colombie-Britannique).
Objectif
Comparer le profil de rétablissement des patients chirurgicaux ayant reçu de la chloroprocaïne ou de la bupivacaïne pour une anesthésie spinale.
Méthodologie
Cet examen rétrospectif multicentrique des dossiers impliquait des patients adultes ayant reçu de la chloroprocaïne ou de la bupivacaïne comme anesthésique spinal pour une chirurgie de courte durée. Le principal critère de l’évaluation était le temps écoulé entre l’admission à l’unité de soins post-anesthésie (USPA) et le congé de l’USPA. Le temps écoulé entre l’admission à l’USPA et la préparation au congé a également été évalué, ainsi que des variables périopératives, soit, le type d’opération réalisée, la durée de la chirurgie, la dose d’anesthésique, la dose d’analgésique peropératoire et le nombre de jours entre l’opération et le congé de l’hôpital.
Résultats
Cet examen portait sur 145 patients âgés de 30 à 94 ans : 72 ayant reçu de la chloroprocaïne et 73, de la bupivacaïne. Concernant le principal critère de l’évaluation, un temps plus court mais non statistiquement significatif entre l’admission à l’USPA et le congé pour la cohorte de patients ayant reçu de la chloroprocaïne (différence moyenne de 36 minutes, p = 0,07) a été constaté. On a par contre constaté une différence significative en faveur de la chloroprocaïne par rapport à la bupivacaïne quant au temps entre l’admission à l’USPA et le moment où les critères de congé de l’USPA étaient remplis (différence moyenne de 48 minutes, p < 0,001). La chloroprocaïne a été associée à une durée de chirurgie plus courte statistiquement significative (différence moyenne 5 minutes, p = 0,026). L’analyse de régression a montré une corrélation significative entre l’anesthésique administré et le temps nécessaire pour répondre aux critères d’admissibilité au congé de l’USPA (p < 0.001).
Conclusion
L’utilisation de la chloroprocaïne a été associée à un délai plus court avant que les patients répondent aux critères de congé de l’USPA. Ces résultats appuient l’avantage potentiel de la chloroprocaïne par rapport à la bupivacaïne sur le plan de la préparation plus précoce au congé de l’USPA, bien que cette différence ne se traduise pas systématiquement en un congé réel plus rapide de l’USPA.
Mots-clés: chloroprocaïne, bupivacaïne, anesthésie spinale, rétablissement post-anesthésique
Chloroprocaine is an amino-ester local anesthetic with a short duration of action, favourable pharmacokinetic profile, and potentially lower incidence of adverse effects,1–3 offering an attractive alternative to its longer-acting counterparts. However, chloroprocaine was removed from the Canadian market in the 1980s due to a series of neurotoxic reactions attributed to the sodium bisulfite preservative used in the formulation.4,5 Preservative-free chloroprocaine re-emerged in 2004 and is widely used in Europe and the United States; however, it is not marketed in Canada.6 In January 2021, Health Canada temporarily permitted importation of US-labelled chloroprocaine under the “No Objection Letter” process, with the goal of reducing postoperative recovery time and expediting discharge from the postanesthetic care unit (PACU) during the COVID-19 pandemic.7
Previous comparative studies between chloroprocaine and bupivacaine have indicated that chloroprocaine is associated with a shorter duration of sensory and motor block,8–13 faster time to ambulation,8–15 and earlier discharge from the PACU,10–12,15,16 thus potentially streamlining patient care processes.
In this study, we aimed to evaluate whether the use of chloroprocaine as spinal anesthesia was associated with expedited postanesthetic recovery relative to bupivacaine at our institutions. The primary outcome was the time from PACU admission to PACU discharge. The secondary outcomes were the time from PACU admission to PACU discharge readiness, correlations with demographic and perioperative variables, and incidences of intraoperative hypotension and bradycardia.
Ethics approval was obtained from the University of British Columbia Clinical Research Ethics Board and Vancouver Coastal Health Research Institute. This study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
We performed a retrospective cohort study analyzing the medical records of patients at 3 hospital sites that were using chloroprocaine for short surgical procedures. A computer-generated search of the Cerner Electronic Medical Record system identified all chloroprocaine orders between August 2021 (when chloroprocaine became accessible) and April 2023. A search for bupivacaine orders in 2019, for purposes of comparison, generated 668 orders for patients whose surgery lasted 1 hour or less. Cases in the bupivacaine cohort were then matched with cases in the chloroprocaine cohort according to procedure type, with random selection (choosing each nth patient) when multiple bupivacaine patient records were available. We selected bupivacaine orders from 2019 to reflect pre-COVID-19 surgical workflow. Patients with incomplete records for PACU admission and discharge were excluded.
Data collected from the electronic medical records were demographic variables (age, sex, American Society of Anesthesiologists [ASA] class, and weight), procedure type, duration of anesthesia and of surgery, and times from PACU admission to discharge and to discharge readiness. Time of discharge was based on the recorded timestamp of physical discharge from the PACU. Time of discharge readiness was the time when nursing staff recorded that Vancouver Coastal Health’s PACU discharge criteria had been met.17 We also recorded the presence of intraoperative hypotension or bradycardia (defined as 3 consecutive readings in the operating room lower than 90 mm Hg or 60/minute, respectively).
Analysis was completed using Microsoft Excel spreadsheet software, version 2412. Unpaired t tests with assumption of unequal variances were used to compare chloroprocaine and bupivacaine groups for the various outcomes. Linear regression was used to investigate the potential influence of demographic and perioperative variables on outcomes. Statistical significance was set a priori to p < 0.05, and 95% confidence intervals were determined for all analyses.
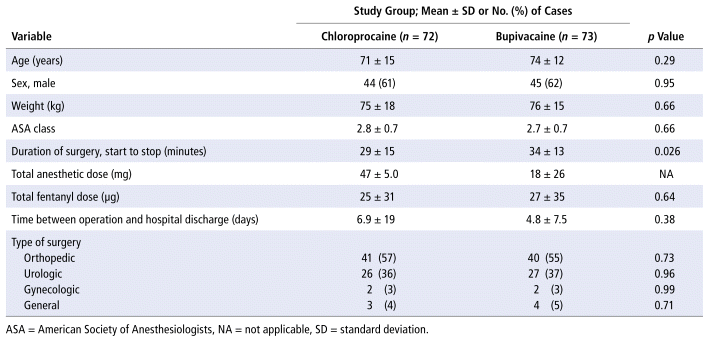
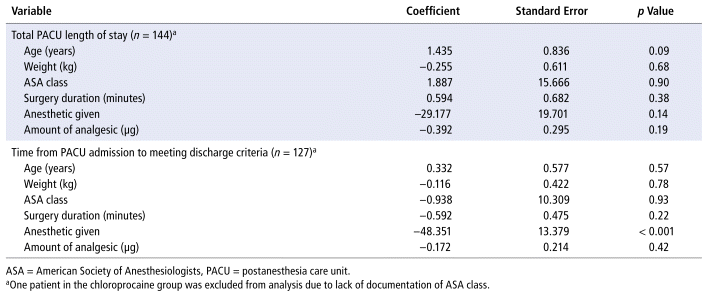
After elimination of patients without data for PACU admission and discharge times, the analysis of total PACU length of stay included 72 patients in the chloroprocaine group and 73 patients in the bupivacaine group; the patients ranged in age from 30 to 94 years. PACU discharge readiness was documented for 66 chloroprocaine patients and 62 bupivacaine patients. Patient demographic and perioperative details are presented in Table 1, along with surgery department distribution. Outcome data are presented in Table 2. The p values represent the differences between chloroprocaine and bupivacaine cohorts for each variable. Regression analysis is presented in Table 3.
TABLE 1 Baseline Demographic and Perioperative Variables

TABLE 3 Linear Regression Analysis of Demographic and Perioperative Data with Outcome Variables
Chloroprocaine patients were discharged from the PACU 36 minutes earlier, on average, than the bupivacaine group, although the difference was not statistically significant (p = 0.07) (Table 2). Chloroprocaine recipients met the PACU discharge eligibility 48 minutes earlier, on average, than the bupivacaine group, a difference that was statistically significant (p < 0.001). The average difference in time from PACU admission to PACU discharge was 57 minutes among admitted patients and 27 minutes among day surgery patients (p < 0.001).
The overall mean surgical time was 32 minutes, with the chloroprocaine group having a significantly shorter mean surgery duration (29 vs 34 minutes; p = 0.026) (Table 1). The most common surgeries performed were orthopedic and urological (Table 1), with the highest prevalence documented for open reduction and internal fixation of the hip and cytoscopy with transurethral prostatectomy, respectively. The most commonly reported anesthetic doses were 50 mg of chloroprocaine (n = 48 [67%]) and 15 mg of bupivacaine (n = 10 [14%]). In terms of intraoperative analgesia, 56 (78%) of the chloroprocaine group and 64 (88%) of the bupivacaine group received IV or intrathecal fentanyl. In total, 12% of patients experienced intraoperative hypotension and 12% experienced bradycardia, with no statistically significant difference between cohorts (Table 2). Day surgery cases accounted for 54 (37%) of our total sample. Those who stayed as inpatients had a mean hospital stay between surgery and discharge of 5.8 (standard deviation 14 days, range 0–125 days), with no significant difference between cohorts (Table 1).
We examined correlations of demographic and perioperative variables with the time of PACU discharge and discharge eligibility. No statistically significant correlations were found, except between anesthetic given and time to meeting PACU discharge eligibility (Table 3).
We hypothesized that our findings would align with earlier literature, confirming that chloroprocaine facilitates faster postanesthetic recovery and discharge relative to bupivacaine. The hypothesis was partially supported, as chloroprocaine was associated with faster PACU discharge readiness but not faster PACU discharge. These results may inform decisions on whether chloroprocaine should be added to hospital formularies if it is remarketed in Canada.
Timely discharge of patients from the PACU increases available bed space in the recovery room, thus improving patient flow and allowing for more operative procedures to be performed. Increasing the number of procedures was the main reason for reintroducing the short-acting local anesthetic chloroprocaine at our sites, at a time when there was substantial pressure to decrease surgical waitlists. The primary objective of our study was to determine whether PACU length of stay differed between patients who received chloroprocaine and those who received bupivacaine for surgeries of short (≤ 60 minutes) duration. We found that chloroprocaine accelerated PACU discharge by an average of 36 minutes (although this result was not statistically significant) and additionally led to significantly earlier PACU discharge readiness (by 48 minutes on average).
Regression analysis supported the internal validity of our study. The statistically significant influence of anesthetic choice on time to PACU discharge eligibility, but not on time to PACU discharge, aligned with our study findings. These results are consistent with a study by Lacasse and others,9 who observed similar PACU length of stay despite significantly shorter time to discharge eligibility in the chloroprocaine group.
These results support the potential for improving case turnover by increasing PACU throughput. However, the observed delay between discharge readiness and actual discharge may negate the benefit gained from using a shorter-acting anesthetic. We sought feedback from PACU staff about this delay; they suggested that the delay might be attributable to external factors such as a lack of inpatient bed space and general staff and resource shortages. The statistically significant difference of 30 minutes between admitted patients and day surgery patients in terms of time to PACU discharge supports the feedback we received. Future studies could focus on patient movement out of PACU, particularly in terms of inpatient flow, to determine ways to improve efficiency.
Patients receiving chloroprocaine had significantly shorter surgery durations (Table 1), which may reflect the concept that anesthesiologists intentionally select chloroprocaine for shorter surgeries. Although the lack of control for surgery duration as a potential confounder in our sample complicates interpretation, the difference of 5 minutes in surgical duration is unlikely to account for the 48-minute difference in time to PACU discharge readiness.
The doses of chloroprocaine and bupivacaine administered align with accepted standards; notably, bupivacaine dosing exhibits higher variability related to variation in patients’ physical condition and surgery type.18,19 Although not reflected in our results, previous literature suggests higher analgesia doses with chloroprocaine, possibly due to faster regression of spinal anesthesia with this drug.9,20,21 We observed no significant differences in intraoperative hypotension or bradycardia between the chloroprocaine and bupivacaine groups (Table 2). These end points were recorded to compare safety profiles, as they are considered primary adverse effects of these medications. However, there are challenges in attributing spinal anesthesia as the causative factor for these effects, given the presence of potential confounders, especially the concurrent intraoperative administration of fentanyl. Inconsistent findings in previous research support the concept of confounder influence.1,9,12–16 We were interested in capturing additional adverse effects, such as urinary retention and transient neurologic symptoms, but the recording of these effects in patients’ charts was inconsistent.
Anesthetic costs are higher for chloroprocaine than for bupivacaine. As such, and given our results, another future study could be a cost-effectiveness analysis. It is unclear whether using chloroprocaine rather than bupivacaine would provide meaningful benefit in terms of costs, considering staff and space constraints that hindered immediate PACU discharge even with decreased patient recovery time.
Our study had several limitations. The retrospective design relied on the accuracy of computerized provider order entry and charting. However, the operating room is a highly controlled environment with well-practised workflows, which would support the validity of our results. Our sample size was limited by the recent date of chloroprocaine adoption, and the observational nature of the study introduced biases and confounders. Nonstandardized doses of various drugs, such as sedatives, vasopressors, and analgesics, were given at the discretion of health care providers, which further contributed to variability within our study groups. Despite these limitations, this study serves as a preliminary investigation of the ability of chloroprocaine to decrease patient recovery times at our institutions. Any future studies at our sites should adopt more rigorous randomized controlled designs with larger sample sizes and standardized protocols to enhance validity and minimize confounding factors.
This multicentre observational study has illustrated the impact of using chloroprocaine instead of bupivacaine as spinal anesthetic at our institutions. By showing that chloroprocaine elicits faster PACU discharge eligibility, our findings support the potential of using chloroprocaine rather than bupivacaine to achieve faster patient discharge readiness. We recognize that addressing barriers to prompt PACU discharge soon after achievement of patient readiness is crucial to realizing the potential gains from chloroprocaine use.
1 Herndon C, Martinez R, Sarpong N, Geller J, Shah R, Cooper H. Spinal anesthesia using chloroprocaine is safe, effective, and facilitates earlier discharge in selected fast-track total hip arthroplasty. Arthroplast Today. 2020;6(3):305–8.
Crossref PubMed PMC
2 Capdevila X, Aveline C, Delaunay L, Bouaziz H, Zetlaoui P, Choquet O, et al. Impact of chloroprocaine on the eligibility for hospital discharge in patients requiring ambulatory surgery under spinal anesthesia: an observational multicenter prospective study. Adv Ther. 2020; 37(1):541–51.
Crossref PMC
3 Szerb J. Reviving older drugs to deal with anesthesia drug shortages. Can J Anaesth. 2015;62(10):1042–4.
Crossref PubMed
4 Kim DH, Kahn R, Lee A, Mac PD, Chiu YF, Yadeau J, et al. Chloroprocaine provides safe, effective, short-acting spinal anesthesia ideal for ambulatory surgeries: a retrospective review. HSS J. 2020;16(Suppl 2): 280–4.
Crossref PMC
5 Ghisi D, Bonarelli S. Ambulatory surgery with chloroprocaine spinal anesthesia: a review. Ambul Anesth. 2015;2:111–20.
6 Szerb J, Abbass S, Banfield J, Uppal V. A survey assessing the need for spinal chloroprocaine to provide subarachnoid neuraxial anesthesia for short-duration surgeries in Canada. Can J Anaesth. 2021;68(7):1096–9.
Crossref PubMed PMC
7 Section Focus – regional and acute pain. CAS Anesth News. 2022 [cited 2023 Jun 24];37(2):19–20. Available from: https://www.cas.ca/CASAssets/Documents/Anesthesia-News/CASnewsletterSummer2022.pdf
8 Koti VR, Nehha S, Rooma, Asraruddin M, Fatima SH. Comparison of chloroprocaine and bupivacaine for spinal anaesthesia in patients undergoing day care surgery: a double blind randomized controlled study. Indian J Public Health Res Dev. 2023;14(2):377–83.
Crossref
9 Lacasse MA, Roy JD, Forget J, Vandenbroucke F, Seal RF, Beaulieu D, et al. Comparison of bupivacaine and 2-chloroprocaine for spinal anesthesia for outpatient surgery: a double-blind randomized trial. Can J Anaesth. 2011;58(4):384–91.
Crossref PubMed
10 Lee A, Shatil B, Landau R, Menon P, Smiley R. Intrathecal 2-chloroprocaine 3% versus hyperbaric bupivacaine 0.75% for cervical cerclage: a double-blind randomized controlled trial. Anesth Analg. 2022; 134(3):624–32.
Crossref
11 Saporito A, Ceppi M, Perren A, La Regina D, Cafarotti S, Borgeat A, et al. Does spinal chloroprocaine pharmacokinetic profile actually translate into a clinical advantage in terms of clinical outcomes when compared to low-dose spinal bupivacaine? A systematic review and meta-analysis. J Clin Anesth. 2019;52:99–104.
Crossref
12 Singh B, Anand A, Attri JP. A prospective open-label randomized controlled trial to compare intrathecal 1% 2-chloroprocaine versus 0.5% bupivacaine in ambulatory elective surgeries. Anesth Essays Res. 2020;14(2):266–70.
Crossref
13 Sugumar M, Singh AK, Rath A, Jayanthi A. A comparative study of isobaric 1% chloroprocaine with 0.5% isobaric bupivacaine for ureteroscopic lithotripsy under spinal anaesthesia: a prospective randomised study. Cureus. 2022;14(4):e24633.
14 Camponovo C, Wulf H, Ghisi D, Fanelli A, Riva T, Cristina D, et al. Intrathecal 1% 2-chloroprocaine vs. 0.5% bupivacaine in ambulatory surgery: a prospective, observer-blinded, randomised, controlled trial. Acta Anaesthesiol Scand. 2014;58(5):560–6.
Crossref PubMed
15 Hejtmanek MR, Pollock JE. Chloroprocaine for spinal anesthesia: a retrospective analysis. Acta Anaesthesiol Scand. 2011;55(3):267–72.
Crossref PubMed
16 Gu L, Smith CR, Ihnatsenka B, Zasimovich Y, Le-Wendling L. Comparing spinal chloroprocaine to hyperbaric and isobaric bupivacaine for total hip and knee arthroplasties: a retrospective study. Cureus. 2023; 15(3):e35729.
PubMed PMC
17 Post anesthesia care unit (PACU) discharge criteria PACU to inpatient unit [guideline]. Vancouver Coastal Health; 2019 Jan [cited 2023 Aug]. Available from: http://shop.healthcarebc.ca/vch/VCHDSTs/D-00-07-30231.pdf
18 Clorotekal® (chloroprocaine HCl injection USP): temporary importation of US-labelled Clorotekal (chloroprocaine HCl, inj USP 1%) to address a medical need in Canada. B Braun of Canada Ltd; 2018 [cited 2024 Aug]. Available from: https://www.bbraun.ca/en/products/b1/clorotekal-chloroprocainehciinjectionusp.html
19 Bupivacaine hydrochloride injection, USP [product monograph]. Baxter Corporation; 2019 May 23 [revised 2023 Dec 6; cited 2024 Aug]. Available from: https://pdf.hres.ca/dpd_pm/00074123.PDF
20 Ghisi D, Boschetto G, Spinelli AM, Giannone S, Frugiuele J, Ciccarello M, et al. Spinal anesthesia with chloroprocaine HCl 1% for elective lower limb procedures of short duration: a prospective, randomized, observer-blind study in adult patients. BMC Anesthesiol. 2021;21(1):58.
Crossref PubMed PMC
21 Alhefeiti K, Patrascu AM, Lustig S, Aubrun F, Dziadzko M. Perioperative outcomes in patients who received spinal chloroprocaine for total hip or knee arthroplasty, consecutive case series study. J Clin Med. 2022;11(19):5771.
Crossref PubMed PMC
Address correspondence to: Meghan Ludlam, Lower Mainland Pharmacy Services, Vancouver General Hospital, 2733 Heather Street, Vancouver BC V5Z1M9, email: mludlam@providencehealth.bc.ca
Competing interests: Meghan Ludlam has served in the volunteer role of Student Ambassador for the BC Pharmacy Association (unrelated to the study reported here). No other competing interests were declared.
Funding: None received.
Submitted: March 25, 2024
Accepted: September 5, 2024
Published: March 12, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 1, 2025