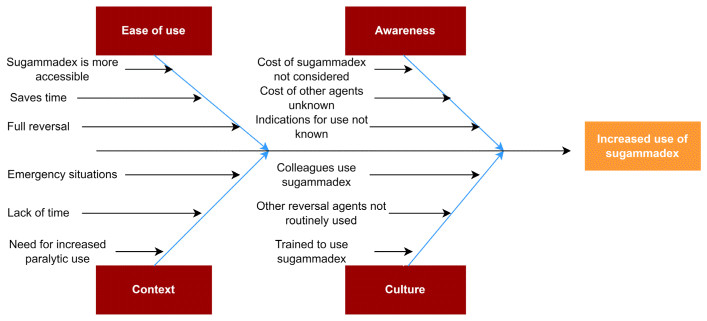
FIGURE 1 Fishbone diagram outlining factors contributing to increased sugammadex usage.
Erin Ingoldsby, Eric Romeril, April Liu, Tal Levit, and Ekta KhemaniTo cite: Ingoldsby E, Romeril E, Liu A, Levit T, Khemani E. Reducing sugammadex expenditure through educational initiatives in an urban tertiary care hospital system: a cost-reduction study. Can J Hosp Pharm. 2025;78(1):e3648. doi: 10.4212/cjhp.3648
ABSTRACT
Background
Sugammadex, a selective relaxant binding agent, works by encapsulating rocuronium or vecuronium to reverse neuromuscular blockade. It is an asset in emergencies requiring expedient reversal of neuromuscular blockade, such as situations involving difficult airways. A prevalent concern relating to sugammadex use is its higher cost compared with traditional reversal agents, contributing to more than 20% of operating room drug expenditures at Hamilton Health Sciences (Hamilton, Ontario) in 2019/20.
Objective
To use iterative Plan-Do-Study-Act cycles to reduce annual sugammadex expenditure at Hamilton Health Sciences by 50% from costs in the 2020/21 fiscal year.
Methods
Five Plan-Do-Study-Act cycles were implemented (January to April 2021) to increase awareness about the cost of sugammadex and appropriate indications for its use. The organization’s Department of Pharmacy supplied data regarding acquisition of sugammadex before, during, and after the Plan-Do-Study-Act cycles, which were analyzed quantitatively. A data set regarding difficult airways at Hamilton Health Sciences was analyzed alongside the sugammadex data to aid in understanding the need for accessibility of this drug.
Results
Use of sugammadex in fiscal year 2021/22 at Hamilton Health Sciences decreased by 75% from the previous fiscal year, without compromising patient safety or drug availability. Analysis of the difficult airway data revealed that both anticipated and unanticipated cases of difficult airway were still prevalent across all hospitals, which highlights the importance of maintaining access to and knowledge of how to use sugammadex in the setting of difficult airways.
Conclusions
This cost-reduction project showed that user-based program designs can have a meaningful impact on resource utilization. Additionally, the difficult airway data set emphasized the clear need to maintain availability of and capabilities for administering sugammadex, given the prevalence of emergency airway situations.
Keywords: cost reduction, anesthesia, resources, patient safety, drug utilization review, sugammadex, hospital pharmacy service
RÉSUMÉ
Contexte
Le sugammadex, un agent de liaison sélectif des relaxants, agit en encapsulant le rocuronium ou le vécuronium pour inverser le blocage neuromusculaire. Il constitue un atout précieux en cas d’urgence nécessitant une inversion rapide du blocage neuromusculaire, comme dans les situations impliquant une gestion difficile des voies aériennes. Une préoccupation courante liée à l’utilisation du sugammadex est son coût plus élevé par rapport aux agents d’inversion traditionnels. Il représentait plus de 20 % des dépenses en médicaments des salles d’opération de l’autorité sanitaire Hamilton Health Sciences (à Hamilton, en Ontario) en 2019–2020.
Objectif
Utiliser des cycles itératifs Planifier-Faire-Vérifier-Agir (PFVA) pour réduire de 50 % les dépenses annuelles de sugammadex de l’autorité sanitaire Hamilton Health Sciences par rapport aux coûts de l’exercice 2020–2021.
Méthodologie
Cinq cycles PFVA ont été mis en oeuvre (de janvier jusqu’en avril 2021) pour sensibiliser le personnel au coût du sugammadex et aux indications appropriées relatives à son utilisation. Le département de pharmacie de l’organisme a fourni des données sur l’acquisition de sugammadex avant, pendant et à la fin de la série de cycles PFVA, qui ont été analysées quantitativement. Un ensemble de données concernant la gestion difficile des voies aériennes à l’autorité sanitaire Hamilton Health Sciences a été analysé en parallèle avec les données sur le sugammadex afin de mieux comprendre la nécessité d’assurer l’accessibilité de ce médicament.
Résultats
L’utilisation du sugammadex à l’autorité sanitaire Hamilton Health Sciences pendant l’exercice 2021–2022 a diminué de 75 % par rapport à l’exercice précédent, sans compromettre la sécurité des patients ni la disponibilité du médicament. L’analyse des données portant sur la gestion difficile des voies aériennes a révélé que les cas anticipés et non anticipés de gestion difficile demeuraient fréquents dans tous les hôpitaux, ce qui souligne l’importance de maintenir l’accès au sugammadex et de maîtriser son utilisation dans le contexte de la gestion difficile des voies aériennes.
Conclusions
Ce projet de réduction des coûts a démontré que des approches centrées sur les utilisateurs peuvent avoir des conséquences importantes sur l’utilisation des ressources. Par ailleurs, l’analyse des données relatives à la gestion difficile des voies aériennes a mis en évidence la nécessité indéniable de maintenir la disponibilité du sugammadex et les compétences pour son administration, compte tenu de la fréquence des situations d’urgence impliquant des voies aériennes.
Mots-clés: réduction des coûts, anesthésie, ressources, sécurité des patients, examen de l’utilisation des médicaments, sugammadex, service de pharmacie hospitalière
Neuromuscular blockade (NMB) serves as a critical instrument in the anesthesiologist’s toolbox, facilitating the establishment of optimal surgical conditions and enabling the anesthesiologist to safely intubate and ventilate the patient.1,2 While the benefits of neuromuscular blocking agents (e.g., succinylcholine, rocuronium) are clear, their use also carries potential risks, such as residual NMB and postoperative pulmonary complications, emphasizing the importance of NMB monitoring and reversal.2,3
Traditionally, neostigmine was used to reverse NMB, despite limitations such as inability to reverse moderate or deep NMB, delayed onset of action, risk of residual NMB, and possible muscarinic adverse events.3,4 Recent guidelines of the American Society of Anesthesiologists5 and the European Society of Anaesthesiology and Intensive Care6 have recommended the use of neostigmine and glycopyrrolate for reversal only at minimal depth of NMB, preferring other agents such as sugammadex at deep, moderate, and shallow depths of NMB. Both guidelines similarly recommend the use of quantitative train-of-four monitoring when a neuromuscular blocking agent is used, which can help determine the degree of NMB and decrease postoperative complications.5,6
Sugammadex is a selective relaxant binding agent, the importance of which has become rapidly established in the operating room (OR). Sugammadex works by encapsulating rocuronium or vecuronium, thus reversing NMB and providing rapid and predictable recovery from NMB during surgery.1,7 The drug’s exceptional efficiency and specificity have made it a dependable asset in a variety of scenarios, including emergencies in which expedient NMB reversal is required, such as the management of anticipated and unanticipated difficult airway, where the patient can be ventilated but intubation is unsuccessful.8 In light of these factors, the 2023 Canadian Anesthesiologists’ Society guidelines state that health facilities must make selective relaxant binding agents readily available.9
Sugammadex significantly reduces the risk of residual NMB and has a high safety profile, appearing to be well tolerated across diverse patient populations, including pediatric, elderly, and morbidly obese populations, as well as patients with cardiac, renal, hepatic, or specific neuromuscular diseases.1,10 Furthermore, sugammadex allows for flexibility with planning and management of anesthesia by permitting a method for reversing profound NMB at the end of surgery, promoting faster OR turnover times, and ultimately enhancing efficiency.1,6,11 The disadvantages of sugammadex, as described in the literature, include increased patient pain, as well as nausea, vomiting, drug interactions (e.g., decreased effectiveness of oral hormonal contraceptives), hypersensitivity reactions, arrhythmias, QT interval prolongation, and effects on anticoagulation.1,7,10 However, these events are rare, and their incidence is lower than with traditional reversal agents, such as neostigmine.12
Despite the potential of sugammadex as a life-saving intervention, a prevalent concern is its higher cost (about $110 for 2 mg/2 mL vial and $220 for 5 mg/5 mL vial in 2019) compared with traditional reversal agents, such as neostigmine (about $9.50 for 10 mg/10 mL vial in 2019).13 This cost difference is especially pronounced in countries with a publicly funded health care system, such as Canada, and is an issue we experienced in the Hamilton Health Sciences system.14 In recent years, the push from publicly funded health care systems to reduce operating costs has forced hospital systems to attempt to identify areas where cost-saving efforts can be employed. Previous quality improvement studies have demonstrated that decreasing the use of sugammadex can reduce monetary expenditures; however, this may confer a risk to patient safety that needs to be considered and addressed.15 More specifically, with decreased use of a product, it is expected that both availability and technical proficiency will decline; in the case of sugammadex, such decline could ultimately compromise patient safety in emergency situations where a difficult airway is encountered. This problem inspired a cost-reduction study, in which we sought to reduce annual expenditure on sugammadex by 50% while improving education about appropriate indications for use of this drug and ensuring no compromise of patient safety.
Following the authorization of sugammadex use in Canada in 2016, the popularity of this agent rapidly increased among staff anesthesiologists. Through discussion with Hamilton Health Sciences staff, we were able to identify causative factors for the increased use and organized our findings into a fishbone diagram (Figure 1). The main factors were ease of use, departmental culture, lack of awareness regarding the costs of and/or indications for sugammadex use, and convenient and rapid full reversal of NMB. Departmental review showed that an increasing proportion of the budget each year was dedicated to the acquisition of sugammadex, reaching more than 20% of OR drug expenditures in 2019/20. This led Hamilton Health Sciences to conduct a survey comparing annual expenditure for sugammadex across several regional hospital systems. Among the 4 hospital systems with more than 500 beds that completed the survey, Hamilton Health Sciences was one of the largest consumers of sugammadex (unpublished data). These results raised questions as to whether sugammadex was being accessed inappropriately and, consequently, what the indications for appropriate sugammadex use are. These investigations also made apparent the variation in availability of sugammadex across different locations within each hospital. The research team, composed of staff members from the Department of Anesthesia and the Department of Pharmacy at Hamilton Health Sciences, drew on these findings to develop a study aimed at recognizing current reasons for use of sugammadex and providing education to staff regarding appropriate reasons for use, with the ultimate goal of reducing costs.
|
| ||
|
FIGURE 1 Fishbone diagram outlining factors contributing to increased sugammadex usage. | ||
Through discussion with anesthesia staff at Hamilton Health Sciences, the following issues contributing to injudicious use of sugammadex were identified: poor understanding of the reasons to access the reversal agent, lack of knowledge of the indications approved by the Pharmacy & Therapeutics Committee, lack of awareness of Canadian Anesthesiologists’ Society guidelines regarding neuromuscular blocking agent reversal and train-of-four monitoring, scant information provided on sugammadex costs, and widespread availability of both sizes (2- and 5-mL vials) in all ORs across the hospital system. Five Plan-Do-Study-Act (PDSA) cycles were planned between January and April 2021 to increase awareness about appropriate indications for sugammadex use and thus to reduce annual expenditure on the drug.
The first PDSA cycle, which occurred in January and February 2021, consisted of displaying a medication cost poster at all narcotic cupboards to increase awareness about the cost of sugammadex relative to other medications and reversal agents. PDSA cycles 2 and 3 were implemented together in February and March and consisted of holding educational sessions for staff regarding indications for sugammadex use and effective train-of-four monitoring, as well as implementing “reason for use” sign-out sheets at all places where staff accessed sugammadex. The reasons for use, as determined by the Pharmacy & Therapeutics Committee, were difficult airway situations, profound/deep residual NMB, ineffective reversal with maximum doses of neostigmine and glycopyrrolate, unexpected termination of surgery, contraindication to neostigmine and glycopyrrolate, persistent blockade in patients with morbid obesity, rocuronium anaphylaxis, and procedures requiring profound block until the end of surgery. PDSA cycles 4 and 5 were implemented together in March and April, just before Hamilton Health Sciences installed new automated drug cabinets (ADCs) in the clean core area of each OR. These last 2 cycles focused on removing the majority of 5-mL vials from previous locations, leaving 4 vials of this size on each difficult airway cart and 3 vials of this size on every drug cart for emergencies, thus encouraging use of the 2-mL vials as the first-line option. Additionally, to help ensure a smooth transition, educational sessions concerning these changes and associated new processes for accessing sugammadex from the ADCs and difficult airway carts were held.
A data set collected between September 2019 and June 2020 regarding the number of difficult airways at Hamilton Health Sciences was also analyzed to assess the need for access to sugammadex in numerous locations.
The primary outcome measure of this cost-reduction project was annual spending on sugammadex following implementation of the 5 PDSA cycles. The secondary measures were the number of difficult airways and reasons for their occurrence at Hamilton Health Sciences, to allow evaluation of the necessity of making reversal agents such as sugammadex readily accessible.
Cost data were collected by the Department of Pharmacy at Hamilton Health Sciences based on monthly acquisition of sugammadex and were analyzed in R software (2020 version; RStudio Team). Reasons for trends in the data were identified through discussion between Department of Anesthesia and Department of Pharmacy colleagues. Notably, the purchasing spike in early 2022 correlated with the period when pharmacy had to stock a fleet of ADCs and was thus not attributable to use by the anesthesia team; analysis of the data took this anomaly into account.
The difficult airway data were made available by the Department of Anesthesia at Hamilton Health Sciences, which had previously recorded occurrences of difficult airway intubations in an anonymized, secure fashion. Quantitative data regarding the number of discrete “difficult airway” situations were collected from documentation by anesthesiologists between September 2019 and June 2020 and were recorded in a spreadsheet. These data were then accessed by the research team and analyzed to identify trends such as prediction of difficult airways, reasons for difficulty, and techniques for achieving ventilation and intubation.
All data were anonymized and contained no patient-specific information. Additionally, no patient information was accessed at any stage of this cost-reduction project. A primary ethical consideration when designing this cost-reduction study was ensuring that despite any changes to patterns of use, acquisition, and distribution of sugammadex, this drug would be available when required and in all locations, ensuring adherence to Canadian Anesthesiologists’ Society guidelines and improving patient safety.9 No other ethical considerations were involved in the development of this cost-reduction study.
Before the intervention, the baseline annual cost of sugammadex had been increasing at a rate of 38% annually between fiscal years 2018/19 and 2020/21, which put Hamilton Health Sciences on track to spend about $310 000 in fiscal year 2021/22, as predicted by the line of best fit (Figure 2). However, actual sugammadex expenditure in fiscal year 2021/22, following the PDSA intervention, was $62 805, including returns for expired products that occurred in 4 of the 12 months (Figure 3); this represented a 75% reduction from the $246 950 expenditure in the previous fiscal year (2020/21). Monthly expenditures were reviewed by the Department of Pharmacy before, during, and after the PDSA cycles and were then used to calculate the total annual expenditure. Over the period January to June 2021, during which the PDSA cycles occurred, the monthly expenditure declined from $24 824 in January to $7472 in June, which clearly illustrated the immediate effects of this educational initiative.
|
| ||
|
FIGURE 2 Spending on sugammadex in consecutive fiscal years before the intervention, along with line of best fit showing projected spending in fiscal year 2021/22. | ||
|
| ||
|
FIGURE 3 Monthly expenditure on sugammadex at Hamilton Health Sciences before, during, and after implementation of the Plan-Do-Study-Act (PDSA) cycles. The PDSA cycles took place between January and April 2021. Negative values shown in late 2021 are due to pharmacy returns of sugammadex. | ||
The combined annual costs of neostigmine and glycopyrrolate were compared over the same timeframe and showed a 6% decrease from 2017/18 to 2018/19 and a 0.05% increase from 2018/19 to 2019/20. Furthermore, annual costs of rocuronium over the same periods showed a 0.05% increase, which indicates that the increased use of sugammadex was not related to a greater volume of OR cases overall or an increase in the proportion of cases requiring paralysis. These data demonstrate that the increases in sugammadex use and cost in the years before the PDSA intervention did not result from replacing neostigmine and glycopyrrolate use (Figure 4) or from higher case volumes.
|
| ||
|
FIGURE 4 Monthly expenditure on common operating room drugs at Hamilton Health Sciences. Susp = suspension. For each drug, the solid line represents the rolling average, whereas individual data points represent monthly values. | ||
Between September 2019 and June 2020, a total of 32 cases of documented difficult airway occurred at Hamilton Health Sciences, 5 of which involved pediatric patients. Of these difficult airway cases, 24 had been predicted to be difficult, 6 were documented as unpredicted, and 2 did not have a documented prediction status. Among those with predicted difficult airways, the most frequently cited reasons for difficult intubation were anterior larynx, immobile epiglottis, and large tongue. Among those with unpredicted difficult airways, the first 2 aforementioned factors remained the most prevalent, with the third being hypertrophied or significantly calcified arytenoid structures. Among both groups, the most frequently documented technique used to achieve manual ventilation was a combination of mask, oral airway, deep general anesthesia, and NMB, whereas the most cited method for achieving intubation was video laryngoscopy. Of note, these data are suspected to underestimate the actual number of difficult airways, given that not all anesthesiologists documented or submitted encountered instances of difficult airways.
Sugammadex is a valuable medication in anesthesia practice. Its remarkable efficacy, rapid onset of action, and predictability in reversing NMB make it indispensable for dealing with emergencies that arise in the OR and elsewhere. One drawback of this medication is its substantial cost, which necessitates thoughtful consideration to optimize access in situations where the drug is essential for patient safety, while decreasing inappropriate use and ensuring appropriate resource allocation. The interventions outlined in this cost-reduction study aimed to reduce annual sugammadex expenditure by more than 50% from the previous fiscal year by focusing on educating staff about the indications for appropriate use of sugammadex, and this goal was met.
Following implementation of the 5 PDSA cycles, sugammadex use at Hamilton Health Sciences declined by 75% from the previous fiscal year, with no compromise in patient safety. We demonstrated that with adequate educational initiatives focused on the cost of and reasons for using sugammadex, hospitals can reduce use of this drug and relieve financial strain on annual OR budgets. However, any changes must also ensure ready accessibility of sugammadex in situations where it is necessary.
Analysis of the data regarding difficult airway situations at Hamilton Health Sciences revealed that both anticipated and unanticipated difficult airways are still prevalent across all hospitals. These data demonstrate that difficult airways can occur for numerous reasons and highlight the importance of having access to reversal agents such as sugammadex and knowing how to effectively administer them. Emphasizing these data is important because although reducing sugammadex utilization has economic benefits, patient safety must not be compromised; in particular, the guidelines of the Canadian Anesthesiologists’ Society should be followed.
The study had potential limitations. Education about appropriate reasons for using sugammadex was delivered during PDSA cycles, and “reasons for use” sign-out sheets were implemented to assess understanding. However, the completion rate for these forms was exceedingly low, likely due to factors such as staff motivation and time constraints. As such, this study relied mainly on changes in sugammadex expenditures, as tracked by the Department of Pharmacy, to assess the effectiveness of the PDSA cycles. Moreover, data on sugammadex use pertaining to the 5-mL and 2-mL vials following PDSA cycles 4 and 5 could not be collected as accurately as the expenditure data, which limited our ability to determine the effectiveness of these initiatives and meant that we relied solely on expenditures for analysis of those cycles. Additionally, it was intended that time spent in the postanesthetic care unit would be monitored during the implementation of and following the PDSA cycles, as a balancing measure; however, these data were significantly confounded by other variables, such as postoperative nausea and vomiting and availability of ward beds, and thus were not representative as a balancing measure. Finally, the difficult airway data set was obtained through requests to anesthesiologists at Hamilton Health Sciences that they record this information; however, it is likely that some staff did not do so, such that the actual number of difficult airway cases was likely higher than the recorded data indicate.
Using quality improvement methodology, specifically PDSA cycles, we were able to safely and effectively reduce annual expenditure on sugammadex by more than 50% while ensuring that adequate stocking and availability of sugammadex were not compromised. These data show that user-based program designs can have a meaningful impact on resource utilization while also demonstrating the clear need to maintain availability of and capabilities for administering sugammadex, given the prevalence of emergency airway situations. The financial results of our initiative show that with proper education and appropriate use of sugammadex, economic savings can be achieved; however, it is also clear that difficult airways continue to be prevalent in the setting of anesthesia, so any changes in approach need to be balanced with adequate availability of and user experience with sugammadex for emergency airway situations.
1 Keating GM. Sugammadex: a review of neuromuscular blockade reversal. Drugs. 2016;76(10):1041–52.
Crossref PubMed
2 Cook D, Simons DJ. Neuromuscular blockade. In: StatPearls. StatPearls Publishing; 2023 Jan- [updated 2022 Sep 24; cited 2023 April 16]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538301/
3 Adeyinka A, Layer DA. Neuromuscular blocking agents. In: StatPearls. StatPearls Publishing; 2023 Jan- [updated 2022 Nov 28; cited 2023 April 13]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537168/
4 Shah SB, Chawla R, Pahade A, El-Molla A. Neuromuscular blockers and their reversal: have we finally found the on-off switches? Ain Shams J Anesthesiol. 2021;13:15.
Crossref
5 Thilen SR, Weigel WA, Todd MM, Dutton RP, Lien CA, Grant SA, et al. 2023 American Society of Anesthesiologists practice guidelines for monitoring and antagonism of neuromuscular blockade: a report by the American Society of Anesthesiologists Task Force on Neuromuscular Blockade. Anesthesiology. 2023;138(1):13–41.
Crossref
6 Fuchs-Buder T, Romero CS, Lewald H, Lamperti M, Afshari A, Hristovska AM, et al. Peri-operative management of neuromuscular blockade: a guideline from the European Society of Anaesthesiology and Intensive Care. Eur J Anaesthesiol. 2023;40(2):82–94.
Crossref
7 Yang LPH, Keam SJ. Sugammadex. Drugs. 2009;69(7):919–42.
Crossref PubMed
8 Motamed C. Sugammadex in emergency situations. J Pers Med. 2023; 13(1):159.
Crossref PubMed PMC
9 Dobson G, Chau A, Denomme J, Fuda G, McDonnell C, McIntyre I, et al. Guidelines to the practice of anesthesia: revised edition 2023. Can J Anesth. 2023;70(1):16–55.
Crossref PubMed
10 Honing G, Martini CH, Bom A, van Velzen M, Niesters M, Aarts L, et al. Safety of sugammadex for reversal of neuromuscular block. Expert Opin Drug Saf. 2019;18(10):883–91.
Crossref PubMed
11 Carron M, Baratto F, Zarantonello F, Ori C. Sugammadex for reversal of neuromuscular blockade: a retrospective analysis of clinical outcomes and cost-effectiveness in a single center. Clinicoecon Outcomes Res. 2016;8:43–52.
Crossref PubMed PMC
12 Hristovska AM, Duch P, Allingstrup M, Afshari A. The comparative efficacy and safety of sugammadex and neostigmine in reversing neuromuscular blockade in adults. A Cochrane systematic review with meta-analysis and trial sequential analysis. Anaesthesia. 2018;73(5): 631–41.
Crossref
13 Voluntary compliance undertaking of Merck Canada Inc. to the Patented Medicine Prices Review Board. Patented Medicine Prices Review Board; 2019 [cited 2024 Jul 20]. Available from: https://www.pmprb-cepmb.gc.ca/view.asp?ccid=1343.
14 Cohen JC, Latchford KJ. Sugammadex in Ontario hospitals: access and institutional policies. J Eval Clin Pract. 2020;26(1):50–5.
Crossref
15 Pimentel, MP, Billings F, Sivashanker KS, Pankaj S, Fields K, Cotugno M, et al. Reducing medication waste while improving access to sugammadex: a quality improvement project in medication stewardship. A A Pract. 2020;14(7):e01223
Crossref PubMed
Address correspondence to: Dr Ekta Khemani, Hamilton General Hospital, 237 Barton Street East, Hamilton ON L8L 2X2, email: khemane@mcmaster.ca
Competing interests: For activities unrelated to the study reported here, Eric Romeril has received consulting fees from the Institute for Safe Medication Practices Canada, speaker’s honoraria from McMaster University and Mohawk College, and in-kind support from Stryker and Daniels; and Ekta Khemani has received speaker’s honoraria from Niagara College. No other competing interests were declared.
Funding: None received.
Submitted: May 10, 2024
Accepted: August 27, 2024
Published: February 12, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 1, 2025