Case Report
Management of Status Epilepticus and Infections in a Patient with Lennox Gastaut Syndrome
Amanda Y Leong, Tom Gieni, Christopher Doig, Daniel J Niven, and Julie Kromm
To cite: Leong AY, Gieni T, Doig C, Niven DJ, Kromm J. Management of status epilepticus and infections in a patient with Lennox Gastaut syndrome. Can J Hosp Pharm. 2025;78(1):e3667. doi: 10.4212/cjhp.3667
INTRODUCTION
The management of antiseizure medications (ASMs) can pose several therapeutic challenges for critically ill patients. Appropriate management of drug interactions for patients in the intensive care unit (ICU) reduces adverse events and morbidity, but requires consideration of complex medication regimens, altered pharmacokinetics and pharmacodynamics, and pathophysiology.1 In a retrospective cohort study in the ICU setting, the 144 adult patients had a total of 395 medication orders, for which there were 1776 potential drug–drug interactions (based on the Lexi-Interact database). 2 Major interactions, defined as literature-supported possible clinical effects and potentially life-threatening effects, accounted for 23.5% of these potential interactions, and moderate interactions, defined as having possible clinical relevance, for 71.4%.2
We describe here the considerations for management of drug–drug interactions between ASMs and anti-infective agents, as well as drug–disease interactions involving anti-infectives, in a critically ill patient.
CASE REPORT
A 38-year-old woman with Lennox-Gastaut syndrome underwent corpus callosotomy.* She was admitted to the ICU on March 14, 2022 (postoperative day 4) for nonconvulsive status epilepticus, which caused altered level of consciousness and airway compromise. Her past medical history included asthma, chronic headache, and fractures from falls due to atonic seizures.
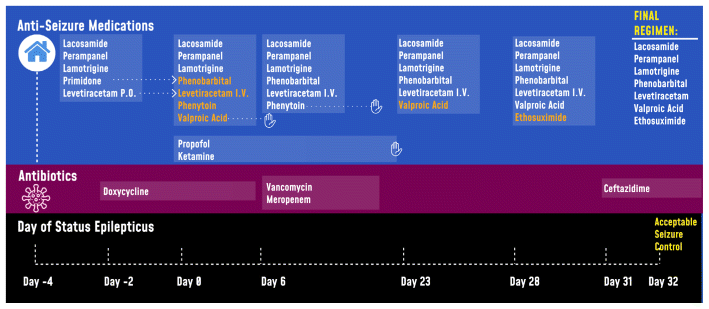
At baseline, the patient was experiencing generalized tonic seizures twice daily; atonic seizures 4 times per month; focal impaired awareness seizures twice weekly; and typical and atypical absence seizures 2 and 4 times daily, respectively. Home ASMs were lacosamide 300 mg twice daily; perampanel 10 mg daily; primidone 187.5 mg twice daily; levetiracetam 1500 mg in the morning, 500 mg in the afternoon, and 1500 mg at bedtime; and lamotrigine 350 mg twice daily (Figure 1).
| |
| |
|
FIGURE 1 Timeline of antiseizure medications and antibiotic therapy in the intensive care unit (ICU). The patient underwent corpus callosotomy on day −4, with status epilepticus occurring on day 0 (March 14, 2022), at which time she was admitted to the ICU. |
During her 64-day course in ICU, the patient experienced 3 distinct episodes of refractory status epilepticus. The cause was thought to be a combination of perioperative sleep deprivation, postoperative cerebral edema, missed administration of oral ASMs (secondary to altered level of consciousness), and culture-negative sepsis.
Upon admission to the ICU, the patient’s benzodiazepine doses were limited due to concerns regarding airway compromise before intubation. Phenytoin 2 g intravenous load (followed by 125 mg orally 3 times daily) was administered, given previous success with this drug in terminating the patient’s status epilepticus; unfortunately there was no sustained benefit. Therefore, the decision was made to intubate and initiate propofol and ketamine. The intention was to reduce the burden of electrographic seizures (as shown by continuous electroencephalography [EEG] monitoring) during titration of ASMs. Given that refractory status epilepticus is characterized by downregulation of γ-aminobutyric acid (GABA) receptors and upregulation of N-methyl-d-aspartate (NMDA) receptors,3 propofol was given for GABA antagonism, with ketamine used as an adjunct anesthetic to provide NMDA antagonism.
The patient’s preoperative home ASM regimen was continued in the ICU. To optimize time to peak concentration, in the context of missed oral doses and ongoing seizures, levetiracetam was administered intravenously, and primidone therapy was switched to phenobarbital 1 g intravenous loading dose, followed by 200 mg daily. Phenobarbital is an active metabolite of primidone that enhances inhibition mediated by GABA type A (GABAA) receptors, reduces glutamate-mediated excitation, and alters sodium and potassium conductance.4 Valproic acid was subsequently added to the regimen, initially as a 4 g intravenous loading dose, then as a maintenance dose of 1 g orally 3 times daily to target NMDA receptor inhibition and to further increase levels of GABA, by both activating GABA synthesis through glutamic acid decarboxylase and inhibiting GABA degradation at the synaptic cleft.4 Given the patient’s baseline seizure frequency, freedom from seizures was not the goal. Instead, ASMs were titrated to achieve fewer than 6–10 seizures daily and to improve the patient’s level of consciousness.
Doxycycline initiated on the postoperative ward for suspected aspiration was continued in the ICU, and by status epilepticus day 6, the regimen was changed to a broad course of antibiotics, including meropenem and vancomycin for culture-negative sepsis and empiric coverage for possible nosocomial meningitis. On the postoperative ward, valproic acid levels were therapeutic (353 μmol/L) but had begun to fall in the ICU, likely due to a hypermetabolic state. In light of known drug interactions with meropenem, the already-dropping subtherapeutic level, and the minimal initial effect on seizure burden, valproic acid was discontinued. During this infectious period, phenobarbital levels declined from 85 μmol/L to 62 μmol/L, and the medication was titrated to 210 mg intravenously twice daily. Phenytoin levels were monitored to ensure optimization (total phenytoin 26 μmol/L, albumin 17 g/L, corrected phenytoin 59 μmol/L).
By day 23 of status epilepticus, the patient was weaned off propofol and ketamine in an attempted transition to non–infusion-based antiseizure therapy. Due to ongoing subtherapeutic phenytoin levels, this ASM was discontinued, and valproic acid was re-initiated, with an increase to 2000 mg orally 3 times daily. Lamotrigine was also empirically reduced because of the drug–drug interaction with valproic acid. Meropenem was stopped after completion of a 10-day course because no source of infection was isolated and because of this drug’s severe interaction with valproic acid. By day 28 of status epilepticus, ethosuximide was added to inhibit T-type calcium channels, and the patient remained off propofol and ketamine with no increase in seizure burden.3 On day 31 of status epilepticus, pan-sensitive Escherichia coli urosepsis was diagnosed. To treat this infection, ceftazidime was chosen over piperacillin–tazobactam, because penicillins lower the seizure threshold, and there was concomitant concern about the possibility of ventilator-acquired pneumonia. Acceptable seizure control was achieved on day 32, as confirmed by continuous EEG, with a regimen of phenobarbital 210 mg intravenously twice daily; valproic acid 2500 mg orally twice daily; ethosuximide 750 mg orally twice daily; lacosamide 300 mg orally twice daily; lamotrigine 150 mg orally twice daily; levetiracetam 1500 mg orally in the morning, 500 mg orally in the evening, and 1500 mg orally at bedtime; and perampanel 10 mg orally at bedtime. A percutaneous tracheostomy was placed on April 19, 2022 (day 36 of status epilepticus), with decannulation on June 20, 2022. The patient was transferred out of the ICU after 64 days.
DISCUSSION
The management of status epilepticus in critically ill patients requires careful attention to the choice of ASMs. Identifying and addressing any underlying cause for the seizures is crucial, as doing so may limit the need for multiple ASMs. The selection of ASMs depends on the type of seizure; the patient’s home regimen, compliance, and ASM levels; intolerances or allergies; comorbidities, including acute or chronic renal and/or hepatic dysfunction; other required medications; and strategic mixing of ASM mechanisms when more than 1 medication is required. Therapeutic drug monitoring, drug–drug interactions, and drug–disease interactions are also important considerations during management. Strategic and systematic selection and titration of ASMs are pivotal. Agents should be trialled individually, with close monitoring of the effect on seizure burden and other clinical indices, including potential adverse effects.
Both the administration of new medications and the withholding of home ASMs may contribute to lowering the seizure threshold, particularly in patients with epilepsy. For example, it is well known that specific antimicrobials can interact with ASMs, resulting in sub- or supra-therapeutic levels of the ASMs. The epileptogenic properties of some anti-infective agents are also an important consideration when selecting agents to treat infections (Table 1).
TABLE 1 Antimicrobials that Lower the Seizure Threshold
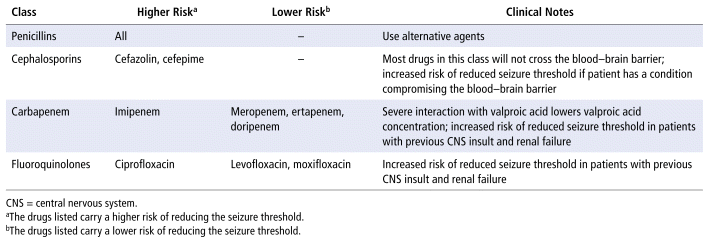
Penicillins may induce seizures because the β-lactam ring inhibits GABAA receptors and reduces the duration that GABAA receptors are open.5 Risk factors for penicillin-induced seizures include renal injury and increased permeability of the blood–brain barrier.6 Seizure risk is highest for penicillin G.5 In one case, a 63-year-old woman with Parkinson disease was treated with piperacillin–tazobactam for urinary sepsis and experienced EEG-confirmed nonconvulsive status epilepticus, which resolved after discontinuation of the antimicrobial.7 In another case, a 54-year-old man with microscopic polyangiitis experienced clinically diagnosed seizures with the use of piperacillin-tazobactam to treat a Legionella infection.8 Penicillin antibiotics should be used with caution to treat infections in patients with a seizure history, given the propensity of these drugs to reduce the seizure threshold. Alternative anti-infective agents should be considered.
Cephalosporins are thought to induce seizures through GABAA receptor antagonism and NMDA receptor agonism. 6 The seizure risk is reported to be highest for cefazolin and cefepime.6 Published case series have identified both electrographic status epilepticus and metabolic/toxic encephalopathy patterns on EEG tracings of patients with renal failure (cases involving chronic renal failure being reported more often than cases involving acute renal failure) and/or existing intracranial pathologies.6,9,10 Notably, in a review of 34 prior case reports with EEG findings, only 1 case was consistent with electrographic status epilepticus as defined by the American Clinical Neurophysiology Society’s critical care EEG terminology11; in the other 33 cases, EEG demonstrated triphasic waves suggestive of metabolic/toxic encephalopathy.10 A separate case report identified EEG-confirmed, ceftazidime-induced encephalopathy, which resolved with administration of clonazepam (as evidenced by subsequent EEG findings).12 Most cephalosporins do not easily cross the blood–brain barrier, and increased dosage is often required to optimize penetration for infections such as meningitis, ventriculitis, and encephalitis. Thus, patients with a disease or condition that increases the permeability of the blood–brain barrier are at risk for reduced seizure threshold.6 In the case reported here, ceftazidime was used, with careful monitoring by means of continuous EEG, to treat the patient’s ongoing sepsis.
Carbapenems have high variability in their epileptogenic properties.6 A meta-analysis of 169 randomized controlled trials showed that among patients exposed to carbapenems, seizures were experienced by an additional 2 (95% confidence interval [CI] 0.001 to 0.004) per 1000 persons, relative to those not exposed to carbapenems.13 The risk was driven by imipenem, with a risk difference of an additional 4 (95% CI 0.002 to 0.007) per 1000 persons experiencing seizures, whereas meropenem, ertapenem, and doripenem were not associated with significantly increased seizure risk.13 Yet when imipenem was compared directly with meropenem, the difference in risk of seizure was not statistically significant (risk difference 0.001, 95% CI −0.003 to 0.006).13 The odds of seizure with imipenem were further interrogated with sensitivity analyses (based on study design, use of dialysis, seizure history, and meningitis), which consistently showed elevated odds ratios for seizure, except in studies using a single-blind design.13 Risk factors for the development of seizures with carbapenems include higher dose, renal injury, history of central nervous system injury, and decreased albumin levels.6,13 Lower albumin levels and worse renal function increase the circulating concentration of carbapenem and its toxic metabolite.6
An important consideration with carbapenems is their known drug–drug interaction with valproic acid. A meta-analysis of 11 observational studies showed that the interaction of carbapenems and valproic acid resulted in a mean difference of −305.1 (95% CI −445.2 to −276.0) μmol/L of serum valproic acid concentration, dropping below the recommended reference range of 350 to 700 μmol/L over a median of 4 days.14 Notably, the meta-analysis had high heterogeneity. Sensitivity analyses for meropenem, imipenem, and ertapenem revealed that this heterogeneity was driven by the meropenem studies, but the reduction in serum levels of valproic acid for each carbapenem was still statistically significant, ranging from −216.3 to −311.3 μmol/L.14
In patients with a history of seizures, imipenem should be avoided. Meropenem, ertapenem, and doripenem may pose risks, and their use should be weighed against those of alternative treatment options with lower seizure risk. In patients who are taking valproic acid, avoidance of carbapenems is recommended; in scenarios where this is not possible, regular therapeutic drug monitoring (if available) is recommended. In the case reported here, we used strategies of discontinuing valproic acid upon initiation of meropenem and subsequently changing meropenem to an antibiotic with no known drug–drug interaction and lower risk of reducing the seizure threshold.
Fluoroquinolones have epileptogenic properties due to their ability to bind to GABA type B receptors and activate NMDA receptors, thus reducing the seizure threshold.6 Risk factors for seizures secondary to fluoroquinolones include history of central nervous system disorders, renal or hepatic failure, and electrolyte abnormalities.6 A systematic review of 143 studies showed that among fluoroquinolones, occurrence of generalized seizures was most frequently reported with ciprofloxacin, with fewer case reports for norfloxacin, levofloxacin, and moxifloxacin.15 In patients who may benefit from fluoroquinolone therapy and are at risk of seizure, it may be prudent to choose alternatives to ciprofloxacin, such as levofloxacin or moxifloxacin.
CONCLUSION
Clinical considerations for the treatment of infections in patients with seizures, epilepsy, or status epilepticus include careful selection and judicious use of antimicrobials for empiric therapy and culture-proven infection. When selecting and dosing antibiotics, clinicians should consider patient factors that affect the clearance, metabolism, and free fraction of ASMs, including renal or liver failure, reduced albumin levels, and intracranial pathologies. The dose of each antibiotic should be carefully targeted to the indication to avoid unnecessarily high doses, while ensuring optimized therapy. Close monitoring of ASMs, by means of therapeutic drug monitoring, if available, can help to ensure optimization of the patient’s therapy as well as management of infectious complications.
References
1 Spoelhof B, Farrokh S, Rivera-Lara L. Drug interactions in neuro-critical care. Neurocrit Care. 2017;27(2):287–96.
Crossref PubMed
2 Dagdelen MS, Gulen D, Ceylan I, Girgin NK. Evaluation of potential drug-drug interactions in intensive care unit. Eur Rev Med Pharmacol Sci. 2021;25(18):5801–6.
PubMed
3 Rossetti AO, Lowenstein DH. Management of refractory status epilepticus in adults: still more questions than answers. Lancet Neurol. 2011; 10(10):922–30.
Crossref PubMed PMC
4 Porter RJ, Meldrum BS. Antiseizure drugs. In: Katzung BG, Masters SB, Trevor AJ, editors. Basic and clinical pharmacology. 12th ed. McGraw-Hill Companies, Inc; 2012. p. 403–28.
5 Chow KM, Hui AC, Szeto CC. Neurotoxicity induced by beta-lactam antibiotics: from bench to bedside. Eur J Clin Microbiol Infect Dis. 2005; 24(10):649–53.
Crossref PubMed
6 Wanleenuwat P, Suntharampillai N, Iwanowski P. Antibiotic-induced epileptic seizures: mechanisms of action and clinical considerations. Seizure. 2020;81:167–74.
Crossref PubMed
7 Fernández-Torre JL, Santos-Sánchez C, Pelayo AL. De novo generalised non-convulsive status epilepticus triggered by piperacillin/tazobactam. Seizure. 2010;19(8):529–30.
Crossref PubMed
8 Man BL, Fu YP. Piperacillin/tazobactam-induced myoclonic jerks in a man with chronic renal failure. BMJ Case Rep. 2015;2015: bcr2015210184.
Crossref PubMed PMC
9 Bora I, Demir AB, Uzun P. Nonconvulsive status epilepticus cases arising in connection with cephalosporins. Epilepsy Behav Case Rep. 2016;6:23–7.
Crossref PubMed PMC
10 Triplett JD, Lawn ND, Chan J, Dunne JW. Cephalosporin-related neurotoxicity: metabolic encephalopathy or non-convulsive status epilepticus? J Clin Neurosci. 2019;67:163–6.
Crossref PubMed
11 Hirsch LJ, Fong MWK, Leitinger M, LaRoche SM, Beniczky S, Abend NS, et al. American Clinical Neurophysiology Society’s standardized critical care EEG terminology: 2021 version. J Clin Neurophysiol. 2021;38(1):1–29.
Crossref PubMed PMC
12 Jackson GD, Berkovic SF. Ceftazidime encephalopathy: absence status and toxic hallucinations. J Neurol Neurosurg Psychiatry. 1992; 55(4):333–4.
Crossref PubMed PMC
13 Cannon JP, Lee TA, Clark NM, Setlak P, Grim SA. The risk of seizures among the carbapenems: a meta-analysis. J Antimicrob Chemother. 2014;69(8):2043–55.
Crossref PubMed
14 Chai PYC, Chang CT, Chen YH, Chen HY, Tam KW. Effect of drug interactions between carbapenems and valproate on serum valproate concentration: a systematic review and meta-analysis. Expert Opin Drug Saf. 2020;20(2):215–23.
Crossref PubMed
15 Sutter R, Rüegg S, Tschudin-Sutter S. Seizures as adverse events of antibiotic drugs: a systematic review. Neurology. 2015;85(15):1332–41.
Crossref PubMed
Amanda Y Leong, BSc, BScPharm, ACPR, is with the Department of Pharmacy Services, Alberta Health Services, and the Department of Critical Care Medicine, Cumming School of Medicine, University of Calgary, Calgary, Alberta.
Tom Gieni, BSP, BCCCP, is with the Department of Pharmacy Services, Alberta Health Services, Calgary, Alberta.
Christopher Doig, MSc, MD, FRCPC, is with the Department of Critical Care Medicine, Alberta Health Services, and the Department of Critical Care Medicine, Cumming School of Medicine, University of Calgary, Calgary, Alberta.
Daniel J Niven, MD, MSc, PhD, FRCPC, is with the Department of Critical Care Medicine, Alberta Health Services, and the Department of Critical Care Medicine, Cumming School of Medicine, University of Calgary, Calgary, Alberta.
Julie Kromm, BMSc, MD, FRCPC, is with the Department of Critical Care Medicine, Alberta Health Services, and the Departments of Critical Care Medicine and Clinical Neurosciences, Cumming School of Medicine, University of Calgary, Calgary, Alberta.
Address correspondence to: Dr Daniel J Niven, Department of Critical Care Medicine, Cumming School of Medicine, University of Calgary, 1403 29 Street NW, Calgary AB T2N 2T9, email: Daniel.niven@albertahealthservices.ca
(Return to Top)
*Written informed consent for publication of the clinical details in this case was obtained from the patient’s parent.
Competing interests: None declared.
Funding: None received.
Submitted: June 17, 2024
Accepted: July 29, 2024
Published: January 15, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 1, 2025

