Chun-Yip Hon, Jackie Ellis, Tina I En Chiang, Matty Jeronimo, Rita Ciconte, and A Dana MénardTo cite: Hon CY, Ellis J, Chiang TIE, Jeronimo M, Ciconte R, Ménard AD. Adoption of closed-system drug transfer devices: effectiveness in reducing occupational exposure to hazardous drugs and the change management process. Can J Hosp Pharm. 2025;78(3):e3676. doi: 10.4212/cjhp.3676
ABSTRACT
Background
Closed-system drug transfer devices (CSTDs) are known to be effective in reducing hazardous drug contamination and, in turn, the risk of exposure for health care workers. In response, the Fraser Health Authority in British Columbia had plans to introduce CSTDs into practice.
Objectives
To confirm the effectiveness of CSTDs in reducing hazardous drug contamination and to understand health care workers’ perspectives regarding the change management process for CSTD implementation.
Methods
Surface wipe samples were collected at 4 health care facilities within the health authority over 3 time points: T0, 1 month before CSTDs were introduced; T1, 1 month after CSTDs were introduced; and T2, 5 months after implementation. Comparative analysis of drug contamination levels was conducted between 2 pairs of sampling times: T0 vs T1 and T1 vs T2. To understand the change management process, health care workers at the same departments (as those where wipe samples were collected) were surveyed.
Results
A total of 156 wipe samples were collected, consisting of 13 samples at each of the 4 sites at each of the 3 time points. There was a statistically significant reduction (p < 0.01) in the sum of the mass of drug contaminants from T0 to T1 (25.82 vs 4.01 ng/cm2) and from T1 to T2 (4.01 vs 0.068 ng/cm2). About 50 individuals responded to each question of the survey, and respondents had generally positive comments regarding the transition to CSTDs. Nevertheless, suggestions for improvement included offering various forms of training (e.g., online video, hands-on sessions) and ensuring ongoing communication.
Conclusions
CSTDs were confirmed to be effective in reducing surface contamination levels, and the change management process employed by the health authority appeared to be well received.
Keywords: health care, occupational exposure, hazardous drugs, closed-system drug transfer devices, change management
RÉSUMÉ
Contexte
Les dispositifs de transfert de médicaments en système fermé (CSTD) sont reconnus pour leur efficacité à réduire la contamination par des médicaments dangereux et, par conséquent, le risque d’exposition des travailleurs de la santé. Ainsi, l’autorité sanitaire Fraser Health en Colombie-Britannique prévoyait de les introduire dans la pratique.
Objectifs
Confirmer l’efficacité des CSTD pour réduire la contamination par des médicaments dangereux, et comprendre les points de vue des professionnels de la santé concernant le processus de gestion du changement lié à leur mise en œuvre.
Méthodologie
Des échantillons de surface ont été prélevés dans 4 établissements de santé relevant de l’autorité sanitaire, à 3 moments différents : à T0, soit un mois avant l’introduction des CSTD; à T1, soit un mois après leur mise en œuvre; et à T2, soit cinq mois après la mise en place. Une analyse comparative des niveaux de concentration des médicaments a été réalisée entre deux paires d’intervalles (T0 contre T1 et T1 contre T2). Afin de mieux comprendre le processus de gestion du changement, les travailleurs de la santé des services où les échantillons avaient été prélevés ont répondu à un sondage.
Résultats
Au total, 156 frottis de surface ont été recueillis, soit 13 échantillons à chacun des 4 établissements, à chacun des 3 moments d’échantillonnage. Une réduction statistiquement significative (p < 0,01) de la somme de la masse des contaminants médicamenteux a été observée entre T0 (25,82 ng/cm2) et T1 (4,01 ng/cm2), puis entre T1 (4,01 ng/cm2) et T2 (0,068 ng/cm2). Environs 50 personnes ont répondu à chaque question du sondage, et les répondants commentaient généralement positivement le passage aux CSTD. Néanmoins, des suggestions d’amélioration ont été formulées, notamment l’offre de formations dans différents formats (p. ex. : vidéos en ligne, séances pratiques) et en assurant une communication continue.
Conclusions
L’efficacité des CSTD pour réduire les niveaux de contamination de surface a été confirmée, et le processus de gestion du changement utilisé par l’autorité sanitaire semblait bien reçu.
Mots-clés: soins de santé, exposition professionnelle, médicaments dangereux, dispositifs de transfert de médicaments en système fermé, gestion du changement
Health care workers’ exposure to hazardous drugs is associated with adverse health effects, including reproductive toxicities (e.g., difficulty conceiving, spontaneous abortion), genetic abnormalities, and an increased risk of cancer.1–5 The US National Institute for Occupational Safety and Health (NIOSH) has indicated that more than 250 hazardous drugs may be found in health care settings,6 and it has been demonstrated that any worker involved with the hospital’s medication circuit (i.e., the process flow of drugs) is at risk of exposure to hazardous drugs.7 Essentially, such exposure can occur during receipt of drug shipments, while preparing drugs, when delivering prepared drugs to another department, while administering drugs, and during disposal of hazardous drug waste.7 CAREX (CARcinogen EXposure) Canada has estimated that approximately 79 000 Canadians, including health care workers, are occupationally exposed to antineoplastic agents.8
Exposure to hazardous drugs occurs primarily through dermal contact with drug-contaminated surfaces and/or objects.9,10 One Canadian study concluded that “frequently contacted surfaces at every stage of the hospital medication system had detectable levels of drug residue”.11 Those work surfaces with confirmed drug contamination included, but were not limited to, countertops, the biological safety cabinet (BSC), waste bins, IV pumps, and carts.11 Moreover, urinary samples from health care workers at the facilities where surface contamination was found had detectable levels of cyclophosphamide, a hazardous drug commonly used as a marker.12 These results demonstrate that physiological uptake is possible. As such, and because there are no established regulatory limits on occupational exposure to hazardous drugs, best practice dictates that exposure should be kept as low as reasonably practicable (a principle also known as ALARP).
One means of reducing the exposure risk is to employ closed-system drug transfer devices (CSTDs). A CSTD is defined as “a device that mechanically prohibits the transfer of environmental contaminants into the system and the escape of the hazardous drug or vapor concentrations outside the system”.13 Numerous studies have been conducted demonstrating the effectiveness of CSTDs in reducing surface contamination.14–17 As a result, a team of oncology clinicians and researchers renowned for their expertise in exposure to hazardous drugs recommended the use of CSTDs in health care settings.5 Moreover, CSTDs are referenced as a best practice in safe drug handling guidelines by various professional groups.13,18,19
In response, the Fraser Health Authority, in the province of British Columbia, had plans to adopt CSTDs for hazardous drug preparation and administration. The health authority intended to fully implement CSTDs for the preparation (in pharmacy) and administration (in clinical settings) of all hazardous drugs, where clinically appropriate (examples of clinically inappropriate situations include, but are not limited to, drug incompatibility with CSTD parts and administration requiring use of a needle, such as intramuscular administration). For the purposes of this study, hazardous drugs were those listed in the BC Provincial Health Authorities Hazardous Drug List, with the hazardous drugs in British Columbia being categorized into Groups 1 and 2 based on Tables 1 and 2, respectively, of the draft NIOSH List of Hazardous Drugs in Healthcare Settings, 2020 (https://www.cdc.gov/niosh/docket/review/docket233c/pdfs/DRAFT-NIOSH-Hazardous-Drugs-List-2020.pdf). The implementation of CSTDs into practice is analogous to the introduction of a novel control measure. For any newly introduced control measure, it is considered best practice to evaluate the intervention to ensure that it achieves the desired outcome,20,21 in this case, more specifically, whether CSTDs are indeed reducing levels of drug contamination. This type of evaluation should be done within each organization where the controls are introduced, as implementation may vary between workplaces.21 Furthermore, although CSTDs have been found to reduce drug contamination levels on surfaces, they do not eliminate contamination completely5,16,17; therefore, the potential for exposure remains and, from an occupational health and safety perspective, an organization must assess and respond to this possible risk.21
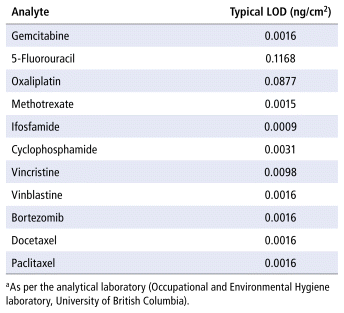
TABLE 1 Typical Limits of Detection (LODs)a for Hazardous Drugs Analyzed in This Study

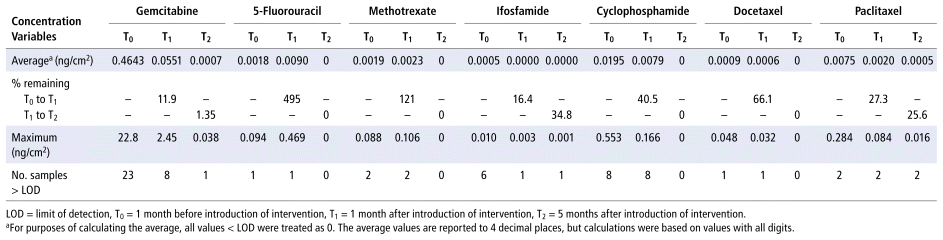
TABLE 2 Summary Statistics for Concentration Levels over Time for Hazardous Drugs That Were above LOD
The adoption of CSTDs will result in a change in practice for those who prepare or administer hazardous drugs. Best practice dictates that an organization should have a process to manage such a change, as it will affect workers’ health and safety.21,22 In addition, Maheshwari and Vohra23 concluded that the employer’s efforts with respect to culture, leadership, training, and communication during the change management process can influence employees’ perceptions and their resulting commitment to change. Given that this initiative to adopt CSTDs involved a large BC health authority, it was important to ascertain the perceptions of workers regarding the change management process (details of the 6-step change management process are available by request to the corresponding author). This undertaking served several purposes. First, the survey results are likely to benefit other facilities with plans to implement CSTDs. Second, the results may provide useful feedback for improvement of the change management process for other health and safety initiatives in the future and, in turn, facilitate a smoother transition.
Overall, the current study had 2 key objectives: to confirm the effectiveness of CSTDs in reducing surface contamination levels at several hospitals within the health authority and to ascertain workers’ perceptions about the process surrounding the adoption of CSTDs into practice. To our knowledge, no previous study has evaluated surface contamination levels before and after a CSTD intervention while also examining the change management process of incorporating these devices into routine practice.
The first part of the current study had a before-and-after design, the most common design employed by similar studies evaluating CSTD effectiveness.24 Our study specifically examined outcomes with the ChemoLock CSTD (ICU Medical), the model adopted for use at the Fraser Health Authority following a multidisciplinary contract selection process. The implementation of CSTDs occurred in October and November 2022. Three time points were selected to assess CSTD effectiveness: T0, 1 month before the introduction of CSTDs at the sites; T1, 1 month after initial introduction of the CSTDs; and T2, 5 months after introduction. The times for T1 and T2 were consistent with similar published studies.25–28
Wipe samples were collected from 4 participating acute care facilities within the health authority, and similar types of surfaces from each site were sampled at the 3 time points. The surfaces selected for sampling were in pharmacy and oncology units and are known (from discussions with front-line workers or the literature11,29) to be contaminated with hazardous drugs and/or are likely to be contacted by workers’ hands. Examples of such surfaces included the refrigerator door handle, the BSC, the floor in front of the BSC, and IV pumps. A total of 13 surface wipe samples were collected at each of the 4 participating sites at T0, with sample collection being replicated at T1 and T2, for a total of 156 wipe samples.
Surface wipe samples were collected using a validated method developed by Jeronimo and others.30 Briefly, sample collection involved using a Whatman filter paper moistened with 0.5 mL of a water–methanol (20:80) solution with 0.1% formic acid. A 10 cm × 10 cm template was used to establish a fixed sampling area for flat surfaces. For objects that were not flat, measurements were taken, and the surface area was calculated manually.
Samples were analyzed using high-performance liquid chromatography/tandem mass spectrometry, the gold standard for analyzing hazardous drugs from wipe samples29; the analyses were performed at the Occupational and Environmental Hygiene laboratory at the University of British Columbia. The concentration of each drug analyte was reported in nanograms per square centimeter (ng/cm2). This validated analytical method can simultaneously quantify 11 drugs per wipe,31 and the laboratory’s typical limit of detection for each of the various analytes is presented in Table 1.
Surface contamination levels were summarized using frequency distributions for each of the 3 time points, which included the proportion of samples above the limit of detection, the percent change in contamination for all wipe samples, and the percent change in contamination by each individual analyte (drug). Comparative analyses, for all drugs combined and for each individual analyte, were conducted using the Wilcoxon signed-rank test to determine whether there was a difference in contamination levels over time (2 separate pairs of sampling times: T0 vs T1 and T1 vs T2). The purpose of this analysis was to evaluate the effectiveness of the intervention before and after CSTD implementation (i.e., change from T0 to T1), as well as the sustained effect of the CSTD over time (i.e., change from T1 to T2). All analyses were performed using SPSS version 29.0 (IBM Corporation), and the level of statistical significance was set at p < 0.05.
No previous studies were known to have examined the CSTD change management process; therefore, a new online survey was developed to capture health care employees’ attitudes and experiences related to the transition to these devices. Questions were created based on existing surveys in the areas of human resources23 and change management (SampleForms software; https://www.sampleforms.com/questionnaire-form-sample.html), with input from experts in occupational health and quantitative survey design. The questions focused on overall perceptions of the implementation of CSTDs, the leadership demonstrated within individual departments and across the health authority during the transition, communications during the transition process, and training provided on the appropriate use of CSTDs. For example, one of the survey questions was “How would you rate your level of satisfaction with your department’s leadership provided throughout the entire process of transitioning to CSTDs?” Participants responded to each question on a 5-point Likert scale. Participant demographic characteristics were also captured, which included age, gender, work site, department, job title, job status (i.e., full- or part-time), and years of experience. Lastly, participants were invited to submit comments in response to open-ended questions about negative experiences during the transition process, suggestions for improvements related to the transition, and overall comments about leadership.
A study team member (T.I.E.C.) sent the survey link to managers of pharmacy and oncology units, who subsequently shared the link with workers by email. Managers also received a recruitment poster that they could print and display within their departments as an additional (optional) dissemination method. Interested participants were invited to click on a link within the email message, which took them to the electronic survey. At the survey website, they were first shown an informed consent form and told that completion and submission of the survey implied consent to participate. The survey questions followed, starting with those related to demographic characteristics. Participants who provided their email address received a $5 electronic gift card as recognition for their time.
Survey results are reported primarily through descriptive statistics, including frequencies. Missing data were not replaced, given that no inferential statistics were deployed. Group differences between the 2 job cohorts (i.e., pharmacy staff and oncology clinic personnel) were calculated using χ2 tests. All analyses were performed using SPSS software, version 29.0 (IBM Corporation), and the level of statistical significance was set at p < 0.05.
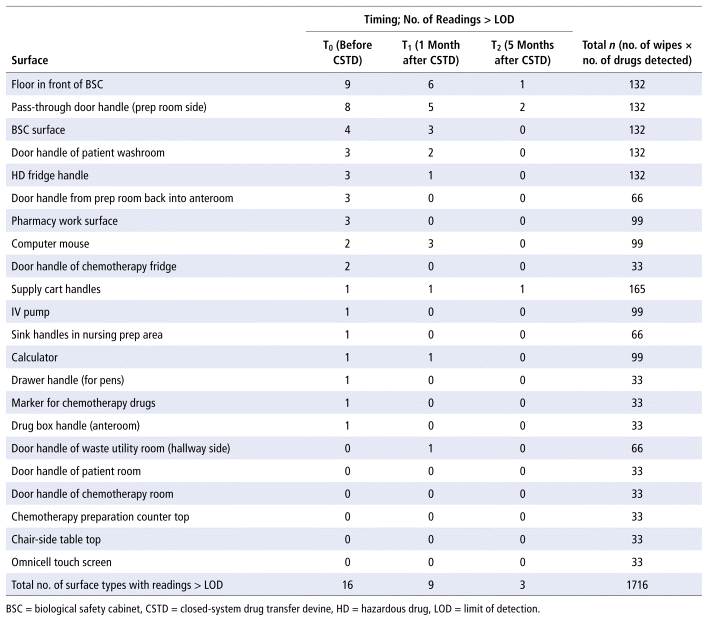
For each of the 3 time points examined, the sum of the mass of all drugs extracted from the surface wipes was 25.817 ng/cm2 for T0, 4.010 ng/cm2 for T1, and 0.068 ng/cm2 for T2. The maximum reported concentration for each drug was typically observed at T0; in fact, more analytes were above the limit of detection at T0 than at the other times (numbers of analytes above the limit of detection were 43 at T0, 23 at T1, and 4 at T2). From T0 to T1, the reduction in drug contamination level (in terms of total mass) was approximately 85%, and from T1 to T2, the decrease was roughly 98%. The decrease in surface contamination levels for both comparisons was statistically significant (p < 0.01). Figure 1 depicts the extent of decrease for selected hazardous drugs, and Table 2 provides detailed surface contamination data stratified by the various analytes. An additional table, in Appendix 1, lists the surfaces evaluated and the corresponding number of samples with contamination above the limit of detection for each of the 3 time points.
|
| ||
|
FIGURE 1 Change in contamination levels at the 3 evaluated time points for 3 commonly prepared hazardous drugs. T0 = before implementation of closed-system drug transfer devices. T1 = 1 month after implementation of the devices, T2 = 5 months after implementation. Note: The y axis has a logarithmic scale. | ||
The survey was open for 6 weeks (May 1 to June 20, 2023), and up to 3 reminder emails were sent. We estimated that 136 staff worked directly with the CSTDs or had a role in their implementation. Since participants were not required to answer every question, the sample size for each question may have varied. On average, 50 people answered the survey questions representing a response rate of approximately 35%.
Participants were primarily female (43/50), ranged in age from 26 to 45 years (31/50), and had worked at the health authority for up to 10 years (29/50). They were employed full-time as nurses (18/50 [36%]) or pharmacy technicians (21/50 [42%]) in inpatient or outpatient oncology (17/50 [34%] and 7/50 [14%], respectively) or in pharmacy (26/50, [52%]). Most of the participants (43/53 [81%]) reported working directly with CSTDs, but most (41/49 [84%]) had no previous experience with these devices before implementation at the health authority, despite the majority (29/50 [58%]) having 1 to 10 years of experience working with hazardous drugs.
For purposes of the analysis, the original 5-point Likert response scale was converted to a simpler dichotomous scheme (i.e., negative/neutral and positive outcomes). Overall, most respondents (60%) had positive perceptions of the CSTD transition process and felt that there had been a sufficient level of support throughout the transition (Table 3). Respondents were quite satisfied with leadership from their department (73%) but were less satisfied with leadership from the health authority itself (57%). Respondents had mainly positive views regarding communications related to the transition (63%) but were not as satisfied regarding the thoroughness of the communications (55%).
TABLE 3 Responses to Questions about Change Management Process
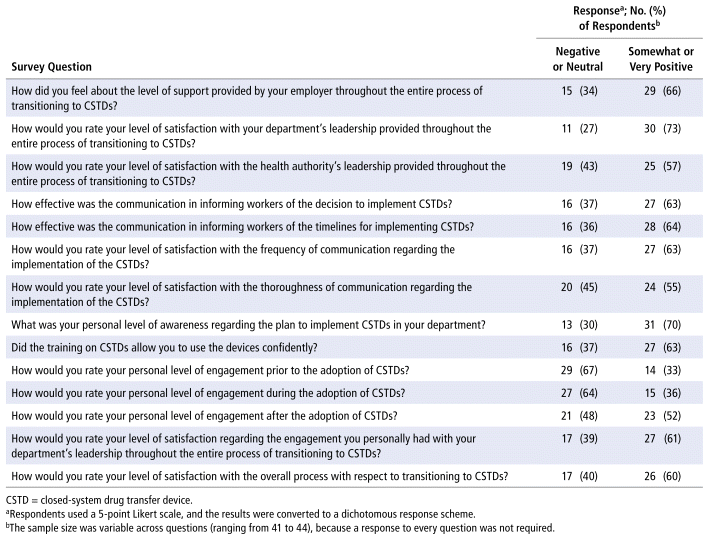
The majority of respondents rated their personal awareness of the plan to implement CSTDs as positive (70%), although there was a significant between-group difference between those working in oncology and those working in pharmacy (18/21 vs 13/23, respectively, were somewhat or very aware; χ2(df 1) 4.49, p < 0.05). Among all respondents, personal engagement was not rated highly before or during the adoption of CSTDs (33% and 35%, respectively; Table 3). Another significant between-group difference emerged with regard to whether training allowed for use of these devices with confidence: most of those working in pharmacy stated that they were somewhat or very confident, whereas fewer than half of those working in oncology felt the same (18/22 vs 9/21, respectively; χ2(df 1) 6.98, p < 0.05).
About half of the respondents answered one or more of the open-ended questions. Some felt there had not been sufficient training and/or support during CSTD implementation and suggested that greater site-based support was required. One respondent stated that “[the] go live date was before the vendor came to do an intro on the products. We had to just figure them out ourselves with non-hazardous drugs.” Another cited “poor follow-up with supplemental teaching after implementation.” Suggested improvements included more and/or different training, especially after implementation, including hands-on practice, training videos, and availability of hard copies of procedures. Others requested better communication and support: “Some type of pharmacy wide meeting to make sure more of the staff was informed.”
This study was intended to evaluate the adoption of CSTDs into practice at a Canadian health authority. Specifically, it aimed to confirm the effectiveness of the selected CSTD device in reducing surface contamination, as well as to ascertain workers’ perceptions of the change management process. Analysis of wipe samples showed that surface contamination had declined by 1 month after the introduction of CSTDs, and the decrease in contamination was maintained at 5 months after implementation. This reduction was observed with respect to the total mass of residual drug, as well as the proportion of wipe samples with contamination above the limit of detection. These findings are comparable to those of previous studies. Siderov and others14 reported that cyclophosphamide contamination was reduced to nondetectable levels following introduction of CSTDs. We had similar findings at T1 and T2 (i.e., below the limit of detection), not just for cyclophosphamide, but for all drugs examined in the current study. Similar to other studies,25,26,32 we found a statistically significant difference in surface contamination levels before and after implementation of CSTDs. Like Bartel and others,15 we found that contamination levels were reduced in both the drug preparation (pharmacy) and drug administration (oncology) departments following the introduction of CSTDs. The novelty of the current study lies in the particular CSTD model used and how and where the samples were obtained. To our knowledge, only 2 other studies have evaluated the effectiveness of the ChemoLock CSTD. One study, by Soefje and others,33 was a laboratory-controlled evaluation; the other, by Bishay and others,34 examined the effectiveness of the ChemoLock CSTD in tandem with cleaning protocols. Our study builds on this limited body of work with respect to evaluating the effectiveness of the ChemoLock CSTD in reducing surface contamination with hazardous drugs.
According to the results of our survey of health care workers, the change management process employed by the health authority appeared to be well received. Participants had positive perceptions about the leadership demonstrated and the support they received with respect to the transition process. These findings are noteworthy given the conclusion of DeJoy and others35 that there is greater compliance with use of these engineering controls when employees perceive management as having a higher commitment to safety. In addition, adherence to CSTD use, as well as to other elements of safe drug handling protocols, can improve the overall safety culture of an organization.36 However, participants also raised some concerns and suggested areas for improvement, notably, with respect to training and communication. Not surprisingly, these 2 factors were also deemed by Boiano and others37 to represent deficiencies with respect to improving nurses’ compliance with safe drug handling guidelines. We also noted significant differences in a few areas between respondents from oncology and those in pharmacy. Going forward, other health authorities may want to perform a pre-implementation assessment to ascertain whether a particular department might need more or different support and/or training during the transition to CSTDs.
Although this study had many positive findings, it also had some limitations. The scope of our project allowed us to examine only 2 time points after CSTD implementation. To confirm that these devices continue to remain effective, it would be important to assess surface contamination levels over a longer period. To our knowledge, time trend analysis of surface contamination levels has not been conducted in British Columbia. In addition, this study examined only one model of CSTD, but several types of CSTD are available on the market. To get an overall understanding of the effectiveness of CSTDs, it would be important to evaluate all commercially available CSTDs in Canada using a similar study design. Lastly, only 4 acute care facilities participated in the study, and only about 50 health care workers completed the survey; it is suggested that future similar studies increase the sample size for greater generalizability. Moreover, it would be valuable to examine surface contamination levels and the change management process at various types of facilities (e.g., long-term care, veterinary clinics).
Overall, the CSTD adopted for use was found to be effective in reducing surface contamination levels and, in turn, the risk of occupational exposure. However, drug contamination was not completely eliminated and, therefore, health care workers who handle hazardous drugs must still use precautionary measures (e.g., gloves, gowns, hand hygiene). The change management strategy employed for implementation of the CSTDs appeared to be well received. However, feedback from survey respondents indicated that more thorough training and ongoing communication are important considerations when implementing CSTDs as the standard of practice.
1 Connor TH, Lawson CC, Polovich M, McDiarmid M. Reproductive health risks associated with occupational exposures to antineoplastic drugs in health care settings. J Occup Environ Med. 2014;56(9):901–10.
Crossref PubMed PMC
2 Ratner PA, Spinelli JJ, Beking K, Lorenzi M, Chow Y, Teschke K, et al. Cancer incidence and adverse pregnancy outcome in registered nurses potentially exposed to antineoplastic drugs. BMC Nurs. 2010;9:15.
Crossref PubMed PMC
3 Power LA, Coyne JW, Hawkins B. ASHP guidelines on handling hazardous drugs. Am J Health Sys Pharm. 2018;75(24):1996–2031.
Crossref
4 Quansah R, Jaakkola JJ. Occupational exposures and adverse pregnancy outcomes among nurses: a systematic review and meta-analysis. J Womens Health (Larchmt). 2010;19(10):1851–62.
Crossref PubMed
5 McDiarmid MA, Polovich M, Power LA, Connor TH, Kienle PC. Published review of closed-system drug-transfer devices: limitations and implications. Am J Health Syst Pharm. 2018;75(24):1982–5.
Crossref PubMed
6 NIOSH list of antineoplastic and other hazardous drugs in healthcare settings, 2016. Publ no. 2016–161. National Institute for Occupational Safety and Health; 2016 [cited 2023 Mar 3]. Available from: https://www.cdc.gov/niosh/docs/2016-161/default.html
7 Hon CY, Teschke K, Chua P, Venners S, Nakashima L. Occupational exposure to antineoplastic drugs: identification of job categories potentially exposed throughout the hospital medication system. Saf Health Work. 2011;2(3):273–81.
Crossref
8 Antineoplastic agents profile. CAREX Canada; 2019 [cited 2019 Dec 12]. Available from: https://www.carexcanada.ca/profile/antineoplastic_agents/
9 Fransman W, Vermeulen R, Kromhout H. Dermal exposure to cyclophosphamide in hospitals during preparation, nursing and cleaning activities. Int Arch Occup Env Health. 2005;78(5):403–12.
Crossref
10 Connor TH. Hazardous anticancer drugs in health care: environmental exposure assessment. Ann N Y Acad Sci. 2006;1076:615–23.
Crossref
11 Hon CY, Teschke K, Chu W, Demers P, Venners S. Antineoplastic drug contamination of surfaces throughout the hospital medication system in Canadian hospitals. J Occup Environ Hyg. 2013;10(7):374–83.
Crossref PubMed
12 Hon CY, Teschke K, Shen H, Demers PAPA, Venners S. Antineoplastic drug contamination in the urine of Canadian healthcare workers. Int Arch Occup Env Health. 2015;88(7):933–41.
Crossref
13 Preventing occupational exposure to antineoplastic and other hazardous drugs in health care settings. Publ no. 2004-165. National Institute for Occupational Safety and Health; [cited 2011 Nov 30]. Available from: http://www.cdc.gov/niosh/docs/2004-165/
14 Siderov J, Kirsa S, McLauchlan R. Reducing workplace cytotoxic surface contamination using a closed-system drug transfer device. J Oncol Pharm Pract. 2010;16(1):19–25.
Crossref
15 Bartel SB, Tyler TG, Power LA. Multicenter evaluation of a new closed system drug-transfer device in reducing surface contamination by antineoplastic hazardous drugs. Am J Health Sys Pharm. 2018;75(4):199–211.
Crossref
16 Salch SA, Zamboni WC, Zamboni BA, Eckel SF. Patterns and characteristics associated with surface contamination of hazardous drugs in hospital pharmacies. Am J Health Sys Pharm. 2019;76(9):591–8.
Crossref
17 Sessink PJM, Trahan J, Coyne JW. Reduction in surface contamination with cyclophosphamide in 30 US hospital pharmacies following implementation of a closed-system drug transfer device. Hosp Pharm. 2013;48(3):204–12.
Crossref
18 Randolph SA. Hazardous drugs: handling in health care settings. Workplace Health Saf. 2018;66(5):264.
Crossref PubMed
19 Model standards for pharmacy compounding of hazardous sterile preparations. National Association of Pharmacy Regulatory Authorities; 2015 [revised 2016 Nov; cited 2023 Mar 3]. Available from: https://www.napra.ca/wp-content/uploads/2022/09/NAPRA-Mdl-Stnds-Pharmacy-Compounding-Hazardous-Sterile-Preparations-Nov-2016-Revised-b.pdf
20 Robson LS, Shannon HS, Goldenhar LM, Hale AR. Guide to evaluating the effectiveness of strategies for preventing work injuries: how to show whether a safety intervention really works. Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health; 2011 [cited 2019 Dec 11]. Available from: https://www.cdc.gov/niosh/media/pdfs/Guide-to-Evaluating-the-Effectiveness-of-Stratgies-for-Preventing-Work-Injuries-2001-119.pdf
21 CSA Z1000:14 (R2019). Occupational health and safety management [standard]. CSA Group; 2019 [cited 2019 Dec 19]. Available from: https://www.csagroup.org/store/product/CAN-CSA-Z1000-14/
22 The impact of new technology and management of change. In: Final report: mining health, safety and prevention review. Ontario Ministry of Labour, Training and Skills Development; 2015 [cited 2019 Dec 13]. Available from: https://www.labour.gov.on.ca/english/hs/pubs/miningfinal/technology.php
23 Maheshwari S, Vohra V. Identifying critical HR practices impacting employee perception and commitment during organizational change. J Organ Change Manag. 2015;28(5):872–94.
Crossref
24 Gurusamy KS, Best LMJ, Tanguay C, Lennan E, Korva M, Bussières JF. Closed-system drug-transfer devices plus safe handling of hazardous drugs versus safe handling alone for reducing exposure to infusional hazardous drugs in healthcare staff. Cochrane Database Syst Rev. 2018;3(3):CD012860.
PubMed PMC
25 Simon N, Vasseur M, Pinturaud M, Soichot M, Richeval C, Humbert L, et al. Effectiveness of a closed-system transfer device in reducing surface contamination in a new antineoplastic drug-compounding unit: a prospective, controlled, parallel study. PLoS One. 2016;11(7):e0159052.
Crossref PubMed PMC
26 Sripakdee W, Subongkot S, Wattanawong W, Onlaor S. Evaluation of chemotherapy contamination following the implementation of closed system transfer devices. Appl Environ Res. 2015;37(1):83–91.
Crossref
27 Guillemette A, Langlois H, Voisine M, Merger D, Therrien R, Mercier G, et al. Impact and appreciation of two methods aiming at reducing hazardous drug environmental contamination: the centralization of the priming of IV tubing in the pharmacy and use of a closed-system transfer device. J Oncol Pharm Pract. 2014;20(6):426–32.
Crossref PubMed
28 Yoshida J, Tei G, Mochizuki C, Masu Y, Koda S, Kumagai S, et al. Use of a closed system device to reduce occupational contamination and exposure to antineoplastic drugs in the hospital work environment. Ann Occup Hyg. 2009;53(2):153–60.
PubMed
29 Connor TH, Zock MD, Snow AH. Surface wipe sampling for antineoplastic (chemotherapy) and other hazardous drug residue in healthcare settings: methodology and recommendations. J Occup Environ Hyg. 2016;13(9):658–67.
Crossref PubMed PMC
30 Jeronimo M, Colombo M, Astrakianakis G, Hon CY. A surface wipe sampling and LC-MS/MS method for the simultaneous detection of six antineoplastic drugs commonly handled by healthcare workers. Anal Bioanal Chem. 2015;407(23):7083–92.
Crossref PubMed
31 Colombo M, Jeronimo M, Astrakianakis G, Apte C, Hon CY. Wipe sampling method and evaluation of environmental variables for assessing surface contamination of 10 antineoplastic drugs by liquid chromatography/tandem mass spectrometry. Ann Work Expo Health. 2017;61(8):1003–14.
Crossref PubMed
32 Sessink PJM, Connor TH, Jorgenson JA, Tyler TG. Reduction in surface contamination with antineoplastic drugs in 22 hospital pharmacies in the US following implementation of a closed-system drug transfer device. J Oncol Pharm Pract. 2011;17(1):39–48.
Crossref
33 Soefje S, Rickabaugh K, Rajkumar R, Wall KP. Evaluation of closed-system transfer devices in reducing potential risk for surface contamination following simulated hazardous-drug preparation and compounding. Int J Pharm Compd. 2022;26(1):72–9.
PubMed
34 Bishay S, Michalowska-Suterska M, Edling A, Battle J. Evaluation of the hazardous drug surface contamination in pharmacy compounding and administration clinical setting after adoption of standardized cleaning workflow and a closed system transfer device. Pharm Technol Hosp Pharm. 2022;7(1):20220004.
Crossref
35 DeJoy DM, Smith TD, Woldu H, Dyal MA, Steege AL, Boiano JM. Effects of organizational safety practices and perceived safety climate on PPE usage, engineering controls, and adverse events involving liquid antineoplastic drugs among nurses. J Occup Environ Hyg. 2017; 14(7):485–93.
Crossref PubMed
36 McDiarmid MA, Condon M. Organizational safety culture/climate and worker compliance with hazardous drug guidelines: lessons from the blood-borne pathogen experience. J Occup Environ Med. 2005;47(7): 740–9.
Crossref PubMed
37 Boiano JM, Steege AL, Sweeney MH. Adherence to safe handling guidelines by healthcare workers who administer antineoplastic drugs. J Occup Environ Hyg. 2014;11(11):728–40.
Crossref PubMed PMC
Address correspondence to: Dr Chun-Yip Hon, Toronto Metropolitan University, 350 Victoria Street, DCC304, Toronto ON M5B 2K3, email: cyhon@torontomu.ca
Competing interests: For activities not directly related to the study reported here, Jackie Ellis received a travel honorarium from the Pharmacy Technician Society of British Columbia, and Tina I En Chiang received a travel honorarium from WorkSafeBC Innovation at Work. No other competing interests were declared.
Funding: This study was funded by WorkSafeBC Innovation at Work through a grant administered by the Fraser Health Authority.
Acknowledgements: The authors wish to thank the participating sites, as well as all the survey respondents. They are also indebted to Samar Hejazi, formerly of the Fraser Health Authority, who provided assistance with the nonparametric tests.
Submitted: July 3, 2024
Accepted: February 24, 2025
Published: July 9, 2025
APPENDIX 1 Summary of surfaces evaluated and corresponding number of samples above detection for all 3 time points.
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 3, 2025