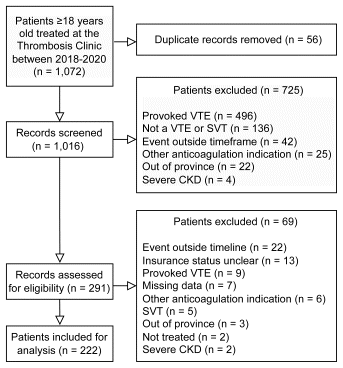
FIGURE 1 Patient selection flowchart. CKD = chronic kidney disease, SVT = superficial vein thrombosis, VTE = venous thromboembolism.
Alex Robin, Daniel Liwski, Sudeep Shivakumar, and Allen TranTo cite: Robin A, Liwski D, Shivakumar S, Tran A. Impact of provincial pharmacare coverage for direct oral anticoagulants on long-term anticoagulation for unprovoked venous thromboembolism. Can J Hosp Pharm. 2025;78(4):e3688. doi: 10.4212/cjhp.3688
ABSTRACT
Background
Unprovoked venous thromboembolism (VTE) has a high risk of recurrence, typically warranting indefinite anticoagulation. Direct-acting oral anticoagulants (DOACs) have several advantages over warfarin and are thus recommended preferentially for long-term use. Until January 2024 in Nova Scotia, DOAC use beyond 6 months was often cost-prohibitive due to limited provincial funding.
Objective
To evaluate the impact of drug coverage on duration of anticoagulation for unprovoked VTE in Nova Scotia.
Methods
A retrospective chart review was completed for patients with unprovoked VTE seen in the Halifax Thrombosis Clinic between 2018 and 2020. Patients were grouped by anticoagulant class, type of insurance (private, provincial, or neither), and anticoagulation continuation or noncontinuation beyond 6 months. The primary outcome was the difference in anticoagulation use at 6 months according to type of insurance based on χ2 testing.
Results
The chart review identified 1016 patients seen during the study period, of whom 222 were included in the analysis. No significant difference in treatment duration was found among patients with private insurance, those covered under provincial pharmacare, and those with no insurance (mean duration 14.47, 12.39, and 13.89 months, respectively; p = 0.25). Scores for the Charlson comorbidity index and patient age did not significantly affect treatment duration. Patients with private insurance were more likely to receive a prescription for DOACs (p = 0.015 relative to provincial pharmacare, p < 0.001 relative to no insurance).
Conclusions
This study showed no statistically significant difference among types of insurance in terms of duration of anticoagulation after unprovoked VTE. However, patients with private insurance were more likely to use DOACs than warfarin. Since this study was completed, Nova Scotia Pharmacare now covers DOACs, but coverage remains limited in many other provinces. The results of this study may serve as evidence to lobby for extended DOAC funding in other provinces as a way to enhance care.
Keywords: venous thromboembolism, pharmacare, anticoagulation, health promotion, accessibility
RÉSUMÉ
Contexte
Le risque de récidive de la thromboembolie veineuse (TEV) non provoquée est élevé, ce qui justifie généralement un traitement anticoagulant indéfini. Les anticoagulants oraux directs (AOD) ont plusieurs avantages par rapport à la warfarine et sont donc recommandés de préférence pour un usage à long terme. Jusqu’en janvier 2024, en Nouvelle-Écosse, le coût de l’utilisation des AOD au-delà de 6 mois était souvent prohibitif en raison du financement provincial limité.
Objectif
Évaluer les conséquences de la couverture médicaments sur la durée du traitement anticoagulant pour la TEV non provoquée en Nouvelle-Écosse.
Méthodologie
Une revue rétrospective des dossiers des patients atteints d’une TEV non provoquée dans l’unité de thrombose de Halifax a été réalisée entre 2018 et 2020. Les patients ont été regroupés par classe d’anticoagulant administré, type d’assurance (privée, provinciale ou aucune) et continuation ou non du traitement après 6 mois. Le résultat principal a été la différence en matière d’utilisation du traitement anticoagulant à 6 mois en fonction du type d’assurance, basée sur un test du χ2.
Résultats
La revue des dossiers a permis d’identifier 1016 patients vus pendant la période de l’étude, dont 222 ont été inclus dans l’analyse. Aucune différence significative dans la durée du traitement n’a été observée parmi les patients ayant une assurance privée, ceux couverts par le programme d’assurance provincial et ceux sans assurance (moyenne de 14,47, 12,39 et 13,89 mois, respectivement; p = 0,25). Les scores de l’indice de comorbidité de Charlson et l’âge des patients n’ont pas démontré d’effet significatif sur la durée du traitement. Les patients ayant une assurance privée étaient plus susceptibles de recevoir une ordonnance pour des AOD (p = 0,015 par rapport à ceux couverts par l’assurance provinciale; p < 0,001 par rapport aux patients sans assurance).
Conclusions
Cette étude n’a montré aucune différence statistiquement significative entre les types d’assurance en ce qui concerne la durée du traitement anticoagulant pour une TEV non provoquée. Cependant, les patients ayant une assurance privée étaient plus susceptibles d’utiliser des AOD plutôt que de la warfarine. Depuis la réalisation de cette étude, le programme d’assurance de Nouvelle-Écosse couvre désormais les AOD, mais leur couverture reste limitée dans de nombreuses provinces. Les résultats de cette étude pourraient servir de preuve afin de faire pression pour élargir le financement des AOD dans d’autres provinces et améliorer les soins.
Mots-clés: thromboembolie veineuse, assurance médicaments, anticoagulation, promotion de la santé, accessibilité
Venous thromboembolism (VTE), including deep vein thrombosis and pulmonary embolism, is classified as either provoked or unprovoked, depending on the presence or absence of risk factors. This classification has a deciding clinical impact on how to manage long-term anticoagulation. Unprovoked VTE has a high risk of recurrence, exceeding 5% annually, which has prompted national guidelines to recommend indefinite anticoagulation, with periodic reassessment to weigh bleeding risk and patient preferences.1
Direct-acting oral anticoagulants (DOACs) are preferred over vitamin K antagonists and alternative, non-oral anticoagulants for long-term anticoagulation.2 Compared to warfarin, DOACs offer several advantages, including fixed dosing without the need for routine coagulation monitoring, a lower risk of major bleeding, fewer drug interactions, and no known food interactions, except rivaroxaban, which should be taken with food.
Until January 2024, the Nova Scotia Pharmacare Programs3 granted only 6 months of coverage for DOACs (apixaban, dabigatran, edoxaban, and rivaroxaban) for the treatment of patients with VTE, unless an appeal was made. This limited coverage posed an important challenge, as indefinite therapy is typically recommended for unprovoked VTE. Furthermore, lack of financial means is often a barrier to receiving long-term anticoagulation. For example, based on Nova Scotia’s general formulary prices in October 2025, a 6-month course of therapy costs $12.14 for warfarin and $147.02 for apixaban. After 6 months, patients who had been receiving a DOAC often decided to switch to warfarin or stop anticoagulation altogether if they could not personally afford DOACs or could not tolerate warfarin’s inconvenient usage profile, which increased their risk of a recurrent event. Patients in many provinces still face this issue.
Unfortunately, lack of access to prescription drug coverage leads to disparities in care. These systemic barriers disproportionately affect marginalized groups, who are less likely to have private coverage and thus have less access to essential medications when gaps exist.4 Without policy changes to address these gaps, at-risk patients will continue to experience preventable health complications due to inadequate coverage for medications, including DOACs for long-term anticoagulation. This situation highlights the need for targeted improvements and formed the rationale for the current study.
We aimed to understand how Nova Scotia’s provincial coverage of DOACs affected the treatment decisions of patients who experienced unprovoked VTE. We anticipated that our findings might serve as evidence to advocate for increased duration of funding for treatment of unprovoked VTE, ultimately improving patient care.
This single-centre retrospective cohort study involved patients with unprovoked VTE who were seen between January 2018 and January 2020 in the Halifax Thrombosis Clinic, identified using a hospital database. Ethics approval was obtained from the Research Ethics Board of the Nova Scotia Health Authority.
All patients seen in the clinic during the study time frame were eligible if they had experienced a VTE. For patients with multiple VTEs, only the first event was considered. VTE was defined as any episode of deep vein thrombosis at any site and/or pulmonary embolism. Cases of superficial vein thrombosis were excluded. Patients with provoked VTE, including cancer-associated VTE, and those receiving long-term anticoagulation for other indications (e.g., atrial fibrillation, mechanical valve) were excluded, as their access to anticoagulation funding is different. Patients with chronic kidney disease (creatinine clearance < 30 mL/min), for whom DOACs are generally contraindicated, were also excluded. The VTE type was determined from the clinic note documentation by the thrombosis clinic physician.
Data collection involved a manual review of electronic medical records in the hospital database and medication dispensing information in the provincial pharmacy database. Data were collected to determine the demographic characteristics and comorbidities needed to calculate the Charlson comorbidity index (CCI), a method of quantifying comorbidity burden and predicting mortality5; more specifically, this metric predicts the 10-year survival rate in patients with multiple comorbidities. Additionally, details about the VTE and the patient’s anticoagulant treatment were gathered. The type of insurance was determined from clinic notes.
Patients were excluded if their insurance status (i.e., whether or not they had insurance) could not be clearly identified. A patient with both private insurance and pharmacare coverage was categorized as having private insurance. Nova Scotia’s pharmacare plan is available to eligible residents, including adults older than 65 years of age, low-income families, and patients with cancer, with eligibility based on age, income, and medical needs. Patients covered by federal plans, such as certain First Nations and Inuit people and Canadian Armed Forces personnel, were included in the private drug coverage group, as DOACs are typically covered without restriction for these groups, unlike Nova Scotia’s pharmacare plan. The primary anticoagulant was defined as the anticoagulant taken for the longest duration within the study time frame, typically based on dispensing data, unless specified in clinic notes.
The treatment duration was defined as the total dispensing time within the 2-year study period, unless clinic notes specified that the patient was to stop before consuming the entire amount of dispensed medication. The available dispensing data did not provide the number of “days supplied” for warfarin. Therefore, the duration of warfarin treatment was calculated on the basis of a presumed 5-mg daily average dose.6
The primary outcome was tested with χ2 testing to determine if there was a statistically significant difference in the proportions of patients with unprovoked VTE who were still receiving anticoagulation at 6 months after the initial event, in relation to the type of insurance, specifically private insurance relative to pharmacare or no insurance.
For the secondary analysis, the analysis was repeated at 12 months after the VTE. The mean duration of anticoagulation was also compared in the subgroups using analysis of variance. Potential confounding factors, notably age and comorbidity status (CCI score), were investigated using binomial logistic regression to help isolate any variation in anticoagulation duration due to these factors. Potential variance explained was assessed using the Nagelkerke R2, with comorbidity quantified using the CCI score. Lastly, the proportions of patients receiving DOACs were compared across insurance types using the χ2 test of independence.
A p value of 0.05 or below was considered to be statistically significant. The statistical analyses were done with SPSS Statistics software (IBM Corporation). Descriptive statistics (e.g., means, percentages) were calculated for all study variables.
The initial chart review identified a cohort of 1016 patients, of whom 222 patients were deemed eligible for further analysis (Figure 1). Common reasons for exclusion included VTE outside the designated time frame, pre-existing indications for anticoagulation, and apparent provoking factors for the VTE.
|
| ||
|
FIGURE 1 Patient selection flowchart. CKD = chronic kidney disease, SVT = superficial vein thrombosis, VTE = venous thromboembolism. | ||
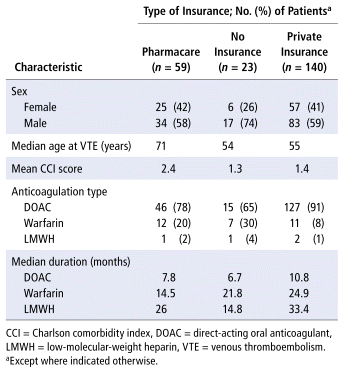
The mean age of patients was 58.4 years, with a median age of 59 years (Table 1), and approximately 40% of the cohort were women. In terms of drug coverage, 23 patients had no insurance, 59 had pharmacare only, and 140 had private insurance. The mean CCI was 1.7 points, out of a maximum of 37 points.5 Notably, the mean CCI was 1.4 points for patients with private insurance and 2.4 points for those with pharmacare. For context, some studies consider a CCI of 2 or greater to indicate significant comorbidity.7,8
TABLE 1 Characteristics of Included Patients
Regarding primary anticoagulation, 188 patients received primarily DOACs, 30 patients received warfarin, and 4 received dalteparin (Figure 2). Over the 2-year time frame, an average of 12.2 months’ worth of medication was dispensed for patients taking DOACs, whereas the average was 22.3 months’ worth for those taking warfarin (based on a presumed 5-mg daily dose, on average). For 32 patients who ever received warfarin, the amount of medication dispensed within the study period was sufficient for more than 2 years of anticoagulation, with the patient who had the most medication dispensed picking up 55.8 months’ worth of warfarin (based on the presumed 5-mg daily dose).
|
| ||
|
FIGURE 2 Type of anticoagulant selected, by type of insurance. AC = anticoagulant, DOAC = direct-acting oral anticoagulant. | ||
There was no significant association between medical insurance status and anticoagulation duration exceeding 6 months in patients with private insurance relative to those without (patients with pharmacare and those with no insurance) (p = 0.24). This lack of significance persisted with the subgroup analysis, comparing individuals with no insurance, those with pharmacare coverage, and those with private insurance (p = 0.51). Additionally, no significant relationship was observed between type of insurance and anticoagulation at the 12-month mark.
As seen in Figure 3, the mean duration of treatment for patients with private insurance, provincial pharmacare, and no insurance did not differ significantly (14.47, 12.39, and 13.89 months, respectively; p = 0.25). Treatment duration among patients who primarily received DOACs was also not statistically different among the 3 insurance coverage types.
|
| ||
|
FIGURE 3 Mean duration of anticoagulant treatment, by type of insurance. | ||
Patients with private insurance received DOACs significantly more frequently than patients with pharmacare coverage (91% vs. 78%, p = 0.015) and those without insurance (91% vs. 65%, p < 0.001).
Age and comorbidity status did not significantly confound the results, as indicated by the Nagelkerke R2 values. Specifically, this analysis suggested that age accounted for 2.5% of the variance (p = 0.55), whereas the CCI explained 1.9% of the variance (p = 0.84).
In this study of patients with unprovoked VTE, there was no statistically significant difference in anticoagulation use beyond 6 months according to type of insurance. As such, it is difficult to determine with certainty if anticoagulation duration is linked to insurance status. However, patients with private insurance were more likely than those with pharmacare coverage to have DOACs prescribed. More specifically, 91% of patients with private insurance were primarily using DOACs, compared to 78% of patients with pharmacare coverage and 65% of patients without insurance (Figure 2).
Current guidance from the American Society of Hematology1 and the CHEST guidelines9 recommend long-term anticoagulation for patients with unprovoked VTE, with DOACs being recommended as first-line treatment. As such, patients without private insurance may be receiving a different standard of care than those with private insurance. Our study supports the hypothesis that provincial reimbursement criteria limit anticoagulant selection for patients with unprovoked VTE.
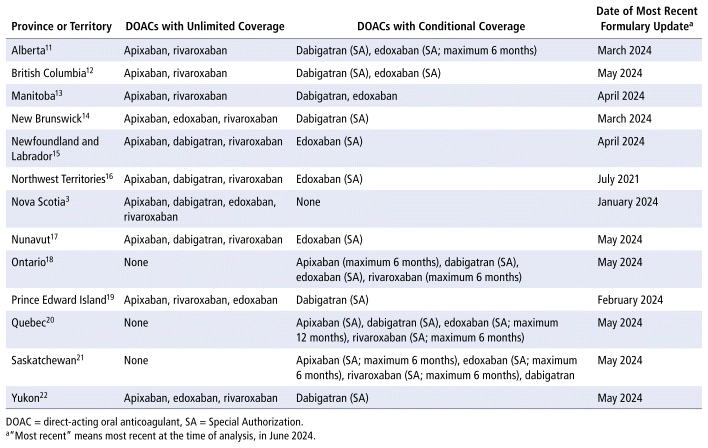
Fortunately for Nova Scotians, DOACs (apixaban, rivaroxaban, dabigatran, and edoxaban) were universally added to the Nova Scotia Pharmacare formulary during this study’s analysis phase, in January 2024. Still, there is significant variation in reimbursement mechanisms across the country, leading to large variation in DOAC utilization across Canada (Figure 4).10 According to a review of formularies in June 2024, almost half of Canadian provinces still impose substantial limitations on DOAC coverage for VTE (Table 2).11–22 Many of these carry the same 6-month limitation or require special authorization for extended coverage.
|
| ||
|
FIGURE 4 Restrictions, as of June 2024, on availability of direct oral anticoagulants (DOACs) for long-term treatment (more than 6 months) of venous thromboembolism across Canada, by province or territory, where green indicates jurisdictions with no DOAC limits or limits on a single DOAC, yellow indicates jurisdictions with limits on 2 DOACs, and red indicates jurisdictions with limitations on 3 or 4 DOACs. Provinces with a single DOAC limitation were included in the “green zone” because the single limitation was typically for dabigatran or edoxaban, both of which are used less often than apixaban and rivaroxaban. | ||
TABLE 2 Provincial Limitations on DOACs for Venous Thromboembolism, as of June 2024
Our primary outcome was not statistically significant. The study may have been underpowered due to the small sample size, which may explain the lack of difference between groups. We excluded more patients than anticipated because of possible provoking factors, missing insurance history, and evaluation of patients whose VTE preceded our study time frame. Our hypothesized confounding factors, age and comorbidity status, did not seem to significantly contribute to the results, with small confounders of variance and nonsignificant p values.
Quantifying some of the other effects of pharmacare limitations was challenging. Many patients were lost to follow-up, which made it impossible to determine the reasons for stopping anticoagulation. For example, insurance changes were rarely documented in follow-up notes, so it was difficult to assess whether anticoagulant switches were driven by coverage changes. Similarly, the shared decision-making surrounding anticoagulant duration in the clinic is often complex and multifactorial. For this reason, obtaining concise information to properly delineate the financial hardship caused by lack of coverage was not feasible.
A more exact process to quantify medication duration would have yielded more dependable results. For instance, patients may have had a full course of medication dispensed without completing the total course. In particular, the inexpensive nature of warfarin often leads pharmacies to dispense it in bulk for titration convenience. For example, one patient’s pharmacy dispensed 55.8 months’ worth of warfarin (based on a presumed 5-mg dose) over the 2-year period. Given that the majority of patients taking warfarin did not have private insurance, this patient’s data likely falsely inflated the duration of anticoagulation within this group. The dispensing information available was not consistent enough to reliably calculate individual warfarin dosing. We therefore estimated that patients received a 5-mg daily dose of warfarin, based on Thrombosis Canada clinical guides6 and a prior retrospective analysis.23
Furthermore, warfarin doses commonly fluctuate over time for individuals, which may have affected our results. Although for each patient we considered the anticoagulant with the longest duration in the analysis, this approach may not have fully addressed the complexity of patients’ treatment transitions.
These findings suggest that future “exceptional status” medication approvals should align with current treatment guidelines to ensure equity for patients without private insurance who are receiving medications. Moving forward, there are promising avenues for further exploration related to this study. Evaluating prescribing patterns before and after Nova Scotia’s DOAC formulary updates could reveal substantial shifts in adherence to standard-of-care practices. Finally, replicating this study in provinces without comprehensive DOAC coverage could shed light on health care disparities, aiding in the pursuit of equitable access to optimal anticoagulation therapy nationwide.
1 Ortel TL, Neumann I, Ageno W, Beyth R, Clark NP, Cuker A, et al. American Society of Hematology 2020 guidelines for management of venous thromboembolism: treatment of deep vein thrombosis and pulmonary embolism. Blood Adv. 2020;4(19):4693–738.
Crossref PubMed PMC
2 Fang MC, Reynolds K, Fan D, Prasad PA, Sung SH, Portugal C, et al. Clinical outcomes of direct oral anticoagulants vs warfarin for extended treatment of venous thromboembolism. JAMA Netw Open. 2023;6(8):e2328033.
Crossref PubMed PMC
3 Nova Scotia formulary. Nova Scotia Department of Health, Nova Scotia Pharmacare; 2024 Jan [cited 2024 Jun]. Available from: https://novascotia.ca/dhw/pharmacare/documents/formulary.pdf
4 Yang FJ, Gupta S. Exploring gaps in prescription drug insurance coverage among men and women in Canada using an intersectional lens. Statistics Canada; 2024 Jan 10 [cited 2025 Feb 5]. Available from: https://www150.statcan.gc.ca/n1/pub/75-006-x/2024001/article/00001-eng.htm
5 Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–83.
Crossref PubMed
6 Warfarin. Thrombosis Canada; 2025 May [cited 2025 Nov 12]. Available from: https://thrombosiscanada.ca/clinical_guides/pdfs/WARFARIN_43.pdf
7 Ofori-Asenso R, Zomer E, Chin KL, Si S, Markey P, Tacey M, et al. Effect of comorbidity assessed by the Charlson Comorbidity Index on the length of stay, costs and mortality among older adults hospitalised for acute stroke. Int J Environ Res Public Health. 2018;15(11):2532.
Crossref PubMed PMC
8 Naeser Y, Mikiver R, Ingvar C, Lambe M, Ullenhag GJ. Survival in patients diagnosed with melanoma in situ compared to the general population. A Swedish population-based matched cohort study. eClinicalMedicine. 2023;65:102284.
Crossref PubMed PMC
9 Stevens SM, Woller SC, Kreuziger LB, Bounameaux H, Doerschug K, Geersing GJ, et al. Antithrombotic therapy for VTE disease. Chest. 2021;160(6):e545–e608.
Crossref PubMed
10 Gao L, Tadrous M, Knowles S, Mamdani M, Paterson JM, Juurlink D, et al. Prior authorization and Canadian public utilization of direct-acting oral anticoagulants. Healthc Policy. 2017;13(2):68–78.
PubMed PMC
11 Formulary: continuing care type A pharmacy – Calgary Zone. Alberta Health Services; updated 2024 Mar [cited 2024 Jun]. Available from: https://www.albertahealthservices.ca/info/page4071.aspx
12 BC PharmaCare formulary search. BC PharmaCare; updated 2024 May [cited 2024 Jun]. Available from: https://pharmacareformularysearch.gov.bc.ca/
13 Drug benefits & interchangeability formulary. Manitoba Health; updated 2024 Apr [cited 2024 Jun]. Available from: https://www.gov.mb.ca/health/mdbif/
14 New Brunswick drug plans formulary. New Brunswick Health; updated 2024 Mar [cited 2024 Jun]. Available from: https://www2.gnb.ca/content/dam/gnb/Departments/h-s/pdf/en/NBDrugPlan/NewBrunswickDrugPlansFormulary.pdf
15 The Newfoundland and Labrador interchangeable drug products formulary. Volume 90. Newfoundland and Labrador, Health and Community Services; updated 2024 Apr [cited 2024 Jun]. Available from: https://www.gov.nl.ca/hcs/files/nlpdp-formularyvol90.pdf
16 Northwest Territories health centre formulary. Government of Northwest Territories; updated 2021 Jul [cited 2024 Jun]. Available from: https://www.hss.gov.nt.ca/professionals/sites/professionals/files/resources/nwt-health-centre-formulary.pdf
17 Government of Nunavut drug formulary. Government of Nunavut, Department of Health; updated 2024 May [cited 2024 Jun]. Available from: https://www.gov.nu.ca/sites/default/files/publications/2024-01/gn_drug_formulary_binder_1_final_dec_2021.pdf
18 Ontario Drug Benefit formulary/comparative drug index (CDI). Government of Ontario; updated 2024 May [cited 2024 Jun]. Available from: https://www.ontario.ca/document/ontario-drug-benefit-odb-formulary-comparative-drug-index-cdi-and-monthly-formulary-0https://www.formulary.health.gov.on.ca/formulary/
19 P.E.I. Pharmacare formulary search. Province of Prince Edward Island, Minister of Health and Wellness; updated 2024 Feb [cited 2024 Jun]. Available from: https://www.princeedwardisland.ca/en/feature/pei-pharmacare-formulary-search/#/service/DrugFormulary/DrugFormularySearch
20 List of medications, May 23, 2024. Gouvernement du Québec, Régie de l’assurance maladie; updated 2024 May [cited 2024 Jun]. Available from: https://www.ramq.gouv.qc.ca/en/media/16796
21 Saskatchewan online formulary database. Government of Saskatchewan, Saskatchewan Drug Plan; updated 2024 May [cited 2024 Jun]. Available from: https://formulary.drugplan.ehealthsask.ca/SearchFormulary
22 Yukon drug test formulary system. Government of Yukon; updated 2024 May [cited 2024 Jun]. Available from: https://ihs.gov.yk.ca/drugs/f?p=161:9000
23 Garcia D, Regan S, Crowther M, Hughes RA, Hylek EM. Warfarin maintenance dosing patterns in clinical practice. Chest. 2005; 127(6):2049–56.
Crossref PubMed
Address correspondence to: Dr Alex Robin, Department of Medicine, Queen Elizabeth II Health Sciences Centre, 1796 Summer Street, Halifax NS B3H 3A6, email: alex.robin@dal.ca
Competing interests: None declared.
Funding: None received.
Submitted: July 22, 2024
Accepted: August 27, 2025
Published: December 10, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 4, 2025