Vincent Leclerc and Nathalie ChâteauvertTo cite: Leclerc V, Châteauvert N. Risk of cytomegalovirus infection after 3 or 6 months of valganciclovir prophylaxis among donor-seropositive, recipient-seronegative heart transplant recipients. Can J Hosp Pharm. 2025;78(2):e3701. doi: 10.4212/cjhp.3701
ABSTRACT
Background
Cytomegalovirus (CMV) disease is frequent following heart transplant, especially among patients with donor-seropositive, recipient-seronegative (D+/R–) CMV status. Valganciclovir prophylaxis for 3 to 6 months is recommended, but few data support the use of 6 rather than 3 months of prophylaxis for patients who have undergone heart transplant.
Objective
To compare the risk of CMV infection and a first occurrence of CMV disease after heart transplant among CMV D+/R− patients who received 3 or 6 months of prophylaxis in the first year after transplant.
Methods
This retrospective analysis included every D+/R− heart transplant recipient at the study centre between October 2015 and October 2022. Patients had to have a minimum of 1 year of follow-up for inclusion.
Results
Thirty-five patients met the inclusion criteria. The duration of valganciclovir prophylaxis (determined by the medical team) was 3 months for 22 patients and 6 months for 13 patients. CMV infection occurred in 68.2% (15/22) and 30.8% (4/13) of the 3-month and 6-month groups, respectively (risk difference 37.4%, 95% confidence interval [CI] 1.75 to 65.8, p = 0.04). CMV disease occurred in 54.5% (12/22) of patients who received 3 months of prophylaxis and 23.1% (3/13) of those who received 6 months of prophylaxis (risk difference 31.4%, 95% CI −3.7 to 59.4, p = 0.09). Most CMV infections and diseases occurred in the 3 months after the end of prophylaxis.
Conclusions
After heart transplant, CMV D+/R− patients who received 3 months of prophylaxis had a higher risk of CMV infection and a non-statistically significant higher risk of CMV disease relative to those who received 6 months of prophylaxis.
Keywords: cytomegalovirus, heart transplant recipients, prophylaxis, valganciclovir
RÉSUMÉ
Contexte
La maladie à cytomégalovirus (CMV) est fréquente après une transplantation cardiaque, en particulier chez les patients ayant un statut CMV donneur-séropositif, receveur-séro-négatif (D+/R–). Une prophylaxie à base de valganciclovir pendant 3 à 6 mois est recommandée, mais peu de données soutiennent l’utilisation de 6 mois de prophylaxie plutôt que 3 mois chez les patients ayant subi une transplantation cardiaque.
Objectif
Comparer le risque d’infection à CMV et la survenue d’une première maladie à CMV après une transplantation cardiaque chez les patients CMV D+/R− ayant reçu 3 ou 6 mois de prophylaxie au cours de la première année suivant la transplantation.
Méthodologie
Cette analyse rétrospective comprenait tous les bénéficiaires d’une de transplantation cardiaque D+/R− au centre d’étude entre octobre 2015 et octobre 2022. Le suivi des patients devait être d’au moins un an pour que les patients soient inclus.
Résultats
Trente-cinq patients répondaient aux critères d’inclusion. La durée de la prophylaxie par valganciclovir était de 3 mois pour 22 patients, et de 6 mois pour 13 patients, selon l’équipe médicale. Une infection à CMV est survenue dans 68,2 % (15/22) et 30,8 % (4/13) des groupes de 3 mois et de 6 mois, respectivement (différence de risque de 37,4 %, intervalle de confiance à 95 % [IC] de 1,75 à 65,8, p = 0,04). La maladie à CMV est survenue chez 54,5 % (12/22) des patients ayant reçu 3 mois de prophylaxie et 23,1 % (3/13) de ceux ayant reçu 6 mois (différence de risque de 31,4 %, IC à 95 % de −3,7 à 59,4, p = 0,09). La plupart des infections et des maladies sont survenues dans les 3 mois suivant la fin de la prophylaxie.
Conclusions
Après une transplantation cardiaque, les patients CMV D+/R− ayant reçu 3 mois de prophylaxie présentent un risque plus élevé d’infection à CMV et un risque plus élevé non statistiquement significatif de maladie à CMV par rapport à ceux ayant reçu 6 mois de prophylaxie.
Mots-clés: cytomegalovirus, transplantés cardiaques, prophylaxie, valganciclovir
Cytomegalovirus (CMV), a member of the herpesvirus family, represents one of the most common opportunistic infections affecting solid organ transplant recipients.1,2 The infection rate depends on the type of organ transplant, the degree of immunosuppression, and the patient’s CMV serostatus.2 Patients with donor-seropositive, recipient-seronegative (D+/R–) CMV status constitute the highest-risk population.2 Although reactivation of the virus or primo-infection can be asymptomatic in the general population, CMV can cause important disease in immunocompromised patients.3 Higher rates of graft failure, cardiac allograft vasculopathy, rejection, and post-transplant lymphoproliferative disease have been seen in heart transplant recipients (HTRs) who experienced CMV disease.3,4 Universal prophylaxis remains the preferred strategy for high-risk D+/R− HTRs.3,5 Canadian, US, and international transplant societies recommend antiviral prophylaxis for 3 to 6 months for D+/R− HTRs, even though the recommendation for longer-duration prophylaxis is based on studies involving only D+/R− kidney and lung transplant recipients.3–7 Recently, however, a single-centre study of 58 D+/R− HTRs who received either 3 or 6 months of valganciclovir prophylaxis showed no difference in terms of the incidence of probable or proven CMV disease.8
Our centre’s current protocol recommends 6 months of valganciclovir prophylaxis for D+/R− HTRs. However, not every health insurance plan offers the same prescription drug coverage. Most D+/R− HTRs receive only 3 months of prophylaxis because of restrictions in their health insurance prescription drug plans.
The objective of this study was to describe the incidence rate of CMV infection and of a first CMV disease among D+/R− HTRs, according to the duration of prophylaxis received (3 or 6 months) in the first year after transplant.
We conducted a single-centre retrospective study involving all new HTRs with D+/R− CMV serostatus between October 1, 2015, and October 31, 2022, who had a minimum of 1 year of follow-up. Data were extracted from the database of electronic medical records. Patient characteristics, including demographic characteristics, comorbidities, and concomitant immunosuppression, were collected. Permission to use the electronic medical records database was granted by the professional service director; informed consent was not required.
According to our centre’s immunosuppression protocol, basiliximab is used for induction in most patients, with antithymocyte globulin used in selected patients. Maintenance immunosuppressive therapy consists of tacrolimus, mofetil mycophenolate or mycophenolic acid, and prednisone. Target 12-hour trough concentrations for tacrolimus follow international recommendations.9 The target dose for mofetil mycophenolate is 1 g twice a day. Prednisone is started at 0.5 mg/kg daily and is gradually weaned over the first year after transplant. Valganciclovir is given orally at a dose of 900 mg daily for prophylaxis starting in the first 7 days after transplant; alternatively, if the oral route is unavailable, IV ganciclovir is given at a dose of 5 mg/kg every 24 hours. For both antiviral medications, the dosage is adjusted for renal function according to the manufacturers’ recommendations. Antiviral prophylaxis is restarted if acute rejection is treated with antithymocyte globulin or high-dose IV corticosteroids. Our centre does not use CMV immunoglobulin.
CMV infection is defined as evidence of CMV replication by isolation of viral DNA, regardless of symptoms. CMV DNAemia is measured every week after the end of prophylaxis for 6 weeks, biweekly for another 6 weeks, then monthly for 6 months, using the Cobas 8800 system (Roche Diagnostics). CMV DNAemia is also measured if clinically indicated. At our centre, CMV DNAemia in D+/R− HTRs is deemed clinically relevant and prompts antiviral treatment at a level of 150 IU/mL. Gastrointestinal endoscopy is not routinely performed at our centre; therefore, to reflect our clinical practice, CMV disease was defined for purposes of this study as a combination of CMV infection with disease-compatible clinical signs and symptoms, either as a viral syndrome or a tissue-invasive condition.7
Quantitative variables are reported as means with standard deviations. Categorical variables are reported as frequencies and percentages and were analyzed using the Fisher exact test.
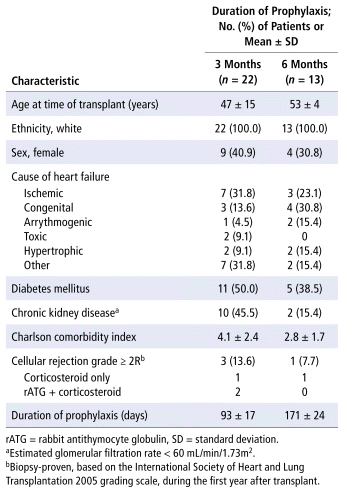
A total of 35 D+/R− HTRs met the inclusion criteria. Of these, 62.9% (22/35) received a 3-month course of prophylaxis, and 37.1% (13/35) received a 6-month course (as determined by the medical team). The baseline characteristics of patients did not differ between the 2 groups (Table 1).
TABLE 1 Baseline Characteristics

Valganciclovir was stopped early due to leukopenia in 13.6% (3/22) and 15.4% (2/13) of patients in the 3- and 6-month groups, respectively. Basiliximab was used for all patients except one, who was in the 6-month group. The mean duration of valganciclovir prophylaxis was 93 (standard deviation [SD] 17) days for the 3-month group and 171 (SD 24) days for the 6-month group.
A total of 19 patients (54.3%) presented with CMV infection during follow-up. A significantly greater proportion of patients receiving the 3-month course of prophylaxis experienced CMV infection relative to those who received a 6-month course (15/22 [68.2%] vs 4/13 [30.8%], risk difference 37.4%, 95% confidence interval [CI] 1.75 to 65.8, p = 0.04) (Figure 1). Overall, 15 patients were considered to have CMV disease, representing 78.9% of those with CMV infection and 42.9% of the total cohort (Figure 2). A greater proportion of patients presented with a first episode of CMV disease in the group who received 3 months of prophylaxis than in the group who received 6 months of prophylaxis, although this difference was not statistically significant (12/22 [54.5%] vs 3/13 [23.1%], risk difference 31.4%, 95% CI −3.7 to 59.4, p = 0.09). Most patients who experienced CMV disease had gastrointestinal involvement (11/12 vs 3/3 for patients with 3 and 6 months of prophylaxis, respectively). The single patient who did not experience gastrointestinal symptoms was considered to have CMV syndrome. No CMV infection was observed in patients who received treatment for acute rejection, and no breakthrough CMV infection was observed.
|
| ||
|
FIGURE 1 Occurrence of cytomegalovirus (CMV) infection and CMV disease in relation to duration of prophylaxis. | ||
|
| ||
|
FIGURE 2 Occurrence of cytomegalovirus (CMV) infection and CMV disease in the overall cohort. | ||
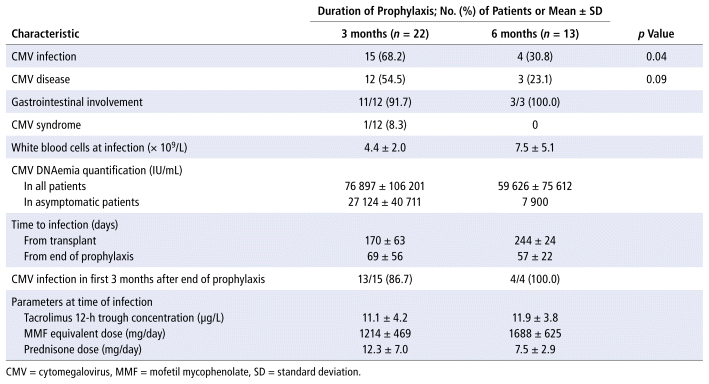
Table 2 presents the characteristics of interest at the time of CMV infection. No differences were observed between the groups in terms of the immunosuppressive agents given, their dosages, or their trough concentrations. Only one patient in the 3-month group was receiving sirolimus instead of mycophenolate at the time of CMV infection. Most CMV infections and diseases occurred in the first 3 months after the end of prophylaxis, regardless of the duration of prophylaxis received.
TABLE 2 Patient Characteristics at Time of CMV Infection
Despite adequate universal antiviral prophylaxis, CMV infection and disease remain significant infectious threats for high-risk D+/R− HTRs. More than half of our cohort (54.3%) had a CMV infection that prompted initiation of antiviral treatment, and most of these patients went on to experience CMV disease. The incidence of CMV infection but not CMV disease was significantly higher among patients who received 3 months of prophylaxis than among those who received 6 months of prophylaxis. The overall incidence of CMV disease in our study (42.9%) corroborated previously reported rates among D+/R− HTRs. Some studies have reported incidence values ranging from as low as 26.5% to as high as 86%, whereas others have reported incidence values more in line with our data (52.6% and 66%).8,10–13 The incidence rate observed in our cohort was high, considering only one of the patients received antithymocyte globulin for induction. Lymphocyte-depleting agents are associated with an increased risk of CMV infection, whereas basiliximab is not.14,15
One explanation for the lower incidence rate of CMV infection among those who received 6 months of prophylaxis is the lower intensity of immunosuppression at the end of prophylaxis. The immunosuppressive regimen is usually more intense during the first 6 months after transplant. The target 12-hour trough concentration for tacrolimus is lowered to between 5 and 10 μg/L after 6 months, and, according to our institution’s protocol, prednisone is weaned somewhere between 9 and 12 months after transplant.7 Although not clearly associated with an increased risk of CMV disease, double compared with triple immunosuppression and lower blood concentrations of calcineurin inhibitors are associated with better early eradication of CMV DNAemia in solid organ transplant recipients with CMV disease.16 A longer duration of prophylaxis can cover this period of high-intensity immunosuppression. Indeed, among kidney transplant recipients, the incidence rate of CMV disease was lower when a longer duration of valganciclovir or ganciclovir prophylaxis was used.6,17 Not only is CMV infection delayed by a longer course of prophylaxis, but it can also be prevented. In our study, patients in the 6-month group who experienced an infection had mean tacrolimus 12-hour trough concentration at the time of infection (mean 244 days after transplant) of 11.9 μg/mL, which is higher than recommended at this time point. This over-immunosuppression could have contributed to their infection.
The results of our study contrast with those reported by Imlay and others,8 who conducted a retrospective analysis of CMV disease and outcomes in CMV D+R− HTRs. In a multivariable model that included 58 patients, Imlay and others8 found no difference in the incidence of proven or probable CMV disease between those who received 6 months of prophylaxis and those who received 3 months (7/18 [38.9%] vs 9/40 [22.5%], respectively). However, the definition of CMV disease used by Imlay and others8 differed from the definition used by us and other authors.12,13,17 In particular, Imlay and others8 excluded a total of 12 possible episodes of gastrointestinal CMV disease from their analysis, without reporting information about the patients’ duration of prophylaxis. In contrast, we deemed that every patient with gastrointestinal symptoms and CMV replication had CMV disease, even without gastrointestinal endoscopy to prove CMV involvement; this approach reflects our current practice of treating such patients with antiviral therapy. Imlay and others8 would have classified these patients as having possible CMV disease and would therefore have excluded them from analysis. This difference in classification could explain, at least in part, the difference in results between our study and theirs.
CMV surveillance after the end of prophylaxis is of the utmost importance, regardless of the duration of prophylaxis. The mean time to CMV infection or disease after the end of prophylaxis was about 60 days in both groups. Extending weekly surveillance for up to 12 weeks after prophylaxis, as recommended by some authors,7 could help to identify patients with CMV infection before the appearance of symptoms. However, frequent surveillance can be a challenge for patients living in rural areas, where results may not be available for a couple of weeks after blood sampling. It is not known whether more frequent or longer duration of surveillance could prevent development of CMV disease, nor was our study designed to test this hypothesis.
The major limitation of this study was the small number of patients, which could have affected study power and our ability to detect a difference between the groups. The small number of CMV infections and diseases also affected the precision of our results, as reflected by the large 95% CIs reported. As such, we cannot draw conclusions about the magnitude of the reduction in CMV infections with a 6-month course of prophylaxis, given it lay somewhere between 1.75% and 65.8%. Our study was not designed to compare patients who did and did not experience CMV disease, or to identify risk factors for infection. Because routine endoscopy is not used in our centre for diagnosis of CMV disease, confounding factors or other gastrointestinal pathology may have been missed or not collected. Given the retrospective nature of our study, we could only collect data for the results of investigations and tests that were done at the time of care. Our definitions of CMV infection and disease were intended to reflect clinical practice at our centre. Therefore, a direct association between duration of prophylaxis and rate of infection cannot be proposed.
In this retrospective, descriptive single-centre study involving high-risk D+/R− HTRs, those who received 6 months of valganciclovir prophylaxis had a lower risk of CMV infection than those who received 3 months of prophylaxis. Most cases of CMV disease and infection occurred in a span of 3 months after the end of prophylaxis, regardless of the duration of prophylaxis. More comparative data are needed to prove the association between a longer duration of prophylaxis and a lower rate of CMV disease. When possible, for high-risk HTRs, 6 months of valganciclovir could be considered.
1 Cytomegalovirus (CMV) and congenital CMV infection. Clinical overview of CMV and congenital CMV. Centers for Disease Control and Prevention (US); 2024 [cited 2025 Mar 31]. Available from: https://www.cdc.gov/cytomegalovirus/hcp/clinical-overview/?CDC_AAref_Val= https://www.cdc.gov/cmv/clinical/overview.html
2 Kotton CN, Torre-Cisneros J, Aguado JM, Alain S, Baldanti F, Baumann G, et al. Cytomegalovirus in the transplant setting: where are we now and what happens next? A report from the International CMV Symposium 2021. Transpl Infect Dis. 2022;24(6):e13977.
Crossref PubMed PMC
3 Razonable RR, Humar A. Cytomegalovirus in solid organ transplant recipients – guidelines of the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transplant. 2019 33(9):e13512.
Crossref PubMed
4 Linares L, Sanclemente G, Cervera C, Hoyo I, Cofan F, Ricart MJ, et al. Influence of cytomegalovirus disease in outcome of solid organ transplant patients. Transplant Proc. 2011;43(6):2145–8.
Crossref PubMed
5 Chih S, McDonald M, Dipchand A, Kim D, Ducharme A, Kaan A, et al. Canadian Cardiovascular Society/Canadian Cardiac Transplant Network position statement on heart transplantation: patient eligibility, selection, and post-transplantation care. Can J Cardiol. 2020;36(3):335–56.
Crossref PubMed
6 Humar A, Lebranchu Y, Vincenti F, Blumberg EA, Punch JD, Limaye AP, et al. The efficacy and safety of 200 days valganciclovir cytomegalovirus prophylaxis in high-risk kidney transplant recipients. Am J Transplant. 2010;10(5):1228–37.
Crossref PubMed
7 Kotton CN, Kumar D, Caliendo AM, Huprikar S, Chou S, Danziger-Isakov L, et al. The third international consensus guidelines on the management of cytomegalovirus in solid-organ transplantation. Transplantation. 2018;102(6):900–31.
Crossref PubMed
8 Imlay H, Dumitriu Carcoana AO, Fisher CE, Wong B, Rakita RM, Fishbein DP, et al. Impact of valganciclovir prophylaxis duration on cytomegalovirus disease in high-risk donor seropositive/recipient seronegative heart transplant recipients. Transpl Infect Dis. 2020;22(3):e13255.
Crossref PubMed PMC
9 Velleca A, Shullo MA, Dhital K, Azeka E, Colvin M, DePasquale E, et al. The International Society for Heart and Lung Transplantation (ISHLT) guidelines for the care of heart transplant recipients. J Heart Lung Transplant. 2023;42(5):e1–e141.
Crossref PubMed
10 Gupta S, Mitchell JD, Markham DW, Mammen PPA, Patel PC, Kaiser P, et al. High incidence of cytomegalovirus disease in D+/R− heart transplant recipients shortly after completion of 3 months of valganciclovir prophylaxis. J Heart Lung Transplant. 2008;27(5):536–9.
Crossref PubMed
11 Kijpittayarit-Arthurs S, Eid AJ, Kremers WK, Pedersen RA, Dierkhishing RA, Patel R, et al. Clinical features and outcomes of delayed-onset primary cytomegalovirus disease in cardiac transplant recipients. J Heart Lung Transplant. 2007;26(10):1019–24.
Crossref PubMed
12 Jewani PK, Pouch SM, Kissling KT. Incidence of cytomegalovirus in cardiac transplant recipients receiving induction immunosuppression with antithymocyte globulin. Clin Transplant. 2018;30(11):e13420.
Crossref
13 Van Aelst LNL, Droogné W, Vörös G, Van Cleemput J. Combination prophylaxis in CMV high risk heart transplant recipients – a single centre experience. Clin Transplant. 2023;37(3):e14848.
Crossref
14 Gala-Lopez BL, Senior PA, Koh A, Kashkoush SM, Kawahara T, Kin T, et al. Late cytomegalovirus transmission and impact of T-depletion in clinical islet transplantation. Am J Transplant. 2011;11(12):2708–14.
Crossref PubMed
15 Portela D, Patel R, Larson-Keller JJ, Ilstrup DM, Wiesner RH, Steers JL, et al. OKT3 treatment for allograft rejection is a risk factor for cytomegalovirus disease in liver transplantation. J Infect Dis. 1995; 171(4):1014–8.
Crossref PubMed
16 Asberg A, Jardine AG, Bignamini AA, Rollag H, Pescovitz MD, Gahlemann CC, et al. Effects of the intensity of immunosuppressive therapy on outcome of treatment for CMV disease in organ transplant recipients. Am J Transplant. 2010;10(8):1881–8.
Crossref PubMed
17 Doyle AM, Warburton KM, Goral S, Blumberg E, Grossman RA, Bloom RD. 24-week oral ganciclovir prophylaxis in kidney recipients is associated with reduced symptomatic cytomegalovirus disease compared to a 12-week course. Transplantation 2006;81(8):1106–11.
Crossref PubMed
Address correspondence to: Vincent Leclerc, Institut universitaire de cardiologie et de pneumologie de Québec, Université Laval, 2725, chemin Sainte-Foy, Québec QC G1V 4G5, email: vincent.leclerc@ssss.gouv.qc.ca
Competing interests: None declared.
Funding: None received.
Acknowledgements: The authors would like to thank Serge Simard, biostatistician at the Centre de recherche de l’Institut universitaire de cardiologie et de pneumologie de Québec – Université Laval, for his help with the statistical analysis performed.
Submitted: August 15, 2024
Accepted: November 11, 2024
Published: May 14, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 2, 2025