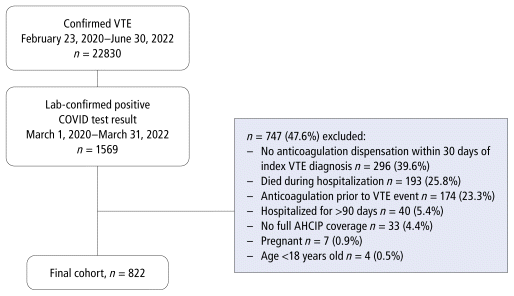
FIGURE 1 Flowchart for patient inclusion. AHCIP = Alberta Health Care Insurance Plan, VTE = venous thromboembolism.
Jesalyn Clarkson, Micheal Guirguis, Xueyi Chen, Timothy Chan, Priya Samuel, and Tammy J BungardTo cite: Clarkson J, Guirguis M, Chen X, Chan T, Samuel P, Bungard TJ. COVID-19 and recurrent venous thromboembolism (CORE-VTE). Can J Hosp Pharm. 2025;78(3):e3702. doi: 10.4212/cjhp.3702
ABSTRACT
Background
Venous thromboembolism (VTE) events occur in association with COVID-19, and the optimal duration of anticoagulant treatment is uncertain.
Objectives
The primary objectives were to determine the duration of anticoagulant prescriptions filled after COVID-19–associated VTE and the proportion of patients experiencing VTE recurrence. The secondary objective was to determine the proportion of patients who experienced bleeding events.
Methods
This retrospective cohort study analyzed data for cases of COVID-19–associated VTE across Alberta, Canada, confirmed between February 23, 2020, and June 30, 2022. Outpatient cases of VTE were identified using codes from the International Classification of Diseases, Ninth Revision plus Alberta Health Service procedure codes. Inpatient cases of VTE and bleeding events were identified using codes from the International Classification of Diseases and Related Health Problems, 10th Revision.
Results
Among the 822 patients included in the analysis, median follow-up was 359.0 days, with the majority of follow-up occurring while patients were off treatment (median 273.0 days). The median age was 59 years, 61.9% of the patients were male, 82.1% had a pulmonary embolism, 71.9% had not received COVID-19 vaccinations, and the median Charlson comorbidity index score was 1. The median duration of treatment was 100.0 (interquartile range 71.0–190.0) days, with 360 patients (43.8%) filling their prescriptions for 0–3 months, 234 (28.5%) for 4–6 months, 181 (22.0%) for 7–12 months, and 47 (5.7%) for more than 12 months. Overall, recurrent VTE occurred in 49 patients (6.0%), 24 of whom had continuously filled their anticoagulant prescriptions up to the time of the event. Bleeding occurred in 72 patients (8.8%).
Conclusions
Given the prolonged follow-up period off anticoagulant treatment for most patients, it is reasonable to consider 3–6 months of therapy for COVID-19–associated VTE.
Keywords: anticoagulants, COVID-19, venous thromboembolism, deep vein thrombosis, pulmonary embolism
RÉSUMÉ
Contexte
Les événements de thromboembolie veineuse (TEV) surviennent en association avec la COVID-19, et la durée optimale du traitement anticoagulant est incertaine.
Objectifs
L’objectif principal consistait à déterminer la durée des ordonnances d’anticoagulants exécutées après une TEV associée à la COVID-19 et la part des patients chez qui la TEV avait récidivé. L’objectif secondaire consistait à déterminer la part des patients vivant des événements hémorragiques.
Méthodologie
Cette étude de cohorte rétrospective a analysé les données des cas de TEV associés à la COVID-19 en Alberta, au Canada, confirmés entre le 23 février 2020 et le 30 juin 2022. Les cas ambulatoires de TEV ont été identifiés à l’aide des codes de la Classification internationale des maladies, neuvième révision, ainsi que des Codes de procédure des Services de santé de l’Alberta. Les hospitalisations pour TEV et les événements hémorragiques ont été identifiés à l’aide des codes de la Classification internationale des maladies et des problèmes de santé connexes, dixième révision.
Résultats
Parmi les 822 patients inclus dans l’analyse, le suivi médian était de 359,0 jours, la majorité du suivi ayant eu lieu lorsque les patients n’étaient plus sous traitement (médian 273,0 jours). L’âge médian était de 59 ans; 61,9 % des patients étaient des hommes; 82,1 % avaient une embolie pulmonaire; 71,9 % n’avaient pas reçu de vaccination contre la COVID-19; et le score médian de l’indice de comorbidité de Charlson était de 1. La durée médiane du traitement était de 100 jours (écart interquartile 71–190 jours), avec 360 patients (43,8 %) ayant exécuté leur prescription pendant 0 à 3 mois, 234 (28,5 %) pendant 4 à 6 mois, 181 (22,0 %) pendant 7 à 12 mois, et 47 (5,7 %) pendant plus de 12 mois. Globalement, une récidive de la TEV est survenue chez 49 patients (6,0 %), dont 24 avaient continuellement exécuté leur ordonnance d’anticoagulants jusqu’à l’événement. Des saignements sont survenus chez 72 patients (8,8 %).
Conclusions
Étant donné la durée prolongée du suivi sans traitement anticoagulant pour la plupart des patients, il est raisonnable de considérer 3 à 6 mois de traitement pour la TEV associée à la COVID-19.
Mots-clés: anticoagulants, COVID-19, thromboembolie veineuse, thrombose veineuse profonde, embolie pulmonaire
Clinical manifestations arising from COVID-19 range from being asymptomatic to experiencing a critically life-threatening condition.1 In particular, patients with COVID-19 experienced increased rates of thrombotic complications.2 Although the situation is not fully understood, it has been demonstrated that endothelial damage, immobilization, and changes in coagulation parameters may contribute to the hypercoagulable state and increased risk of venous thromboembolism (VTE) among patients who test positive for COVID-19.1,3 According to the limited literature available, the median duration of anticoagulation treatment following VTE diagnosis in patients with COVID-19 is 3 to 6 months,4–8 with reported rates of VTE recurrence being consistently low, ranging from 0% to 2.6%.4–12 Many previous studies restricted follow-up to 3 months,9,12 which limits the data about longer-term treatment outcomes. Although some studies have followed patients to about 1 year,4–6,10,11 most of these are limited by small sample size (≤ 100 patients), thereby providing scarce data for estimation of recurrent events. The rates of bleeding events, whether major, clinically relevant non-major, minor, or overall, have ranged from 0% to 20%.4,7,9–13 The majority of recurrent thrombotic events and bleeding events have occurred within the first 30 days.9,12
It is uncertain how strongly provoking COVID-19 is as a risk factor for VTE, which means decisions about treatment duration can be less than clear.14,15 As such, using administrative data for Alberta, we sought to assess practices for anticoagulant treatment and outcomes among patients with VTE in association with positive COVID-19 test results.
This retrospective cohort study received ethics approval from the University of Alberta (Pro00124559). A waiver of consent was granted due to the retrospective design. The study was conducted in Alberta, Canada, and used administrative data spanning the period from February 23, 2020, to September 30, 2022. More specifically, patients with index VTE between February 23, 2020, and June 30, 2022, were identified, with follow-up extending to September 30, 2022, to allow a minimum of 3 months of follow-up data for each patient.
Adult patients with a diagnosis of deep vein thrombosis or pulmonary embolism within 1 week before or 90 days after a confirmed positive result on COVID-19 testing were included. For outpatients, VTE was confirmed using codes from the International Classification of Diseases, Ninth Revision in conjunction with diagnostic imaging (e.g., compression ultrasonography, computed tomography) within 7 days.16 For inpatients, VTE was confirmed with codes from the International Classification of Diseases and Related Health Problems, 10th Revision (ICD-10). Patients had to have filled an anticoagulant prescription in the community within 30 days after the index VTE to be included. Patients were excluded if they were less than 18 years old, were pregnant, were receiving anticoagulants in the 90 days before the index VTE, were hospitalized for 90 days or more, died during the hospital stay, or had no Alberta Health Care Insurance Plan (AHCIP) coverage within 3 years before the index VTE.
The Alberta Strategy for Patient Oriented Research SUPPORT unit (AbSPORU) extracted and collated all relevant administrative data. The National Ambulatory Care Reporting System (NACRS), Discharge Abstract Database (DAD), and Alberta Practitioner Claims databases were used to gather comorbidity data. VTE events were identified using the DAD for inpatients and the Practitioner Claims database for outpatients, whereas bleeding events for the entire cohort were identified using the DAD and the NACRS. Additional data were linked using the following databases: Pharmaceutical Information Network for anticoagulant prescription fill data, based on Anatomical Therapeutic Chemical classification system codes and Drug Identification Numbers; Provincial Registry for death records; and the Provincial Laboratory database for results of COVID-19 testing and other laboratory values.
Our primary objectives included reporting, for patients with COVID-19–associated VTE, the duration of anticoagulant prescriptions filled (median time and proportions categorized as fills for 0–3, 3–6, 7–12, and > 12 months), with classification of the anticoagulant as direct oral anticoagulant (DOAC), warfarin, low-molecular-weight heparin (LMWH), fondaparinux, heparin, or switch in therapy. Duration of treatment was determined as the date of first dispensation to the final day of the last dispensation before a 31-day break in supply. Drug switches captured cases involving a change in anticoagulant after dispensation of the first drug for 7 days or longer.
We also aimed to report the proportion of patients with recurrent VTE (pulmonary embolism or deep vein thrombosis), both on and off treatment, and to describe details about the timing of recurrence in relation to the last anticoagulant prescription filled. Recurrent VTE was defined in the same way as the index VTE, with the recurrent event occurring beyond 90 days from the initial event. Treatment status was classified in 3 ways: uninterrupted, interrupted, or off treatment. VTE in the context of uninterrupted treatment was defined as a VTE event without a 31-day break in prescription fills for anticoagulant treatment. VTE in the context of interrupted treatment was defined as a VTE event after a 31-day break in prescription fills, but with anticoagulant coverage at the time of the event. VTE while off treatment was defined as a VTE event after a 31-day break in treatment, with no prescription fills in effect at the time of the event. We used a 31-day (full month) break in these definitions, given the acuity of VTE being actively treated, the likelihood of a limited duration of treatment, and the need to have definitions in place for administrative data.
Our secondary objective was to report the proportions of bleeding events that occurred while patients were on and off treatment and to describe details about the timing of these events in relation to the last anticoagulant prescription filled. Bleeding included both major bleeding and clinically relevant non-major bleeding, identified using ICD-10 codes.
Means (with standard deviations) and medians (with interquartile ranges [IQRs]) are used to report continuous variables, and frequencies with percentages are used to report categorical variables. The χ2 test (or the Fisher exact test if n ≤ 5) was used to compare differences between categorical variables of specified cohorts. For continuous variables, a t test or Mann–Whitney test was used to compare differences between 2 groups. A one-way analysis of variance or the Kruskal–Wallis test was used to compare differences involving more than 2 groups.
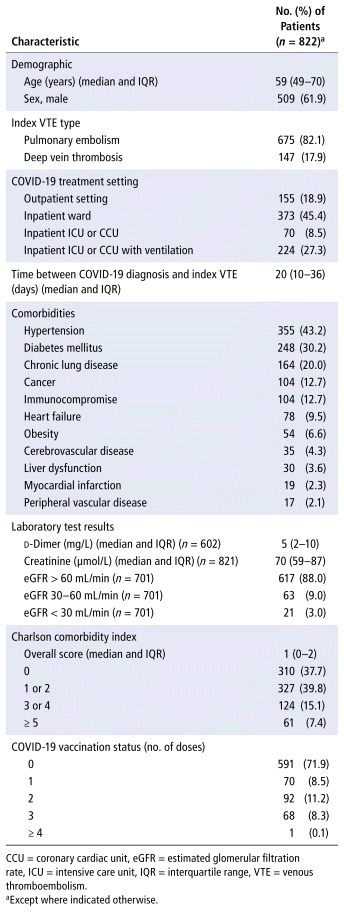
A total of 1569 patients with a positive result on COVID-19 testing and confirmed VTE were screened, of whom 822 (52.4%) were included in the analysis (Figure 1). Most of the 747 exclusions related to not having an anticoagulant prescription dispensed within 30 days of the index VTE (39.6%), death during hospitalization (25.8%), and receiving an anticoagulant before the VTE event (23.3%). Among included patients, the median age was 59 (IQR 49–70) years, more than half were men (61.9%), and the majority had pulmonary embolism (82.1%), with a median time between positive COVID-19 test result and the index VTE of 20 (IQR 10–36) days (Table 1). The majority of patients (81.1%) received their COVID-19 diagnosis as an inpatient (45.4% on the ward, 35.8% in the intensive care unit or coronary cardiac unit), and most (71.9%) were unvaccinated before the index VTE. Overall, the Charlson comorbidity index (CCI) scores were low (median 1 [IQR 0–2]). The most common comorbidities were hypertension (43.2%), diabetes (30.2%), chronic lung disease (20.0%), cancer (12.7%), and immunocompromise (12.7%).
|
| ||
|
FIGURE 1 Flowchart for patient inclusion. AHCIP = Alberta Health Care Insurance Plan, VTE = venous thromboembolism. | ||
TABLE 1 Baseline Characteristics
During the 950-day study period, the median follow-up was 359.0 (IQR 249.0–505.0) days, with patients being off treatment for the majority of the follow-up period (median 273.0 [IQR 162.0–408.0] days). Overall, the median duration of treatment was 100.0 (IQR 71.0–190.0) days. Altogether, 360 patients (43.8%) filled prescriptions for 0–3 months, 234 (28.5%) for 4–6 months, 181 (22.0%) for 7–12 months, and 47 (5.7%) for more than 12 months. Neither the COVID-19 treatment setting (inpatient vs ambulatory) nor the CCI score affected treatment duration (p = 0.58 and p = 0.17, respectively). Of the total population, 696 (84.7%) had a prescription for a DOAC (including 436 [62.6%] for apixaban and 251 [36.1%] for rivaroxaban), 92 (11.2%) received LMWH (with 88 [95.7%] of these receiving tinzaparin), and 33 (4.0%) received warfarin. Most patients (747 [90.9%]) remained on a single agent with no changes in treatment throughout the study period. When a switch did occur (n = 75), it was most often from an injectable agent (usually LMWH) to a DOAC (43 [57.3%] of the switches); the second most frequent type of switch was from a DOAC to an injectable agent (most often LMWH) (11 [14.7%] of the switches). When prescriptions were analyzed in relation to CCI score, we found that the proportion of patients initially filling prescriptions for LMWH increased with increasing CCI score: 8.4% (26/310) of patients with CCI of 0, 8.3% (27/327) of those with CCI of 1 or 2, 16.9% (21/124) of those with CCI of 3 or 4, and 29.5% (18/61) of those with CCI ≥ 5.
The overall number of recurrent VTE events was 49 (6.0%), with similar rates of pulmonary embolism (29 [59.2%]) and deep vein thrombosis (20 [40.8%]). The median time from the index VTE to the recurrent event was 168.0 (IQR 127.0–206.0) days. Of those with recurrent VTE, 24 had uninterrupted treatment, 10 had interrupted treatment, and 15 were off treatment at the time of the event. The COVID-19 treatment setting and CCI score did not influence recurrence rates (p = 0.09 and p = 0.05, respectively). The last anticoagulant prescription filled before or at the time of the recurrent event was most commonly for apixaban (26 [53.1%]), rivaroxaban (11 [22.4%]), or LMWH (8 [16.3%]). Of the 24 patients with recurrent VTE despite uninterrupted therapy, the majority were receiving a DOAC (20 patients [83.3%]), followed by LMWH (3 patients [12.5%]). The findings were similar among the 10 patients with recurrent VTE and interrupted therapy, with DOAC being most common (8 patients [80.0%]), followed by LMWH or warfarin (1 patient [10.0%] for both). Of the 26 patients with recurrent VTE who were receiving apixaban, 24 (92.3%) had a dosage of 5 mg twice daily, and among the 11 patients with recurrent VTE who were receiving rivaroxaban, all (100%) had a dosage of 20 mg daily.
Overall, there were 72 (8.8%) bleeding events, with a median time from the index VTE of 62.5 (IQR 23.0–212.0) days. Of the 72 events, 2 (2.8%) were fatal (intracerebral hemorrhage, with prescription fill data for tinzaparin, and hemoptysis, with prescription fill data for rivaroxaban). Among patients with bleeding events, 51 had uninterrupted treatment (with bleeding occurring a median of 33 days after the index VTE), and 21 were off treatment at the time of the event (which occurred at a median of 288 days after the index VTE). The last anticoagulant prescription filled before or at the time of the bleeding event was most commonly for apixaban (28 [38.9%]) or rivaroxaban (26 [36.1%]); LMWH accounted for another 11 cases (15.3%).
The median duration of anticoagulant treatment for our cohort was 100.0 (IQR 71–190) days, with the majority of the study population (594 [72.3%]) having prescriptions filled for up to 6 months. VTE recurrence rates were higher than expected, at 6.0%, whereas bleeding rates were similar to previous data, at 8.8%. Large proportions of VTE recurrences (49.0%) and bleeding events (70.8%) occurred in association with uninterrupted treatment, whereas 20.4% of VTE recurrences occurred in patients with interrupted treatment. Interestingly, neither the CCI score nor COVID-19 treatment setting seemed to affect the duration of anticoagulation or the rate of recurrent VTE, perhaps implying that risk may not vary once the index VTE is appropriately treated.
In this study, the median time from COVID-19 infection to the index VTE was 20 (IQR 10–36) days, similar to findings reported by Kartsios and others13 (median 11 [IQR 6–33] days), implying that the VTE presentation is likely to occur early in the course of COVID-19 disease. To our knowledge, our study had the largest cohort to date, and the follow-up duration of 359 days was among the longest reported so far, with patients being off treatment for the majority of follow-up. Most patients (72.3%) filled their anticoagulant prescriptions for a finite duration of up to 6 months, consistent with other studies reporting a median duration of therapy of 93 (IQR 84–149) days7 and 4.6 (IQR 3.5–6.3) months.8 For those with treatment longer than 6 months, the rationale for extension was generally the presence of antiphospholipid syndromes, a history of VTE or left ventricular thrombus, or persistent risk factors.4,7 Our exclusion criteria were intended to eliminate those with concomitant conditions warranting long-term treatment; as such, we surmise that the extended duration of treatment for some patients may be the result of variations in prescribing practices for VTE, which our own experience has shown tend to be tremendously heterogeneous.
Consistent with previous studies, discharge prescriptions were most commonly for DOACs (largely apixaban in our study), followed by LMWH and warfarin.4,7,10,12 Interestingly, as the CCI score increased, the number of patients initially started on tinzaparin (rather than a DOAC) also grew. Although we cannot be sure, it may be that as the number of comorbid conditions increased, prescribers felt more confident treating patients with LMWH.
The overall rate of recurrent VTE in this study (6.0%) was higher than reported in other COVID-19–specific trials, although we had a longer duration of follow-up. Buso and others12 reported a recurrence rate of 1.1% within 90 days of follow-up, although it was unclear if events in that study occurred while patients were on or off anticoagulation. In a recent prospective cohort study of 431 patients, a recurrence rate of 2.6% was reported for patients who had discontinued anticoagulation after receiving 3–6 months of treatment.8 Interestingly, half of those with recurrent VTE in our cohort were receiving uninterrupted anticoagulant treatment at the time of the event, and the majority had filled prescriptions for DOAC doses appropriate for treatment of acute VTE. We may thus infer that these recurrent events were not due to inappropriate dosing but may have been related to non-adherence or patient-specific factors and the hypercoagulable state of patients with COVID-19. Although recurrence rates were proportionally low among patients receiving warfarin and LMWH, recurrence occurred more often with these therapies than with DOACs. Although the overall recurrence rate of 6.0% was higher than previously reported in the COVID-19–specific literature, it was similar to the rate among those with recurrence of a nonsurgically provoked VTE (5%–5.8%) and lower than the rate among those with recurrence after an unprovoked VTE (7.9%–10%).15,17
In our cohort, the rate of bleeding was 8.8%, similar to the pooled incidence of 7.8% reported in a systematic review and meta-analysis of COVID-19 patients (with 3.9% considered major)18 and the 10% rate in a recent cohort study.7 The median duration from index VTE to bleeding event was 62.5 (IQR 23.0–212.0) days, similar to data reported elsewhere,19 which may indicate that, in addition to being related to anticoagulation therapy, bleeding events are due to severe inflammation and imbalances in platelet production and destruction in patients with COVID-19.18
This study had some limitations. First, because of the restrictions associated with the COVID-19 pandemic, we were able to conduct only a retrospective observational study of administrative data, so the analysis was subject to limitations related to data availability and coding. Second, we had to rely on dispensing data and therefore assumed that patients took their prescribed medications as instructed. Notably, 39.6% of potential participants were excluded from the analysis because they did not fill a prescription for an anticoagulant in the community within 30 days of the index VTE. The rationale for not filling prescriptions is unclear, but may have been due to distal clots not requiring treatment, the cost of prescriptions, or personal beliefs that treatment was unnecessary. Additionally, an Alberta audit of COVID-19–related VTE revealed that 12.9% of patients lacked anticoagulant prescription fills following acute VTE,20 suggesting that our observation reflects a real issue in this population. Third, we assumed that once a greater than 1-month break in prescription fill data emerged, patients were no longer taking anticoagulants; hence, we may have overestimated the time for which patients were not taking anticoagulants. To help mitigate this potential problem, we explored the number of patients who had anticoagulant dispensing following a 31-day break in treatment until the study end date; this post hoc analysis showed that 699 (85.0%) of the patients did not have a prescription dispensed after the break in treatment, which gave us more confidence that anticoagulation was not restarted.
Within a large cohort of patients with COVID-19–associated VTE, most had anticoagulant prescription fills for only 3–6 months. The median follow-up throughout the study was 359 days, and once anticoagulant prescriptions were not filled, we followed patients for a median of 273 days. The overall rate of VTE recurrence was 6.0%, similar to recurrence rates associated with provoked nonsurgical factors, and this outcome occurred most commonly among those having uninterrupted prescription fill data. The rate of bleeding (8.8%) was also consistent with previous data. Given the prolonged follow-up period without anticoagulant fills, for most patients it is reasonable to consider 3–6 months of therapy for COVID-19–associated VTE.
1 Cuker A, Peyvandi F. COVID-19: hypercoagulability. In: Leung LLK, Tirnauer JS, editors. UpToDate [database on internet]. UpToDate Inc; 2017 [updated 2022 May 24; cited 2022 Jul 27]. Available from: https://www.uptodate.com/contents/covid-19-hypercoagulability
2 COVID-19-associated coagulability. In: DynaMed [database on internet]. EBSCO Information Services; 1995– ; [updated 2018 Nov 30; cited 2022 July 27]. Accessed through institutional subscription.
3 Thachil J, Tang N, Gando S, Falanga A, Cattaneo M, Levi M, et al. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J Thromb Haemost. 2020;18(5):1023–26.
Crossref
4 Delrue M, Stépanian A, Voicu S, Nassarmadji K, Sène D, Bonnin P, et al. No VTE recurrence after 1-year follow-up of hospitalized patients with COVID-19 and a VTE event: a prospective study. Chest. 2022; 162(1):226–9.
Crossref
5 Ageno W, Antonucci E, Poli D, Bucherini E, Chistolini A, Fregoni V, et al. Venous thromboembolism secondary to hospitalization for COVID-19: patient management and long-term outcomes. Res Pract Thromb Haemost. 2023;7(4):100167.
Crossref PMC
6 Filippi L, Tonello D, Milazzo D, Turcato G, Panepinto L, Marchetti M. Follow-up of pulmonary embolism in patients with COVID-19 [abstract]. Chest. 2022;161(6 Suppl):A597.
Crossref
7 Whyte MB, Barker R, Kelly PA, Gonzalez E, Czuprynska J, Patel RK, et al. Three-month follow-up of pulmonary embolism in patients with COVID-19. Thromb Res. 2021;201:113–5.
Crossref PMC
8 Jara-Palomares L, Bikdeli B, Jiménez D, Muriel A, Martin Del Pozo M, Demelo-Rodríguez P, et al. Rate of recurrence after discontinuing anticoagulation therapy in patients with COVID-19–associated venous thromboembolism. JAMA Intern Med. 2022;182(12):1326–8.
Crossref PubMed PMC
9 Demelo-Rodríguez P, Ordieres-Ortega L, Ji Z, Del Toro-Cervera J, de Miguel-Díez J, Álvarez-Sala-Walther LA, et al. Long-term follow-up of patients with venous thromboembolism and COVID-19: analysis of risk factors for death and major bleeding. Eur J Haematol. 2021; 106(5):716–23.
Crossref PubMed PMC
10 Ahmed M, Khan R, Xenou E, Fowler M, Sinha S, Lokare A, et al. After the storm; 6-month follow up outcomes and management of COVID-19 related venous thromboembolism [abstract PB0270]. Res Pract Thromb Haemost. 2021 [cited 2022 Jul 28];5(Suppl 2):e12589. Available from: https://www.sciencedirect.com/science/article/pii/S2475037922021926?via%3Dihub#s3255
11 Maino A, Landolfo M, Conci S, Vettorato E, Cozzio S, Magnoni S, et al. SARS-CoV-2 associated venous thromboembolism – a one-year follow-up. Ital J Med. 2022;16(Suppl 1):3.
12 Buso G, Mazzolai L, Rueda-Camino JA, Fernández-Capitán C, Jiménez D, Bikdeli B, et al.; RIETE Investigators. Pulmonary embolism in patients with COVID-19: comparison between different care settings. Semin Thromb Hemost. 2023;49(1):34–46.
Crossref
13 Kartsios C, Lokare A, Osman H, Perrin D, Razaq S, Ayub N, et al. Diagnosis, management, and outcomes of venous thromboembolism in COVID-19 positive patients: a role for direct anticoagulants? J Thromb Thrombolysis. 2021;51(4):947–52.
Crossref
14 Moores LK, Tritschler T, Brosnahan S, Carrier M, Collen JF, Doerschug K, et al. Prevention, diagnosis, and treatment of VTE in patients with coronavirus disease 2019. Chest. 2020;158(3):1143–63.
Crossref
15 Venous thromboembolism: duration of treatment [clinical guide]. Thrombosis Canada; 2021 Sep 27 [cited 2022 Jul 27]. Available from: https://thrombosiscanada.ca/hcp/practice/clinical_guides?language=en-ca&guideID=DURATIONOFANTICOAGULANTTHERAPY
16 Alotaibi AS, Wu C, Senthilselvan A, McMurtry MS. The validity of ICD codes coupled with imagining procedure codes for identifying acute venous thromboembolism using administrative data. Vasc Med. 2015;20(4):364–8.
Crossref PubMed
17 Iorio A, Kearon C, Filippucci E, Marcucci M, Macura A, Pengo V, et al. Risk of recurrence after a first episode of symptomatic venous thromboembolism provoked by a transient risk factor: a systematic review. Arch Intern Med. 2010;170(19):1710–6.
Crossref
18 Jiménez D, García-Sanchez A, Rali P, Muriel A, Bikdeli B, Ruiz-Artacho P, et al. Incidence of VTE and bleeding among hospitalized patients with coronavirus disease 2019. Chest. 2021;159(3):1182–96.
Crossref
19 Katsoularis I, Fonseca-Rodríguez O, Farrington P, Jerndal H, Lundevaller EH, Sund M, et al. Risks of deep vein thrombosis, pulmonary embolism, and bleeding after covid-19: nationwide self-controlled cases series and matched cohort study. BMJ. 2022;377:e069590.
Crossref PubMed PMC
20 Schultz L, Bungard TJ, Mackay E, Gill P, Guirguis M. Prevalence of venous thromboembolism and anticoagulant use in patients with COVID-19 in Alberta, Canada. Can J Hosp Pharm. 2022;75(4):286–93.
Crossref
Address correspondence to: Dr Tammy Bungard, Division of General Internal Medicine, University of Alberta, 8425 Aberhart Centre, 11402 University Avenue, Edmonton AB T6G 2J3, email: tammy.bungard@ualberta.ca
Competing interests: For activities not directly related to the study reported here, Tammy Bungard has received unrestricted research grants from and served on an advisory board for Pfizer; she also serves as Co-Chair of the Patient and Families Committee, Thrombosis Canada. No other competing interests were declared.
Funding: None received.
Submitted: August 27, 2024
Accepted: November 11, 2024
Published: July 9, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 3, 2025