Niina Kleiber, Brigitte Martin, Grégoire Leclair, Céline Thibault, Jean-Philippe Roy, and Élaine PelletierTo cite: Kleiber N, Martin B, Leclair G, Thibault C, Roy JP, Pelletier É. Safety assessment of povidone K-12 in IV acetaminophen in pediatrics. Can J Hosp Pharm. 2025;78(4):e3722. doi: 10.4212/cjhp.3722
ABSTRACT
Background
The potential toxicity of excipients is a recurrent issue in pediatrics, particularly for neonates. The first IV formulation of acetaminophen approved in Canada (Avir Pharma Inc) contains the excipient povidone K-12, which lacks safety data for individuals with immature renal function, specifically, neonates, infants, and those with anuria. Povidone is eliminated by the kidneys and may accumulate in these populations.
Objective
To assess the safety of IV povidone K-12 in pediatrics.
Methods
The safety of IV povidone K-12 was assessed by first reviewing the available data and then measuring the amount of povidone K-12 exceeding the molecular weight threshold for glomerular filtration (25 000 g/mol). Size exclusion chromatography was used to assess the molecular weight of povidone K-12 to allow estimation of the proportions of povidone K-12 below various molecular weight thresholds.
Results
Case reports of povidone accumulation causing organ failure and death in adults were found in the literature. However, the published data were insufficient to assess the risk of accumulation, as no reliable molecular weight determinations could be found. Measurements by chromatography showed that the amount of povidone exceeding the molecular weight threshold of 25 000 g/mol was less than 2 ppm (0.0002%), which suggests a low risk of accumulation despite immature renal function.
Conclusions
Povidone K-12 is unlikely to accumulate in neonates, infants, or patients with impaired renal function, with the possible exception of patients with anuria.
KEYWORDS: excipient, toxicity, acetaminophen, pain, povidone, povidone storage disease
RÉSUMÉ
Contexte
La toxicité potentielle des excipients est un problème fréquent en pédiatrie, en particulier chez les nouveau-nés. La première formulation IV de l’acétaminophène approuvée au Canada (Avir Pharma Inc) contient l’excipient povidone K-12, pour lequel il n’existe pas de données d’innocuité chez les personnes dont la fonction rénale est immature, notamment les nouveau-nés, les nourrissons et les patients anuriques. La povidone est éliminée par les reins et peut s’accumuler chez ces populations.
Objectif
Évaluer l’innocuité de la povidone K-12 (IV) en pédiatrie.
Méthodologie
L’innocuité de la povidone K-12 intraveineuse a été évaluée en examinant les données disponibles et en mesurant la quantité de povidone K-12 dépassant le seuil de la masse moléculaire de la filtration glomérulaire (25 000 g/mol). La chromatographie d’exclusion stérique a été utilisée pour déterminer la masse moléculaire de la povidone K-12 afin d’estimer les proportions de povidone K-12 en dessous de différents seuils de masse moléculaire.
Résultats
Des cas d’accumulation de povidone ayant causé une défaillance des organes et le décès chez des adultes ont été trouvés dans la littérature. Cependant, les données publiées ne permettaient pas d’évaluer le risque d’accumulation, car aucune détermination fiable de la masse moléculaire n’était disponible. Les mesures par chromatographie ont montré que la quantité de povidone dépassant le seuil de masse moléculaire de 25 000 g/mol était inférieure à 2 ppm (0,0002 %), ce qui laisse supposer un faible risque d’accumulation, malgré une fonction rénale immature.
Conclusions
Il est peu probable que la povidone K-12 s’accumule chez les nouveau-nés, les nourrissons ou les patients ayant une insuffisance rénale, à l’exception possible des patients anuriques.
Mots-clés: excipient, toxicité, acétaminophène, douleur, povidone, accumulation de la povidone
Acetaminophen has been administered by the IV route for more than 20 years in Europe and more than 10 years in the United States, but the IV formulation was approved in Canada more recently, in 2018.1 In Canada, the IV route is officially approved for children above 2 years of age,1,2 although in some other countries, it is approved for neonates.3–5 However, solid pharmacokinetic and pharmacodynamic data support the off-label use of IV acetaminophen for young children in Canada, including during the neonatal period.6–8 Canada has faced additional safety issues because, unlike the IV formulations of acetaminophen approved for neonates and infants in Europe and the United States, the first formulation to become available in Canada (Avir Pharma Inc) contains povidone K-12, a solubilizer for which safety data are lacking for children under 2 years of age.1 The current study was conducted at a time when the only IV formulation of acetaminophen available in Canada contained povidone K-12.
The potential toxicity of excipients is a recurrent issue for children because pediatric and neonatal populations are often excluded from studies on drug development.9 Povidone K-12 is a low-molecular-weight solubilizer used to increase the amount of dissolved drug per volume. The accumulation of povidone in the organs has been associated with povidone storage disease, which can lead to organ dysfunction and death.10–12 The United States Pharmacopeia provides specifications for povidone, defining a K-value for various pharmaceutical grades ranging from K-10 for lower-molecular-weight povidone to K-120 for higher-molecular-weight povidone.13
A 3.5% solution of povidone K-30 was first used as a plasma substitute and volume expander during the Second World War.14,15 Its use was later banned following repeated cases of povidone storage disease.
Povidone K-12 has a lower molecular weight than povidone K-30. Its elimination depends on glomerular filtration rate, which is lower in neonates and infants than in older children, for whom administration of povidone-containing IV acetaminophen is an on-label use.16–18 Whether this grade of povidone can lead to accumulation and povidone storage disease is unknown, and this uncertainty is one of the reasons the manufacturer does not recommend its use in children under 2 years of age. Other IV formulations of acetaminophen available in Canada, even those without povidone K-12, are also not recommended for this population.2,19 The potential long-term effects of povidone accumulation during organ maturation are also unknown.
In anticipation of the introduction of IV acetaminophen to our mother and child hospital, the institutional analgesic governance committee carried out the study reported here to assess the safety of IV povidone K-12 for neonates and infants.
Given the nature of this study and the absence of study participants, no ethics approval was needed.
To assess the risk of accumulation and toxicity associated with the use of IV povidone K-12 in neonates and infants, we reviewed the available published data and determined the molecular weights of povidone K-12 polymers and their distribution. On the basis of these results, we then developed specific recommendations to guide use of povidone K-12 in premature and term neonates and in patients with renal dysfunction.
Searches were carried out in the PubMed database and using the Google Scholar search engine using the conceptual groups [povidone] or [PVP] or [polyvinylpyrrolidone] AND [parenteral] and their related synonyms. Reference lists of included articles and reviews were screened for additional relevant literature. Pharmacopeias, handbooks,14,20 and technical documents provided directly to the researchers by Avir Pharma and BASF (manufacturers of povidone) were also consulted. Conference abstracts were excluded.
All available data, including clinical, chemical, in vitro, animal, and human data, were assessed. Data that allowed delineation of the risks related to povidone K-12 in neonates and infants were assessed and summarized. These data included the general chemical properties of povidone, pharmacokinetics based on these chemical properties. and pharmacodynamics (i.e., adverse events observed).
General chemical information on weight characterization of povidone was extracted from the literature. Data provided by the manufacturers were assessed, and the manufacturers were contacted for additional information as necessary.
Povidone K-12 contained in Avir Pharma’s product is manufactured by the BASF Group, according to current good manufacturing practices; BASF kindly provided a sample for characterization.
As described in further detail below, povidone is a polymer, and therefore the weight average molecular weight (Mw) and polydispersity index (PDI) are needed to accurately characterize molecular weight and its distribution. The Mw and the PDI were determined by size exclusion chromatography.21 Then, a mathematical model was used to determine the number average molecular weight (Mn) and estimate the proportions of povidone K-12 below various thresholds.22
Povidone (synonyms: polyvidone, polyvinylpyrrolidone, PVP) is a nontoxic, non-ionic, inert, water-soluble synthetic polymer used in the pharmaceutical, biomedical, cosmetic, and food industries.10 It is a polydisperse polymer, which means that it is composed of several similar molecules of different molecular weights.
The pharmacokinetics of povidone K-12 were assessed in terms of its absorption, distribution, metabolism, and elimination, as described below.
High-molecular-weight pharmaceutical-grade povidones are not absorbed and can be used as excipients for oral administration.10 They are used to increase the viscosity and physical stability of tablets and liquid preparations. Povidone K-12 is a low-molecular-weight povidone used only for IV administration and is, by definition, fully absorbed.10,14
When administered parenterally, povidone is widely distributed, and its retention in the body is inversely proportional to its viscosity (expressed using a constant K-value). Povidone K-12 has a lower viscosity, lower molecular weight, and lower retention time (time spent in the body after injection) than povidone K-100, which has high viscosity and high molecular weight and is largely retained in the body. Unchanged povidone may accumulate in the reticuloendothelial system.15
Povidone is not metabolized.10,14
Povidones are excreted through glomerular filtration (passive diffusion). The extent of elimination is inversely proportional to the molecular weight. Particles of a size exceeding the size of the kidneys’ glomerular endothelial fenestrae are too large to be eliminated.23 The exact weight threshold preventing povidone filtration has not been clearly determined, but it is usually accepted that molecules with a weight of 25 000 to 60 000 g/mol or larger are not filtered by the kidneys and may accumulate.10,15,23 Povidone K-12 is considered safe for IV use in individuals with normal and mature renal function. Studies in rats have shown that povidone K-12 displays properties similar to those of inulin, a molecule used to quantify glomerular filtration rate because it is solely eliminated by glomerular filtration without significant tubular excretion or reabsorption.10 Povidone K-12 has an average low molecular weight of 2000 to 3000 g/mol. Since it is a polymer, it contains different-sized molecules, including some larger than 25 000 g/mol (threshold for filtration). Unfortunately, only the mean molecular weight of povidone K-12 has been determined and is reported in published data. Therefore, determination of the number of molecules larger than 25 000 g/mol (determined threshold for filtration) is needed to assess its safety in neonates and infants with immature renal function and lower glomerular filtration rate.17,18,24
As mentioned above, only low-molecular-weight pharmaceutical-grade povidones are authorized for parenteral use, because high-molecular-weight povidones are not eliminated by the kidneys and therefore accumulate in the body, mainly in the reticuloendothelial system.15,25 The accumulation of this inert compound25 may lead to povidone storage disease. This disease was reported in 2014 by the European Medicines Agency among people who inject drugs, specifically those misusing methadone by injecting products made for oral use.26
Povidone storage disease is due to the accumulation of povidone in several organs, notably the reticuloendothelial system (lymph nodes, spleen, bone marrow).10
Accumulation in the bones induces osteonecrosis with pathological fractures, whereas medullar infiltration leads to anemia.10,26 Renal failure has also been commonly described.10,26 A skin disease called cutaneous thesaurismosis resulting from the accumulation of povidone contained in certain subcutaneous or intramuscular drugs was described in the 1960s. In its most severe forms, multiple organ dysfunction syndrome occurs and may be fatal.27
The occurrence of povidone storage disease depends on the molecular weight, the dose, the duration, and the route of administration. In adults, clinical complications have been described with povidone K-30 (marketed as a plasma substitute) with doses exceeding 70 g IV (1.2 g/kg) or 200 g SC or IM (3.3 g/kg),10 but histologic abnormalities have been observed with lower doses.20 Despite the absence of reported cases of povidone storage disease with low-molecular-weight pharmaceutical-grade povidones (such as povidone K-12), the German regulatory body has advised caution with repeated use in patients who have poor renal function.10
Our literature review highlighted that povidone K-12 could potentially lead to accumulation in neonates, infants, and patients with renal insufficiency. Quantifying the amount of povidone molecules exceeding the molecular weight threshold associated with a risk of accumulation was needed to infer the risk of toxicity.14
The United States Pharmacopeia provides a standard test to evaluate the average molecular weight of povidone by measuring the viscosity of aqueous solutions. A K-value can then be calculated from the viscosity measurement, with lower viscosities being associated with lower K-values. A sample of povidone comprises a polydisperse mixture of molecules of different molecular weights. The K-value, as well as the average molecular weight, is only indicative of the central tendency of the molecular weight distribution. Therefore, a low average molecular weight does not exclude the presence of some larger molecules in the sample. A full characterization of the sample requires additional information:
The reported Mw of povidone K-12 is 2000 to 3000 g/mol (according to the BASF technical document), which is well below the described molecular weight deemed at risk for accumulation. However, povidone K-12 polymers may display a large range of molecular weights with a Gaussian distribution around the mean.10,25 The Mw and Mn values are mandatory information to estimate weight distribution (expressed by the PDI, which is calculated as Mw/Mn).22 None of these characteristics have been reported by Avir Pharma or BASF.
Moreover, the methods for weight determination have changed significantly over time, which implies that initial reported values may be different from weights measured using new methods.
The lack of data and the potential unreliability of reported povidone molecular weight values motivated our team to carry out a determination of the molecular weights of povidone K-12 polymers and their distribution.
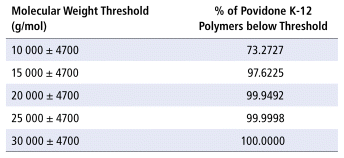
The Mw and PDI of povidone K-12 were measured by gel permeation chromatography. The sample Mw was 8340 g/mol, and the PDI was 1.641. This information allowed us to calculate the proportions of povidone K-12 polymers with molecular weight below various predetermined thresholds (Table 1).
TABLE 1 Proportion of Povidone K-12 Polymers with Molecular Weight below Thresholds

As previously detailed, the molecular weight threshold for glomerular filtration is usually accepted to be approximately 25 000 g/mol. With higher-molecular-weight substances, there is a risk of accumulation. The weight determination conducted in this study (Table 1) suggests that only 2 ppm (0.0002%) of povidone K-12 is above the threshold with risk of accumulation. Therefore, even with decreased renal function, significant accumulation is highly unlikely, except in patients with oligo-anuria. However, the threshold for glomerular filtration in the immature glomeruli of preterm neonates with ongoing nephrogenesis may be inferior to 25 000 g/mol, and therefore caution should be applied in this population.
The administered povidone dose in IV acetaminophen formulations is much lower than the dose previously associated with povidone storage disease in adults.10 Indeed, we estimated that with a maximum daily dose of acetaminophen of 60 mg/kg/day, a neonate would receive 6 mg/kg/day of povidone K-12. This represents less than 1% of the dose associated with povidone storage disease in previous adult case reports (1.2 g/kg of higher-molecular-weight polymers such as povidone K-30).10 However, this comparison is limited by the fact that no minimum toxic dose has been defined for neonates and infants. In addition, given the characterization of molecular weights above, it is expected that almost all of the polymers will be removed, as an amount of only 12 pg/kg is above the glomerular filtration threshold.
Together, these data suggest that even with decreased glomerular filtration rate, the risk of povidone accumulation is low. An exception applies in the case of patients with anuria, because in these patients, none of the administered povidone K-12 can be excreted. Moreover, the likelihood of accumulation increases with repeated administration.
The recommended duration of use was determined empirically and was driven by a risk minimization strategy, considering that (1) the daily acetaminophen dose includes less than 1% of the povidone dose associated with povidone storage disease; (2) multiple periods of administration may not be avoidable in the sickest (and most at risk) patients, who have an associated risk of accumulation; and (3) toxicologic data were missing for neonates with incomplete nephrogenesis and maturing organs.
Therefore, we developed the recommendations presented in Box 1 to ensure the safety of IV acetaminophen containing povidone K-12 in term and preterm neonates and infants.
BOX 1. Recommendations and Conclusions for IV Use of Acetaminophen Containing Povidone K-12, Based on Current Study

The analysis presented here has shown that the povidone K-12 contained in the Canadian formulation of IV acetaminophen (Avir Pharma Inc) is unlikely to accumulate in preterm and term neonates or infants or in patients with impaired renal function but preserved diuresis. However, clinicians must be vigilant in their choice of available formulations and must use only formulations containing excipients that are known, have been studied, and are safe for their intended population. The case of povidone K-12 is not unique; many drugs formulated for adults contain excipients whose effect is poorly characterized in neonates or infants. Given the availability of IV formulations of acetaminophen without povidone, we recommend using those alternatives, consistent with general precautions for use in the neonatal and pediatric populations. Manufacturers should provide a comprehensive safety assessment of their drugs, including the excipients, to avoid excluding populations such as neonates, premature infants, infants, or patients with medical conditions that may benefit from their drugs. Regulatory authorities should require a comprehensive safety assessment of drugs submitted for marketing, including the excipients used in the preparations.
Povidone accumulation can lead to povidone storage disease with deleterious consequences on organ function. The povidone K-12 included in the first available Canadian IV acetaminophen formulation is unlikely to significantly accumulate in neonates, infants, and patients with renal dysfunction. Its use should, however, be avoided in patients with anuria.
1 Acetaminophen injection [product monograph]. AVIR Pharma Inc; 2018 Nov 28 [cited 2025 May 8]. Available from: https://pdf.hres.ca/dpd_pm/00048547.PDF
2 Acetaminophen injection [product monograph]. Baxter Corporation; 2021 Aug 16 [cited 2025 May 8]. Available from: https://www.baxter.ca/sites/g/files/ebysai1431/files/2021-08/EN_ACETAMINOPHEN%20INJECTION.pdf
3 Acetaminophen injection [prescribing information]. Hikma-Pharmaceuticals USA Inc; 2022 Jun [cited 2025 May 8]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/206968s000lbl.pdf
4 Paracetamol B Braun. In: Répertoire des spécialités pharmaceutiques [database on Internet]. Agence nationale de sécurité du médicament et des produits de santé; [cited 2025 May 14]. Available from: https://agence-prd.ansm.sante.fr/php/ecodex/extrait.php?specid=61304982
5 Perfalgan 10mg/ml solution for infusion. Bristol-Myers Squibb Pharmaceuticals Limited; [cited 2025 May 14]. Available from: https://www.medicines.org.uk/emc/product/60/smpc
6 Mian P, Knibbe CAJ, Calvier EAM, Tibboel D, Allegaert K. Intravenous paracetamol dosing guidelines for pain management in (pre)term neonates using the paediatric study decision tree. Curr Pharm Des. 2017;23(38):5839–49.
Crossref PubMed
7 Conroy S, McIntyre J. The use of unlicensed and off-label medicines in the neonate. Semin Fetal Neonatal Med. 2005;10(2):115–22.
Crossref PubMed
8 Thibault C, Pelletier É, Nguyen C, Trottier ED, Doré-Bergeron MJ, DeKoven K, et al. The three W’s of acetaminophen in children: who, why, and which administration mode. J Pediatr Pharmacol Ther. 2023; 28(1):20–8.
PubMed PMC
9 Nellis G, Metsvaht T, Varendi H, Toompere K, Lass J, Mesek I, et al. Potentially harmful excipients in neonatal medicines: a pan-European observational study. Arch Dis Child. 2015;100(7):694–9.
Crossref PubMed
10 Robinson BV, Sullivan FM, Borzelleca JF, Schwartz SL. PVP: a critical review of the kinetics and toxicology of polyvinylpyrrolidone (povidone). CRC Press; 1990.
11 Dunn P, Kuo T, Shih LY, Wang PN, Sun CF, Chang MJ. Bone marrow failure and myelofibrosis in a case of PVP storage disease. Am J Hematol. 1998;57(1):68–71.
Crossref PubMed
12 Leh F, Stalund IV, Bjånes TK, Ohldieck C, Svarstad E, Leh S. Polyvinylpyrrolidone deposition disease in patients with intravenous opioid use: a case series. Hum Pathol. 2021;116:102–11.
Crossref PubMed
13 Povidone [monograph]. USP; 2019 May 1 [cited 2025 May 8]. Available from: https://www.usp.org/sites/default/files/usp/document/harmonization/excipients/m68070.pdf
14 Bühler V. Kollidon®. Polyvinylpyrrolidone excipients for the pharmaceutical industry. 9th ed. BASF; 2008.
15 Ravin HA, Seligman AM, Fine J. Polyvinyl pyrrolidone as a plasma expander; studies on its excretion, distribution and metabolism. N Engl J Med. 1952;247(24):921–9.
Crossref PubMed
16 Atiyeh BA, Dabbagh SS, Gruskin AB. Evaluation of renal function during childhood. Pediatr Rev. 1996;17(5):175–80.
Crossref PubMed
17 Hayton WL. Maturation and growth of renal function: dosing renally cleared drugs in children. AAPS PharmSci. 2000;2:E3.
Crossref
18 Gallini F, Maggio L, Romagnoli C, Marrocco G, Tortorolo G. Progression of renal function in preterm neonates with gestational age < or = 32 weeks. Pediatr Nephrol. 2000;15(1–2):119–24.
Crossref PubMed
19 Acetaminophen injection [product monograph]. B Braun Melsungen AG; 2023 Feb 24 [cited 2025 May 8]. Available from: https://pdf.hres.ca/dpd_pm/00069718.PDF
20 Polyvinylpyrrolidone toxicity profile. British Industrial Biological Research Association; 1991.
21 Held D, Kilz P. Size-exclusion chromatography as a useful tool for the assessment of polymer quality and determination of macromolecular properties. Chem Teach Int. 2021;3(2):77–103.
Crossref
22 Sheu WS. Molecular weight averages and polydispersity of polymers. J Chem Educ. 2001;78(4):554–5.
Crossref
23 Graham RC Jr, Karnovsky MJ. Glomerular permeability. Ultrastructural cytochemical studies using peroxidases as protein tracers. J Exp Med. 1966;124(6):1123–34.
Crossref PubMed PMC
24 Bennett WM. Drug prescribing in renal failure. Drugs. 1979;17(2):111–23.
Crossref PubMed
25 Kurakula M, Rao GSNK. Pharmaceutical assessment of polyvinylpyrrolidone (PVP): as excipient from conventional to controlled delivery systems with a spotlight on COVID-19 inhibition. J Drug Deliv Sci Technol. 2020;60:102046.
Crossref PubMed PMC
26 Assessment report for methadone medicinal products for oral use containing povidone. Procedure no. EMEA/H/A-107i/1395. European Medicines Agency; 2014 Jul 10 [cited 2025 May 8]. Available from: https://www.ema.europa.eu/en/documents/referral/methadone-article-107i-procedure-prac-assessment-report_en.pdf
27 Kuo TT, Hu S, Huang CL, Chan HL, Chang MJ, Dunn P, et al. Cutaneous involvement in polyvinylpyrrolidone storage disease: a clinicopathologic study of five patients, including two patients with severe anemia. Am J Surg Pathol. 1997;21(11):1361–7.
Crossref PubMed
Address correspondence to: Dr Niina Kleiber, CHU Sainte-Justine, 3175, chemin de la Côte Sainte-Catherine, Montréal QC H3T 1C5, email: niina.kleiber.med@ssss.gouv.qc.ca
Competing interests: For activities not related to the study reported here, Niina Kleiber has received grant funding from CHU Sainte-Justine, speaker’s honoraria from Western University (London, Ontario), and payment from Novartis for participation on a data safety monitoring board; she also serves on the Editorial Board of the Journal of Pediatric Pharmacology and Therapeutics. No other competing interests were declared.
Funding: None received.
Acknowledgement: The authors would like to thank Xavier Banquy for his contribution to this work, specifically the characterization of the povidone K-12 sample in his laboratory at the Faculty of Pharmacy, Université de Montréal. They would also like to thank the BASF Group, which kindly provided a sample of povidone K-12 for characterization.
Submitted: September 30, 2024
Accepted: June 24, 2025
Published: October 8, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 4, 2025