Tracy Shu, Simroop Ladhar, Dionzie Ong, Alfie Chung, Isla Drummond, and Aaron M TejaniTo cite: Shu T, Ladhar S, Ong D, Chung A, Drummond I, Tejani AM. Tamper-proofing multidose medications in hospitals: high-value, low-effort interventions to reduce medication waste. Can J Hosp Pharm. 2025;78(3):e3748. doi: 10.4212/cjhp.3748
ABSTRACT
Background
The Canadian health care sector contributes 4.6% of national greenhouse gas emissions, with medications accounting for 25% of that amount. Reducing waste from high-cost multidose items such as eye drops and inhalers can lower environmental and health care costs.
Objectives
To evaluate tamper-proofing practices in hospital pharmacy departments in British Columbia and to explore opportunities for standardization to reduce medication waste.
Methods
Site visits were conducted at 13 hospitals across 3 health authorities in British Columbia—Vancouver Coastal Health, Providence Health Care, and Fraser Health—to gather data on tamper-proofing practices in hospital pharmacy departments.
Results
Methods of tamper-proofing varied across departments. Key recommendations for improvement include the implementation of adhesive stickers or other tamper-evident features, standardization of the placement of tamper-evident adhesive stickers, prioritization of patient-specific medications for tamper-proofing, and development of and adherence to policies/procedures for most multidose medications before they leave the pharmacy.
Conclusions
Standardizing tamper-proofing practices can reduce medication waste and environmental impact, with potential for broader adoption across hospitals.
KEYWORDS: tamper-proofing, medication waste, multidose medications, standardization
RÉSUMÉ
Contexte
Le secteur des soins de santé canadien est responsable de 4,6 % des émissions de gaz à effet de serre nationales et les médicaments représentent 25 % de ce chiffre. La réduction du gaspillage des articles multidoses coûteux tels que les gouttes pour les yeux et les inhalateurs peut diminuer les coûts environnementaux et de santé.
Objectifs
Évaluer les pratiques en matière d’inviolabilité dans les départements de pharmacie hospitalière en Colombie-Britannique et explorer les possibilités de standardisation pour réduire le gaspillage de médicaments.
Méthodologie
Des visites des lieux ont été réalisées dans 13 hôpitaux répartis sur 3 autorités sanitaires en Colombie-Britannique – Vancouver Coastal Health, Providence Health Care et Fraser Health – afin de collecter des données sur les pratiques en matière d’inviolabilité dans les départements de pharmacie hospitalière.
Résultats
Les méthodes d’inviolabilité variaient d’un département à l’autre. Les principales recommandations d’amélioration comprennent la mise en place d’autocollants ou d’autres témoins d’intégrité, la standardisation du positionnement de ces témoins d’intégrité, la priorisation des médicaments spécifiques aux patients pour l’inviolabilité, ainsi que l’élaboration et le respect de politiques/procédures pour la plupart des médicaments multidoses avant leur sortie de la pharmacie.
Conclusions
La normalisation des pratiques d’inviolabilité peut réduire le gaspillage de médicaments et l’impact environnemental, avec un potentiel d’une adoption plus large dans les hôpitaux.
Mots-clés: inviolabilité, gaspillage des médicaments, médicaments multidoses, standardisation
The Canadian health care sector is a significant contributor to the nation’s greenhouse gas emissions, accounting for approximately 4.6% of the country’s total emissions.1 Within this sector, medications—both prescribed and nonprescribed—represent the largest category in terms of emissions, contributing 25% of total health care–related greenhouse gases, with hospitals following closely behind.2 As the climate crisis intensifies, there is increasing urgency to implement strategies to reduce these emissions, which pose a growing threat to human health.3
Medication waste in hospitals is a global issue that negatively affects the environment and increases health care costs.4 A previous survey describing medication recycling patterns in Canadian hospitals found that 30%–50% of unused medications were not reused or redispensed for new patients.5 Multidose medications, such as eye drops and inhalers, are particularly problematic because of their high cost and the larger quantity of medication per unit relative to single-use items. If a multidose item is dispensed for a patient but not actually used, the medication can be sent home with the patient, which prevents waste, but this is not always appropriate. Furthermore, hospital supply does not meet community dispensing criteria,6 yet some patients are nonetheless sent home with multidose medications that should have been returned to the pharmacy. Unused medication can be returned to stock for reuse in a new patient, provided pharmacy staff are assured of its integrity. Implementing tamper-evident features for multidose products, such as an adhesive sticker covering the opening, could provide a practical solution, enabling easy identification and potential recirculation of unused medication for other patients. With this in mind, we sought to gain a better understanding of the medication tamper-proofing process in pharmacy departments in hospitals in the Lower Mainland of British Columbia.
The primary objective of this study was to determine when and how tamper-proofing was occurring in our large hospital system. The secondary objective was to identify opportunities to improve and standardize the ways in which tamper-proofing could be used to reduce medication waste.
Our goal was to understand the tamper-proofing processes and procedures at most acute care hospital sites within Vancouver Coastal Health, Providence Health, and Fraser Health (13 hospitals) located in the southwestern portion of British Columbia. A set of questions was developed by a team of medication-use evaluation pharmacists (the authors), all of whom had previous experience working in all aspects of hospital pharmacy practice, such as dispensary operations, medication safety, and clinical pharmacy services. This wide range of experience meant that the team could understand and interpret survey responses from pharmacy staff working in these various areas. Pharmacy students gathered information about the sites’ procedures for tamper-proofing multidose medications (e.g., inhalers, eye drops, topical agents); workflow procedures, including who is responsible for tamper-proofing; the presence of existing tamper-proofing guidelines; and how medications are returned to the pharmacy from nursing units. Answers to our questions were gathered during interview-type interactions. More specifically, pharmacy coordinators and pharmacy technician supervisors at the various sites were contacted by email to set up in-person or virtual visits in June and July 2024. Virtual visits were used when distance precluded in-person visits. The questionnaire (Appendix 1) was circulated ahead of the site visit. The team felt that the pharmacy coordinators and pharmacy technician supervisors would have direct knowledge of their departments’ respective policies and procedures, but if not, they would reach out to staff members before providing responses to the questions.
At the outset of each site visit, the questions were discussed with the pharmacy coordinator or pharmacy technician supervisor. During the visits, we recorded information using a standard data collection form. Afterward, we asked to observe the work area to see the supplies used for tamper-proofing and took photos of these observations. Each visit lasted about 45 minutes. After each hospital site visit, we went over the notes to add any missing details and organized the photos into one document per site. Another document was created that served as a summary of all the sites visited. This document included sections to note what each site was doing well in terms of tamper-proofing, what was not being done optimally, and our ideas for ways to improve. Our team reviewed the observations from all sites visited to produce a list of recommendations based on informal group discussion leading to consensus. The recommendations were not based on tamper-proofing methods previously known to be useful or practical, as we were unable to find any literature on the subject of tamper-proofing to reduce medication waste. Rather, our recommendations were based on human factors principles, such as the hierarchy of effectiveness.7
A total of 13 sites were visited. Two of these visits were virtual due to distance, whereas the other 11 visits were in person. Of the sites visited, 4 hospitals were tertiary care institutions, 6 were urban community hospitals, and the remaining 3 were rural hospitals.
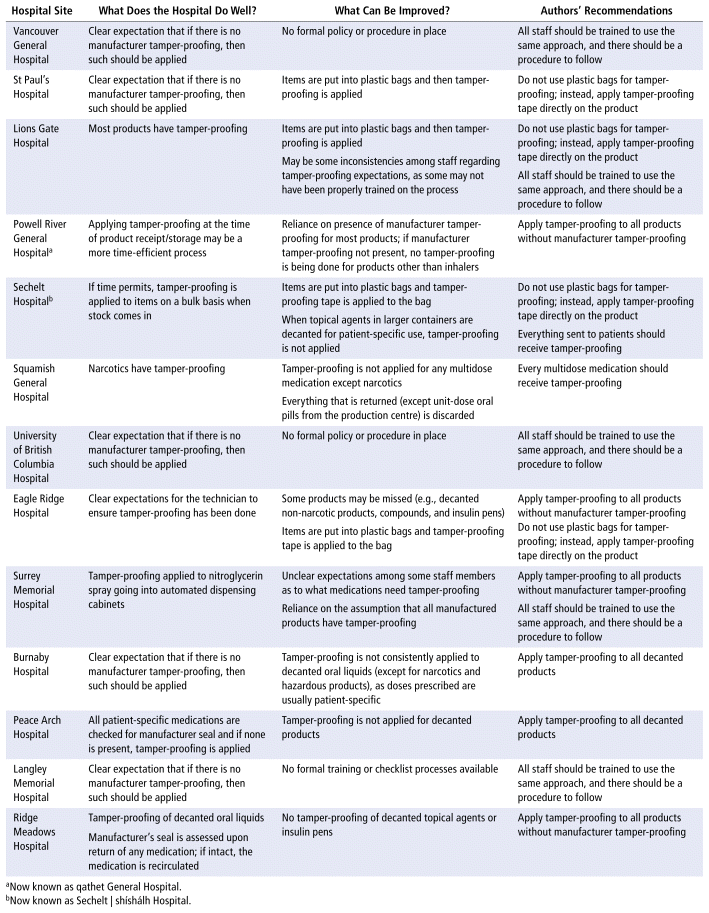
Tamper-proofing practices were highly variable across the 13 sites (Table 1). None of the sites visited had any protocol or policy in place for tamper-proofing. However, 12 of the 13 sites applied tamper-proofing to most multidose products. Four sites made extensive use of plastic bags for tamper-proofing (e.g., placing a bottle of eye drops into a plastic zipper bag).
TABLE 1 Summary of Site Visits

Using the information we learned from all sites, our team produced a set of recommendations (Table 2), which was sorted into recommendations that could be implemented immediately (high impact, low effort) and those that might take more time and effort (high impact, high effort).
The purpose of this study was to better understand the tamper-proofing procedures at hospitals in our region to identify gaps and areas for improvement to reduce medication waste. We could not find any similar evaluations of tamper-proofing from other jurisdictions. Our previous survey of Canadian hospital pharmacies showed that tamper-proofing of multidose medications was not universal.5 According to those findings, approximately 1 in 5 hospital pharmacies did not tamper-proof anything, only 1 in 5 sites used tamper-proofing for inhalers, and just over half of the pharmacies applied tamper-proofing to cream and eye drop bottles.5 Although most of the hospitals in our current study applied tamper-proofing for most multidose medications, we found inconsistencies across sites, including missed opportunities for tamper-proofing. Furthermore, there were opportunities to optimize and/or standardize this process, including when during the dispensing process the tamper-proof marker (adhesive sticker or plastic zipper bag) should be applied and where the sticker should be applied on each product.
A problem faced by many hospital pharmacies is short staffing, which may contribute to challenges in implementing tamper-proofing practices. Initially, our team discussed whether all items being sent from the pharmacy, both those specifically for the patient and those to be stocked in automated dispensing cabinets (ADCs), should be tamper-proofed. In particular, we debated whether tamper-proofing should be employed for medications stored in ADCs, given that nurses might only remove multidose medications from the ADC when they are needed, so such medications would frequently be opened and used and thus would generate very little waste. If so, the effort required to tamper-proof medications destined for ADCs may not be worthwhile. Nonetheless, we ultimately opted to recommend that all multidose medications be tamper-proofed. We felt that tamper-proofing is a low-effort intervention, regardless of the storage location, and it has the potential for substantial impact. Furthermore, this approach standardizes practice and expectations for the health care team. Additionally, nursing practice is not necessarily the same across all sites, and some nurses may proactively access multidose products from the ADCs.
Our study had 2 main limitations. Without a baseline understanding of the amount of medication waste associated with unused multidose containers, it is difficult to determine the amount of waste that could be reduced by tamper-proofing. Additionally, we only evaluated the pharmacy side of the process; the handling of medications by nurses could be a topic for further study to identify additional opportunities for tamper-proofing.
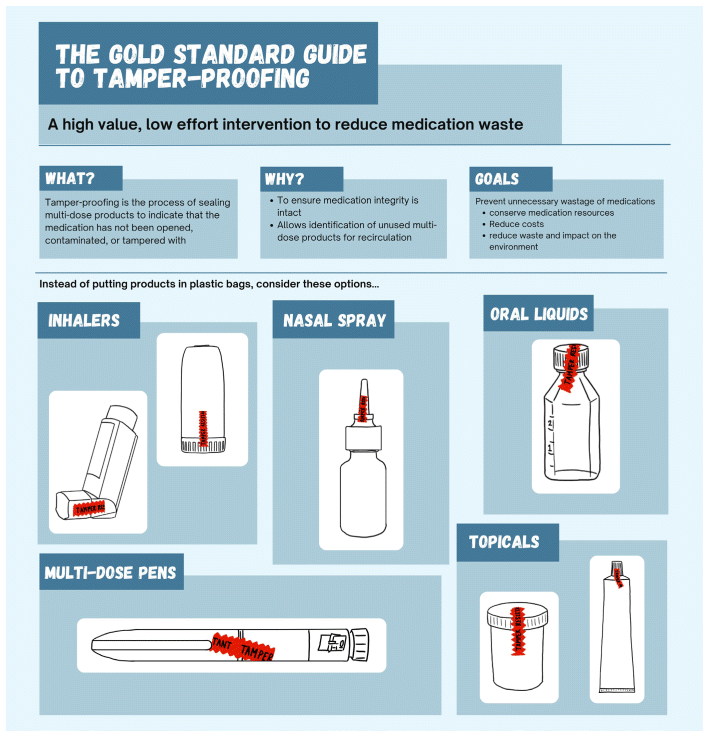
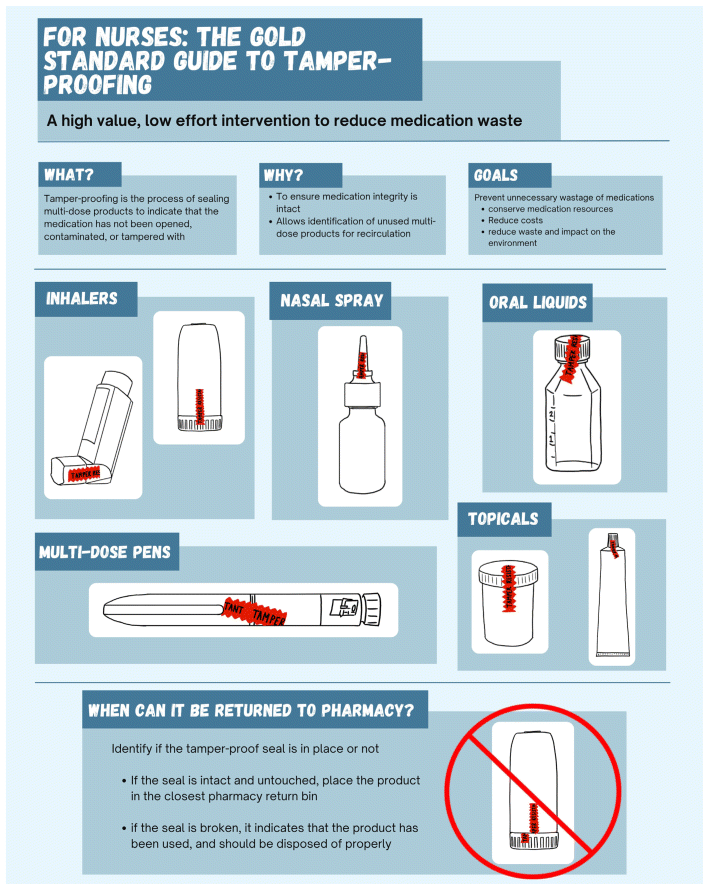
The next step of our project is to implement the high-impact, low-effort recommendations (Table 2) because they are simple and can be started immediately. Longer-term plans include developing a policy and procedure to standardize the tamper-proofing process within pharmacy departments to ensure consistency across hospital sites. In addition, we would like to collaborate with our nursing colleagues to develop educational initiatives and policies and procedures for nurses that focus on their role in broader efforts to reduce medication waste, specifically, strategies for multidose medications and tamper-proofing awareness. We have created an infographic for pharmacy and nursing staff that shows how to tamper-proof specific product types to ensure consistency (Appendix 2). We also plan to evaluate whether standardized tamper-proofing policies, procedures, and education lead to a reduction in medication waste. The evaluation will be 2-fold: first, we will assess the degree to which pharmacy staff adhere to tamper-proofing all multidose medications listed in the policy, and second, we will determine the quantity of unused multidose medications that are returned by nursing staff to the pharmacy department. For the latter evaluation, we will be able to see which patients had orders for multidose medications that were tamper-proofed.
The results of our project indicate that a consistent, standardized approach to tamper-proofing medications could have a high impact on reducing the number of multidose medications that must be discarded. From the small sample of Canadian hospital pharmacy departments we visited, it is clear there is an opportunity to incorporate a standardized protocol to reduce inconsistencies and overall waste. We encourage other hospitals to review their tamper-proofing and medication recycling policies, in hopes of a meaningful step toward reducing the impact that health care has on our environment.
1 Vogel L. Canada’s health system is among the least green. CMAJ. 2019; 191(48):E1342–3.
Crossref PubMed PMC
2 Eckelman MJ, Sherman JD, MacNeill AJ. Life cycle environmental emissions and health damages from the Canadian healthcare system: an economic-environmental-epidemiological analysis. PLoS Med. 2018; 15(7):e1002623.
Crossref PubMed PMC
3 Born KB, Levinson W, Vaux E. Choosing Wisely and the climate crisis: a role for clinicians. BMJ Qual Saf. 2024;33(3):200–4.
Crossref
4 Peltoniemi T, Suomi R. Eliminating medicine waste in a Finnish university hospital — a qualitative study. J Pharm Policy Pract. 2019;12(1):27.
Crossref
5 Zou B, Sung S, Drummond I, Tang L, Tejani AM. Understanding medication recycling practices in Canadian hospitals. Int J Pharm Pract. 2024;32(4):311–5.
Crossref PubMed
6 Schedule “F” – standards of practice. Part 1 – Community pharmacy standards of practice. In: Health Professions Act – bylaws. College of Pharmacists of British Columbia; 2020 Jun 19 [cited 2024 Jun 1]. Available from: https://library.bcpharmacists.org/6_Resources/6-1_Provincial_Legislation/5076-HPA_Bylaws.pdf
7 ISMP Canada. Designing effective recommendations. Ont Crit Incident Learn. 2013 [cited 2024 Jun 1];4:1–2. Available from: https://www.ismp-canada.org/download/ocil/ISMPCONCIL2013-4_EffectiveRecommendations.pdf
Address correspondence to: Dr Aaron M Tejani, Lower Mainland Pharmacy Services (Fraser Health), Langley Fulfillment Centre, 8521 198A Street, Langley BC V2Y 0A1, email: aaron.tejani@fraserhealth.ca
Competing interests: For activities not directly related to this study, Aaron Tejani has received honoraria for presentations given on behalf of the Therapeutics Initiative and for lectures to students in the University of British Columbia Faculty of Medicine, as well as payment for expert testimony. No other competing interests were declared.
Funding: None received.
Submitted: November 12, 2024
Accepted: May 12, 2025
Published: August 13, 2025
Is there a tamper-proofing checklist for technicians to use?
Is everyone in the pharmacy trained?
Is there a formal policy/procedure regarding tamper-proofing?
Where can this be found?
When does tamper-proofing occur?
How often?
Who is responsible for tamper-proofing?
What is the process for tamper-proofing?
How do tamper-proofed medications get returned?
Are medications checked for tamper-proofing integrity?
Are there products that have short shelf lives after being decanted?
Is tamper-proofing worthwhile?
Are there issues with fitting tamper-proofed medications into automated dispensing cabinets? For example, are inhalers placed in plastic zipper bags, which are then given tamper-proofing?
Is there an audit process for tamper-proofing?
If yes, how often is this done?
What is the process?
Who is responsible?
How are new products that may require tamper-proofing treated? (How does the policy treat new products?)
Is there a list that is updated?
Who decides what gets tamper-proofed?
© 2024 Lower Mainland Pharmacy Services. Reproduced by permission.
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 3, 2025