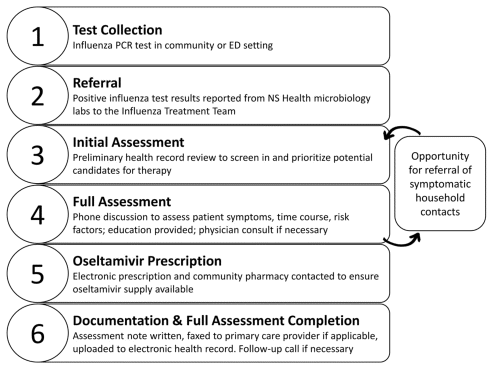
FIGURE 1 Workflow of Influenza Treatment Team. ED = emergency department, NS = Nova Scotia, PCR = polymerase chain reaction.
Emma K Reid, Sarah Opie, Lisa Nodwell, Shelly A McNeil, and Tasha D RamseyTo cite: Reid EK, Opie S, Nodwell L, McNeil SA, Ramsey TD. Nova Scotia Health Influenza Treatment Team: virtual care pathway to antiviral treatment. Can J Hosp Pharm. 2025;78(3):3760. doi: 10.4212/cjhp.3760
Influenza is 1 of the top 10 leading causes of death in Canada.1 Early antiviral therapy with oseltamivir can lessen the burden of influenza illness by reducing symptom duration and complications. The Association of Medical Microbiology and Infectious Disease Canada recommends that antiviral treatment, including oseltamivir, be initiated as soon as possible for adults and for children age 1 year or older who are at risk for complications of influenza, including admission to hospital.2,3
During the 2023/24 acute respiratory virus season, Nova Scotians experiencing respiratory symptoms could self-refer to receive multiplex polymerase chain reaction (PCR) testing for COVID-19, influenza A and B, and respiratory syncytial virus. All patients with a positive result on COVID-19 PCR testing were automatically referred to the Nova Scotia Health COVID-19 Non-severe Therapy Consult Service. However, Nova Scotians without a primary care provider lacked a mechanism to receive their influenza diagnosis, and the risk was high that positive results on influenza testing would go unaddressed.
To support this new self-referral PCR testing program and to reduce strain on local emergency departments, a pilot NS Health Influenza Treatment Team virtual care program was launched to offer oseltamivir treatment to a subset of high-risk outpatients who tested positive for influenza. This pilot program ran simultaneously with the NS Health COVID-19 Non-severe Therapy Consult Service.4
The NS Health Influenza Treatment Team pilot, a strategic initiative of the NS Health Emerging and Re-emerging Infections Network, launched in January 2024, in the middle of the 2023/24 respiratory virus season, and continued until the influenza season was over, in July 2024. The members of the Influenza Treatment Team were NS Health pharmacists (including E.K.R., S.O., and T.D.R.), who worked collaboratively with an on-call physician (S.A.M).
A prescribing protocol (available upon request to the corresponding author) was created to support independent pharmacist prescribing of oseltamivir to nonhospitalized patients with influenza. To qualify for pharmacist-prescribed oseltamivir, patients had to have mild, uncomplicated influenza symptoms that started in the past 5 days (or up to 7 days in some cases) and had to have at least 1 high-risk factor for influenza complications, such as older age (65 years or older) or underlying respiratory disease. Although the product monograph specifies that oseltamivir initiation should occur within 2 days of the onset of influenza symptoms,5 our team prescribed the medication up to 5 days from symptom onset because both the Association of Medical Microbiology and Infectious Disease Canada and the Canadian Paediatric Society recommend initiation of antiviral therapy beyond 2 days if the individual belongs to a group at high risk for severe disease; if the illness is progressive, severe, or complicated; or if the patient requires hospitalization.3,6 We additionally accepted a window of symptoms up to 7 days in duration to allow for prescribing in immunocompromised or otherwise vulnerable individuals who might conceivably have prolonged viral replication. The full list of high-risk criteria in the prescribing protocol reflected the guidance outlined by the Association of Medical Microbiology and Infectious Disease Canada.3 For patients with progressive or complicated symptoms, or if the possibility of postviral bacterial pneumonia was raised, the team contacted the on-call physician to determine an appropriate referral and/or prescribing pathway.
Positive results for influenza PCR tests completed at NS Health microbiology laboratories were provided to the team of pharmacists and electronically populated into a protected folder 4 times a day. The electronic health records for patients with a positive test result were briefly reviewed (initial assessment) for circumstances warranting further assessment (full assessment), which included a phone conversation with the patient or caregiver (Figure 1). Of these laboratory-referred patients, outpatients age 1 year or older who either self-referred for PCR testing without a documented primary care provider or who were tested for influenza in a NS Health emergency department were eligible for a full assessment. Patients for whom oseltamivir was already prescribed, as documented in the NS Drug Information System, were excluded, as were those residing in a long-term care facility, because within Nova Scotia, these facilities have an established process for managing influenza outbreaks.
|
| ||
|
FIGURE 1 Workflow of Influenza Treatment Team. ED = emergency department, NS = Nova Scotia, PCR = polymerase chain reaction. | ||
Assessments were prioritized according to criteria such as age over 65 years and other documented risk factors. A team member made up to 3 attempts over 2 days to reach the patient by telephone; if these efforts were unsuccessful, the patient was considered lost to follow-up. The phone assessment allowed the Influenza Treatment Team pharmacist to assess the patient’s symptoms and their time course, confirm risk factors, discuss the role of oseltamivir, and provide education. Patients with at least 1 risk factor for complications and persistent influenza symptoms but still within 7 days of onset were offered oseltamivir treatment. If the patient agreed, the Influenza Treatment Team pharmacist prescribed oseltamivir electronically and contacted the patient’s community pharmacy to ensure supply was available. Documentation of the assessment was uploaded as a progress note in the patient’s electronic health record and, if applicable, was sent by fax to their primary care provider. A follow-up phone call with the patient was completed on a case-by-case basis.
Occasionally, symptomatic household contacts of influenza-positive patients were assessed for oseltamivir therapy if requested by the initially referred patient. In these cases, a negative result on COVID-19 rapid antigen testing was required before oseltamivir could be prescribed.
Internal tracking of referrals and assessments was documented in shared Excel spreadsheets (Microsoft Corporation), organized by the date of test referral and accessed on a protected drive. Between 1 and 4 pharmacists were scheduled each day, 7 days a week, from 0800 to 1600, to support the Influenza Treatment Team, often alongside other clinical responsibilities. Evening coverage (1600 to 1900) was available occasionally. Over the course of the pilot, influenza test referral volumes ranged from 0 to 67 referrals per day.
During the pilot period (January 23 to July 27, 2024), 1823 patients underwent initial assessment by the team (Figure 2). Fourteen of the referrals were household contacts, and all others were referred from the laboratory. Of these 1823 patients, 1133 were screened in for full assessment, and 690 (37.8%) were excluded. The most common reasons for exclusion were pre-existing prescription for oseltamivir (n = 187, 27.1%), residence in a long-term care facility (n = 178, 25.8%), and inpatient status (n = 144, 20.9%).
|
| ||
|
FIGURE 2 Patient referral and assessment pathway. ED = emergency department, EHR = electronic health record, LTCF = long-term care facility, NS = Nova Scotia, PCR = polymerase chain reaction. | ||
Full assessments were completed for 924 (81.6%) of the 1133 patients screened in for a full assessment. Of the 209 patients for whom a full assessment was not completed, 166 (79.4%) were lost to follow-up because they could not be reached by phone.
Among the patients with a full assessment, the mean age was 34 (standard deviation 24.4) years, with 14.1% age 65 years or older (Table 1). Most (85.8%) of the referred patients were tested in the emergency department setting.
TABLE 1 Characteristics of Patients Who Tested Positive for Influenza and Underwent Full Assessment for Potential Antiviral Therapy
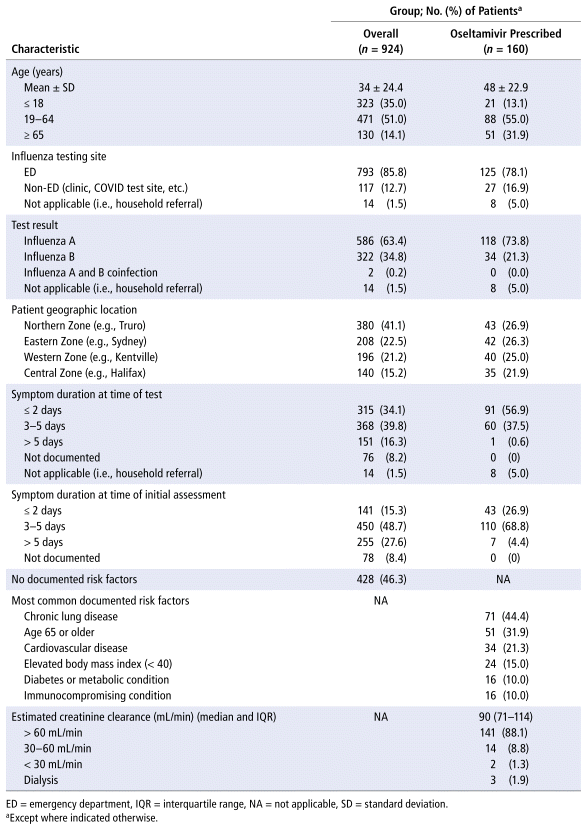
Oseltamivir was prescribed for 160 (17.3%) of the fully assessed patients. Approximately one-third of prescriptions were for those age 65 years of age or older (31.9%). Other risk factors commonly identified among oseltamivir recipients were chronic lung disease (44.4%) and cardiovascular disease (21.3%). Recorded risk factors and comorbidities are summarized in Table 1, although the overall prevalences may be underestimated, given that the assessing pharmacist only had to confirm the presence of 1 risk factor before proceeding with the rest of the assessment. Comprehensive information on risk factors and renal function for the fully assessed population is not reportable, as such information was not necessarily ascertained or documented for all patients who otherwise did not meet the eligibility criteria (e.g., those whose symptoms were much improved at the time of the phone assessment). A lack of risk factors was, however, documented in nearly half of the fully assessed patients (46.3%). Among patients for whom oseltamivir was prescribed, the distribution was relatively even across the 4 NS Health zones: 26.9% in the Northern Zone, 26.3% in the Eastern Zone, 25.0% in the Western Zone, and 21.9% in the Central Zone.
Among patients with a full assessment, the median time from referral to completion of the assessment, including documentation, was 8 (interquartile range [IQR] 4.4–23.0) hours. Among the 160 patients for whom oseltamivir was prescribed, the median time from referral to prescription was 5.5 (IQR 3.0–17.7) hours.
Physician on-call consultation was required for 6 (0.6%) of the fully assessed patients. Follow-up phone calls, for the purpose of monitoring symptoms and providing patient reassurance, were completed for 19 (2.1%) of the fully assessed patients. All of these follow-up phone calls were suggested by the assessing pharmacist after consultation with the patient and were completed by an Influenza Treatment Team pharmacist.
This pilot initiative was successful in reaching a high proportion of outpatients within 12 hours after a positive result on PCR testing for influenza. Nova Scotians living in rural and urban areas received timely, telephone-based care and were able to access oseltamivir from a community pharmacy of their choice. A comprehensive prescribing protocol supported independent pharmacist prescribing in most cases, which can feasibly be replicated by other Canadian pharmacist-led teams, thus offloading physician, other prescriber, and emergency department resources. Our team is aware of community pharmacist-led initiatives in other Canadian jurisdictions for prescribing influenza antivirals, which, to our understanding, require the patient to present to their community pharmacy for assessment.7–10 The automatic referral model, whereby all patients with influenza-positive PCR tests performed through NS Health provincial laboratories were referred to our dedicated Influenza Treatment Team, may offer the advantages of timelier follow-up and prescribing, as well as convenience for the patient. As is the case in many jurisdictions in Canada, a significant number of Nova Scotians do not have a regular primary care provider, so programs such as this, which optimize the scope of practice for pharmacists, can fill an important gap in the management of influenza. The efficiency of prescribing by the Influenza Treatment Team aligns with evidence-based recommendations to initiate influenza therapy as soon as possible after symptoms appear, to optimize the efficacy of antivirals.3
The approach to patient assessment in this pilot was broad, with most patients being screened in for telephone assessment, even those of younger age and those without clear, documented risk factors for influenza complications. Our test-referred patient population was relatively young, and many had no identified risk factors. The overall rate of oseltamivir prescribing was accordingly low, at less than 20%, although the yield was higher among those age 65 or older. This broad screening approach was taken with recognition that some risk factors warranting oseltamivir treatment, such as elevated body mass index, may not be routinely available in the electronic health record and would need to be ascertained through discussion with the patient. Also, many fully assessed patients had been experiencing symptoms for a duration beyond a reasonable window for initiating antiviral therapy or had sufficiently improved influenza symptoms by the time of the phone assessment. The duration and severity of influenza symptoms also required confirmation through a discussion with the patient. Efficiencies could be built into this process if more patient information were made available alongside the positive influenza test result, which would allow more comprehensive screening of high-risk patients warranting full pharmacist assessment. Targeted testing of only high-risk individuals is another strategy to streamline necessary follow-up.
The workflow was relatively rudimentary and could be replicated by other institutions; however, there are opportunities for improvement. Moving forward, incorporating a self-referral process to allow reporting of high-risk criteria and symptom duration, similar to the COVID-19 Non-severe Therapy Consult Service self-referral form used in Nova Scotia, and using automation to support patient triage would optimize prioritization and workflow. Continued prioritization of older individuals will help to ensure efficient resource utilization, with recognition that it is also important to initiate treatment for patients younger than 65 years who have medical comorbidities that increase the risk of influenza complications. Expansion of this pilot to include pharmacist prescribing for patients with influenza in long-term care facilities and inpatient settings has the potential to improve efficiency in initiating appropriate antiviral therapy and thus to improve influenza outcomes for patients in these high-risk settings.
1 Flu (influenza): for health professionals. Government of Canada, Public Health Agency of Canada; 2024 [cited 2024 Dec 8]. Available from: https://www.canada.ca/en/public-health/services/diseases/flu-influenza/health-professionals.html
2 Harrison R, Mubareka S, Papenburg J, Schober T, Allen UD, Hatchette TF, et al. AMMI Canada 2023 update on influenza: management and emerging issues. J Assoc Med Microbiol Infect Dis Can. 2023;8(3):176–85.
PubMed PMC
3 Aoki FY, Allen UD, Mubareka S, Papenburg J, Stiver HG, Evans GA. Use of antiviral drugs for seasonal influenza: foundation document for practitioners—update 2019. J Assoc Med Microbiol Infect Dis Can. 2019; 4(2):60–82.
PubMed PMC
4 Ramsey TD, d’Entremont-Harris M, Nodwell L, Goodall B, Barrett L. Nova Scotia Health COVID-19 Non-severe Therapy Consult Service: lessons from a hospital pharmacy–based model. Can J Hosp Pharm. 2024;77(3):e3578. doi:10.4212/cjhp.3578
Crossref PMC
5 Tamiflu [product monograph]. Hoffmann-La Roche Limited; 2022 Sep 9.
6 Allen UD; Canadian Paediatric Society, Infectious Diseases and Immunization Committee. The use of antiviral drugs for influenza: guidance for practitioners. Canadian Paediatric Society; 2018 Oct 9 [reaffirmed 2024 Jan 11, cited 2025 Apr 24]. Available from: https://cps.ca/documents/position/antiviral-drugs-for-influenza
7 Standards of practice for pharmacists and pharmacy technicians. Alberta College of Pharmacy; 2025 [cited 2025 Apr 24]. Available from: https://abpharmacy.ca/wp-content/uploads/Standards_SPPPT.pdf
8 Community pharmacy practice enactments. Saskatchewan College of Pharmacy Professionals; 2025 [cited 2025 Apr 24]. Available from: https://saskpharm.ca/document/5894/Community_Pharmacy_Practice_Enactments.pdf
9 Pharmacist prescribing: initiating, adapting and renewing prescriptions [guideline]. Version 7.00. Ontario College of Pharmacists; 2024 [cited 2025 Apr 24]. Available from: https://www.ocpinfo.com/regulations-standards/practice-policies-guidelines/adaptations-renewing-prescriptions/
10 Questions de practique. Ordre des pharmaciens du Québec; 2025 [cited 2025 Apr 24]. Available from: https://www.opq.org/pratique-professionnelle/questions-de-pratique/
Address correspondence to: Dr Emma K Reid, Pharmacy Department, Nova Scotia Health, 1276 South Park Street, Halifax NS B3H 2Y9, email: emmak.reid@nshealth.ca
Competing interests: For activities not directly related to the project described here, Tasha Ramsey has served in a volunteer capacity with the Nova Scotia Emerging and Re-emerging Infections Therapeutics and Prophylactics Recommendation Group (Co-chair) and the CSHP Education Committee. No other competing interests were declared.
Funding: None received.
Acknowledgement: The authors acknowledge Gregory Kerry for his assistance with the statistical analyses.
Submitted: December 16, 2024
Accepted: May 12, 2025
Published: August 13, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 3, 2025