Shengyu Yu, Stéphanie Tremblay, Léna Coïc, Cécile Bérard, Philippe-Alexandre Martineau-Cyr, Jean-Marc Forest, Josée Chagnon, Philippe Ovetchkine, and Bruce TapiéroTo cite: Yu S, Tremblay S, Coïc L, Bérard C, Martineau-Cyr PA, Forest JM, et al. Effectiveness, tolerance, and safety of meropenem in a pediatric outpatient parenteral antimicrobial therapy program. Can J Hosp Pharm. 2025;78(3):e3772. doi: 10.4212/cjhp.3772
ABSTRACT
Background
Outpatient parenteral antimicrobial therapy (OPAT) is often used for patients who need long-term antimicrobial therapy. Meropenem is a broad-spectrum antibiotic used to treat polymicrobial and multidrug-resistant bacterial infections.
Objective
To evaluate the efficacy, tolerance, and safety of IV meropenem for pediatric patients in the OPAT program at CHU Sainte-Justine in Montréal, Quebec.
Methods
At the study institution, meropenem solutions (1–40 mg/mL) are prepared in polyvinylchloride bags or cassettes. Each delivery device contains one daily dose, and infusions (at room temperature) take place every 8 hours. Devices are delivered to the patient’s home in batches every 3 or 4 days. With refrigeration, the drug solution has a short period of stability (96 hours). This single-centre retrospective study included all patients under 18 years of age who received IV meropenem therapy as part of the OPAT program between April 2000 and April 2024. For the current analysis, demographic data, clinical data, serum alanine aminotransferase levels, and white blood cell counts were collected using a standardized template.
Results
A total of 349 courses of therapy (for 262 patients) met the inclusion criteria. For most patients, the treatments were successful and well tolerated, with an overall success rate of 93.4% (326/349). Few gastrointestinal symptoms and rashes were reported. Neutropenia and eosinophilia were more frequently observed than in previous studies. OPAT-related adverse events were mainly catheter-related (8.9%, 31/349) or pump-related (6.3%, 22/349) issues.
Conclusions
These results suggest that IV meropenem can be safely and effectively used in pediatric OPAT, although the target concentration of 90% may not be retained, according to current stability data.
Keywords: meropenem, outpatient parenteral antimicrobial therapy, safety, pediatric infections, antibiotics
RÉSUMÉ
Contexte
L’antibiothérapie intraveineuse (IV) à domicile (ABAD) est souvent utilisée chez les patients qui ont besoin d’antibiothérapie à long terme. Le méropénem est un antibiotique à large spectre utilisé pour traiter les infections polymicrobiennes et multirésistantes.
Objectif
Évaluer l’efficacité, la tolérance et l’innocuité du méropénem IV chez les patients pédiatriques du programme ABAD du CHU Sainte-Justine, à Montréal (Québec).
Méthodologie
Au CHU Saint-Justine, le méropénem, à une concentration variant de 1 à 40 mg/mL, est conservé dans un sac en PVC ou une cassette. Chaque dispositif d’administration contient une dose quotidienne et les perfusions (à température ambiante) ont lieu toutes les 8 heures. Les dispositifs sont livrés par lots tous les 3 à 4 jours au domicile des patients. Le méropénem dans ces dispositifs a une courte durée de conservation de 96 heures au réfrigérateur. Cette étude rétrospective monocentrique a inclus tous les patients de moins de 18 ans ayant reçu du méropénem intraveineux, dans le cadre du programme ABAD, entre avril 2000 et avril 2024. Pour cette analyse, les données démographiques, les données cliniques, les taux d’alanine aminotransférase sérique et la numération des globules blancs ont été recueillis à l’aide d’un formulaire standardisé.
Résultats
Au total, 349 traitements (administrés à 262 patients) ont été inclus dans l’étude. Ils ont été efficaces et bien tolérés chez la plupart des patients, avec un succès global de 93,4 % (326/349). Peu de symptômes gastro-intestinaux et d’éruptions cutanées ont été rapportés. La neutropénie et l’éosinophilie ont été observées plus fréquemment que dans les études précédentes. Les effets indésirables liés à l’ABAD concernaient principalement des problèmes liés au cathéter (8,9 %, 31/349) et à l’utilisation de la pompe (6,3 %, 22/349).
Conclusions
Ces résultats semblent indiquer que le méropénem IV est efficace et peut être utilisé en toute sécurité dans l’ABAD pédiatrique, bien que la concentration cible de 90 % puisse ne pas être atteinte dans tous les cas selon les données de stabilité actuelles.
Mots-clés: méropénem, antibiothérapie intraveineuse à domicile, sécurité, infections pédiatriques, antibiotiques
Outpatient parenteral antimicrobial therapy (OPAT) is an effective alternative to hospital admission for patients who need long-term parenteral antimicrobial therapy.1 Meropenem is a broad-spectrum antibiotic used to treat polymicrobial and multidrug-resistant bacterial infections.2 Data regarding meropenem solutions diluted to 22 mg/mL indicated that the solution remained stable for up to 17 hours at room temperature,3 although stability has been variable across studies. Fawaz and others4 determined that concentration declined to 90% of the initial value after 7.4 hours at room temperature and to 71.2% after 20 hours. In contrast, Berthoin and others5 observed more than 10% degradation after about 12 hours at room temperature, with the extent of degradation depending on the initial concentration. These findings suggest that concentration and storage temperature have a significant impact on stability. The variability of stability across studies is worth highlighting, as is its concentration-dependency (whereby less concentrated solutions remain stable for longer3–5) and probably temperature-dependency (whereby cooler temperatures yield better stability, although maintaining lower temperatures would require the use of ice packs in the ambulatory setting, which could substantially reduce the temperature of the infusion, thus inconveniencing the patient, and could also weigh down the ambulatory system).
Where a single-dose elastomeric pump is used, the problem of reduced stability does not arise, because the dose can be refrigerated until the time of administration. To decrease the manipulations required of patients or their parents (in the pediatric setting), to diminish the risk of contaminating the peripherally inserted central catheter (PICC) line, and to maintain venous patency, a pump is used to deliver the antibiotic over 24 hours. In this setting, the decline in stability of meropenem at room temperature could be an obstacle to use of this drug in OPAT, given that antibiotics remain unrefrigerated over a period of 24 hours.
This study aimed to review clinical experience with meropenem, administered with an infusion pump, in the pediatric OPAT program at the CHU Sainte-Justine, Montréal, Quebec, over a period of 24 years.
This single-centre retrospective study included all patients under 18 years of age who received IV meropenem through the OPAT program at the CHU Sainte-Justine between April 2000 and April 2024. Patients were identified from the pharmacy department database. Demographic data (gender and age), clinical data (primary diagnosis, number of meropenem doses administered, and clinical outcomes), serum alanine aminotransferase levels (ALT), and white blood cell counts were collected using a standardized template.
Meropenem has been used in the CHU Sainte-Justine OPAT program since 2000 and is administered at home through a PICC line, using a programmable ambulatory infusion pump (computerized ambulatory delivery device) containing a 24-hour antibiotic dose. For patients receiving meropenem, the choice was determined by a pediatric infectious diseases specialist, based mainly on the microbiological organisms identified (i.e., multidrug-resistant microorganisms), patients’ allergy to β lactam antibiotics, and lack of any other therapeutic option.
For OPAT administration, meropenem, at a concentration ranging from 1 to 40 mg/mL, is prepared in a polyvinylchloride bag or a cassette; the choice of bag or cassette depends on the desired dose in relation to the available cassettes (capacities of 50, 100, or 250 mL) or bag (when desired dose is over 250 mL). Each device contains a single daily dose, and the infusions take place at room temperature every 8 hours, for a period of 30 minutes to 6 hours. Devices are delivered to the patient’s home in batches every 3 or 4 days. With refrigeration, the drug solution has a short period of stability (96 hours). Patients are followed weekly by nurses and pediatric infectious diseases physicians in a specialized clinic at the CHU Sainte-Justine. Evolution of the patient’s clinical status, central line–related dysfunction, results of blood tests, and adverse events related to antibiotic therapy are systematically reported in the patient’s medical record.
This study was retrospective in nature, so approval was granted by the institution’s Department of Medical and Academic Affairs, rather than the ethics committee.
OPAT success was defined as complete resolution of the infectious episode on an outpatient basis, without relapse in the subsequent 30 days.
OPAT failure was defined as an unscheduled readmission or change in antimicrobial therapy, after lack of improvement.
The following OPAT-related adverse events were collected: catheter- or pump-related issues, gastrointestinal symptoms, rash, fever, and seizure. Abnormalities of blood work were defined according to the following predetermined parameters:
Hepatotoxicity was defined as a 2-fold elevation in ALT over the patient’s baseline level (defined as the most recent ALT value before initiation of meropenem).
Eosinophilia was defined as absolute eosinophil count above 0.4 × 109/L, and significant eosinophilia as an absolute eosinophil count above 1.0 × 109/L.
Neutropenia was defined as absolute neutrophil count below 1.5 × 109/L, and profound neutrophil count below 0.5 × 109/L.
Descriptive statistics are expressed as means with standard deviation (SD) and medians with range. Proportions were compared using the χ2 test or the Fischer exact test as appropriate. Statistical significance was defined by a p value less than 0.05.
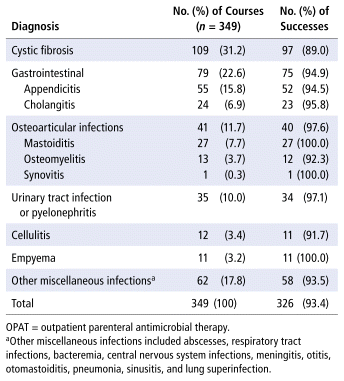
Over the 24-year study period, 379 treatments of IV meropenem were delivered as part of the CHU Sainte-Justine OPAT program. Of these, 30 were excluded from the analysis because the patients were 18 years of age or older (n = 20), were being treated in a rehabilitation centre (n = 6), or were admitted to hospital (n = 4). The remaining 349 treatments were delivered to 262 patients eligible for inclusion in this study (of whom 181 or 69.1% were girls), whose mean age was 9.0 (SD 5.64) years. Meropenem was used for various infections, as detailed in Table 1.
TABLE 1 Clinical Effectiveness of Meropenem in 349 OPAT Courses at CHU Sainte-Justine, April 2000 to April 2024

The mean dosage of meropenem was 92.2 (SD 28.5) mg/kg daily divided into 3 doses, with a mean treatment duration of 18.8 (SD 16.5) days (median 6.5 days). The drug concentration ranged from 6.7 to 40 mg/mL. Meropenem was used as monotherapy in 217 treatments (62.2%). Of the 132 treatments involving meropenem combined with other antibiotics, 100 (75.8%) were used for cystic fibrosis. Tobramycin was the most frequently used antibiotic in combination with meropenem (23.5%, 31/132), followed by ceftazidime (19.7%, 26/132), vancomycin (18.2%. 24/132), and trimethoprim–sulfamethoxazole (16.7%, 22/132).
OPAT success rates, classified by diagnosis, appear in Table 1. Overall, the treatment was successful in 93.4% of cases. The success rate was higher when meropenem was used as monotherapy than when used in combination with other antibiotics (96.8% [210/217] vs 87.9% [116/132]; p = 0.003). Treatment failure did not always result in readmission to hospital (9.7%, 34/349); sometimes, there was a change in the outpatient antimicrobial treatment.
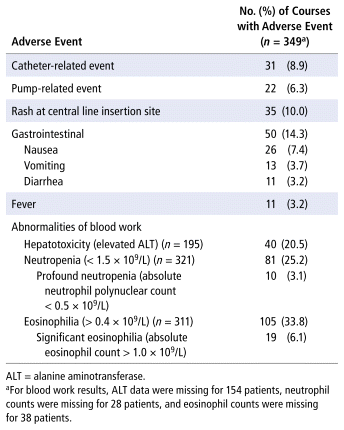
OPAT-related adverse events, detailed in Table 2, were mainly catheter-related (8.9%, 31/349) and pump-related (6.3%, 22/349) issues. The predominant clinical adverse effects were gastrointestinal symptoms, which occurred in 50 (14.3%) cases. Rashes at the site of the central line were reported in 35 cases. Fever occurred in 11 cases, but no seizures were observed. Neutropenia and profound neutropenia were observed in 81 and 10 cases, respectively. Of the 81 patients with neutropenia, 29 had neutropenia before starting meropenem. Eosinophilia and significant eosinophilia occurred in 105 and 19 cases, respectively. Of the 105 patients with eosinophilia, 41 had eosinophilia before starting meropenem. A 2-fold elevation of ALT over baseline was observed in 40 cases. About a third of these 40 cases (n = 12) occurred in patients with appendicitis, 9 in patients being treated for cystic fibrosis, 2 in patients with cholangitis, and 17 in patients with other infections. ALT level, neutrophil count, and eosinophil count during treatments were unavailable for 154, 28, and 38 cases, respectively, possibly because these patients had shorter duration of treatment. The occurrence of elevated hepatic enzymes in a large proportion of the patients may have been attributable to underlying chronic liver disease, including cystic fibrosis and cholangitis secondary to biliary atresia, or to appendicitis.
TABLE 2 Clinical and Blood Work Adverse Events Observed during 349 Courses of Meropenem Therapy at CHU Sainte-Justine, April 2000 to April 2024
When administered by infusion, preparations of meropenem must have at least 90% of their initial concentration to be considered stable.3–5 A recent study showed that at 25°C, meropenem solutions at a concentration of 2 mg/mL delivered with portable elastomeric infusion pumps retained drug content above 90% of the initial amount for 24 hours; for 25 mg/mL solutions, the period of stability was 8 hours.6 Despite uncertainties regarding the stability of meropenem in OPAT, the overall success rate of 93.4% in the current study is consistent with previously reported effectiveness of meropenem in hospitalized children (84%–96%).2 The results are also consistent with the success rates reported for other pediatric OPAT programs, which have ranged between 87% and 96%.1,7,8 The rate of unscheduled readmission to hospital was lower than previously reported for pediatric OPAT (9.7% vs 10%–28%).7–9
Catheter-related issues were reported less often than in previous pediatric OPAT studies (8.9% vs 25%, respectively).10,11 Conversely, rashes were reported more frequently than in previous research (10.0% vs 5%–12%, respectively)7,9; this adverse effect could be attributable to localized contact dermatitis related to the catheter dressing. Nausea was more commonly reported than in previous studies of meropenem in the treatment of hospitalized children (7.4% vs < 2%, respectively).10 The frequency of gastrointestinal symptoms (14.3%) and fever (3.2%) in the current study aligned with previous reports (2.5%–6%).2,9
The frequency of elevation of hepatic enzymes (specifically ALT) was comparable to what has been reported in studies of other antibiotics used in OPAT (20.5% vs 4%–22%, respectively).8,9,12 Meropenem is not known to significantly affect liver enzymes, including ALT.13 As a precautionary measure, and given that patients were receiving treatment at home, an alert threshold of 2-fold ALT elevation was used.
Neutropenia was more frequently observed than in previous studies of pediatric OPAT (25.2% vs 13%, respectively).7 For 2 of the cases in our study, neutropenia might have influenced the treatment outcome, leading to therapeutic failure. This complication is commonly reported in OPAT with other β lactam antibiotics (5%–13%).7,9 The higher incidence of neutropenia in the current study could be explained by patients having other underlying diseases or receiving concomitant medications that would increase their risk. A quick review of the literature did not reveal an obvious difference in the rate of neutropenia for meropenem compared with other β lactams.
Eosinophilia was also more frequently observed here than in previous studies of adult OPAT programs with meropenem (33.8% vs 8%, respectively).11 One case of drug reaction with eosinophilia and systemic symptoms (also known as DRESS) was reported, and this affected the patient’s clinical course. The eosinophilia findings have several possible causes. For example, in pediatrics, meropenem dosage is adjusted for weight, and children receive a relatively higher dose than adults. Also, the duration of treatment in OPAT is longer than for inpatients, and abnormalities of blood work in patients treated with IV meropenem have been reported mainly for hospital inpatients. In the current study, the mean duration of treatment for patients with eosinophilia was 16.8 (SD 9.2) days. Toxic degradation of the antibiotic solution might also explain the onset of blood work abnormalities, although this relation has yet to be confirmed.11 Finally, other underlying diseases or concomitant medications could increase the risk of eosinophilia.
To our knowledge, this is the largest study to date of a pediatric cohort treated with IV meropenem in an OPAT program. However, the study had a retrospective design, and the events reported in the medical record could be incomplete despite weekly follow-ups and the limited number of treating physicians. Moreover, the patients might have had multiple comorbidities and medications that could affect the laboratory results. Published studies do not always define laboratory parameters precisely, which represents a barrier to comparisons with previous results. Furthermore, the absence of a control group prevents comparisons with patients treated with another regimen. It will be interesting to see whether our results will be repeated in future analyses of data from other centres.
Results from the current study suggest that IV meropenem can be safely and effectively used in pediatric OPAT, although the target concentration of 90% may not be retained, according to currently available stability data. Treatments were successful for, and well tolerated by, most of the patients. Weekly follow-up to monitor the clinical tolerance profile and the occurrence of abnormalities of blood work (neutropenia, eosinophilia, and elevated ALT) should be recommended.
1 Chapman ALN, Dixon S, Andrews D, Lillie PJ, Bazaz R, Patchett JD. Clinical efficacy and cost-effectiveness of outpatient parenteral antibiotic therapy (OPAT): a UK perspective. J Antimicrob Chemother. 2009;64(6):1316–24.
Crossref PubMed
2 Cohen-Wolkowiez M, Poindexter B, Bidegain M, Weitkamp JH, Schelonka RL, Randolph DA, et al.; Meropenem Study Team. Safety and effectiveness of meropenem in infants with suspected or complicated intra-abdominal infections. Clin Infect Dis. 2012;55(11):1495–502.
Crossref PMC
3 Walker SE, Varrin S, Yannicelli D, Law S. Stability of meropenem in saline and dextrose solutions and compatibility with potassium chloride. Can J Hosp Pharm. 1998;51(4):156–68.
4 Fawaz S, Barton S, Whitney L, Swinden J, Nabhani-Gebara N. Stability of meropenem after reconstituion for administration by prolonged infusion. Hosp Pharm. 2019;54(3):190–6.
Crossref PMC
5 Berthoin K, Le Duff CS, Marchand-Brynaert J, Carryn S, Tulkens PM. Stability of meropenem and doripenem solutions for administration by continuous infusion. J Antimicrob Chemother. 2010;65(5):1073–5.
Crossref PubMed
6 Esteban-Cartelle B, Serrano DR, Pérez Menendez-Conde C, Vicente-Oliveros N, Álvarez-Díaz A, Fortún Abete J, et al. Stability of meropenem in portable elastomeric infusion devices: which protocol should be implemented in clinical practice? Microbiol Spectr. 2024;12:e0206423.
Crossref
7 Gomez M, Maraqa N, Alvarez A, Rathore M. Complications of outpatient parenteral antibiotic therapy in childhood. Pediatr Infect Dis J. 2001;20(5):541–3.
Crossref
8 Madigan T, Banerjee R. Characteristics and outcomes of outpatient parenteral antimicrobial therapy at an academic children’s hospital. Pediatr Infect Dis J. 2013;32(4):346–9.
Crossref
9 Fernandes P, Milliren C, Mahoney-West HM, Schwartz L, Lachenauer CS, Nakamura MM. Safety of outpatient parenteral antimicrobial therapy in children. Pediatr Infect Dis J. 2018;37(2):157–63.
Crossref
10 Le J, San Agustin M, Hernandez EA, Tran TT, Adler-Shohet FC. Complications associated with outpatient parenteral antibiotic therapy in children. Clin Pediatr (Phila). 2010;49(11):1038–43.
Crossref PubMed
11 Manning L, Wright C, Ingram PR, Whitmore TJ, Heath CH, Manson I, et al. Continuous infusions of meropenem in ambulatory care: clinical efficacy, safety and stability. PLoS One. 2014;9(7):e102023.
Crossref PubMed PMC
12 Hornik CP, Herring AH, Benjamin DK Jr, Capparelli EV, Kearns GL, van den Anker J, et al. Adverse events associated with meropenem versus imipenem/cilastatin therapy in a large retrospective cohort of hospitalized infants. Pediatr Infect Dis J. 2013;32(7):748–53.
Crossref
13 Severity grading in drug induced liver injury. In: LiverTox: clinical and research information on drug-induced liver injury. National Institute of Diabetes and Digestive and Kidney Diseases; 2012 [updated 2019 May 4; cited 2024 Nov 10]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK548241/
Address correspondence to: Jean-Marc Forest, Pharmacy Department, CHU Sainte-Justine, 3175, chemin de la Côte Sainte-Catherine, Montréal QC H3T 1C5, email: jean-marc.forest.hsj@ssss.gouv.qc.ca
Funding: This work was supported by the Department of Pharmacy, CHU Sainte-Justine.
Submitted: January 12, 2025
Accepted: March 31, 2025
Published: July 9, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 3, 2025