Caitlin Chew, Roxane Carr, Chelsea Elwood, and Vanessa PaquetteTo cite: Chew C, Carr R, Elwood C, Paquette V. Safety of cefazolin in pregnant patients with documented penicillin allergy: a retrospective cohort study. Can J Hosp Pharm. 2025;78(4):e3774. doi: 10.4212/cjhp.3774
ABSTRACT
Background
The antibiotic cefazolin is commonly used in pregnancy, as first-line prophylaxis for cesarean section wounds or as an alternative to penicillin for prophylaxis against group B Streptococcus. About 10% of people report a penicillin allergy, and clinicians have historically avoided the use of β-lactams (including cefazolin) in these individuals. Instead, they have used non-β-lactams, which can lead to poorer maternal outcomes.
Objective
To evaluate the safety of cefazolin in pregnant patients with a documented history of penicillin allergy.
Methods
This retrospective cohort study included all pregnant patients with a documented penicillin allergy at the time of receiving their first dose of cefazolin at a large tertiary care hospital (January 2016 to August 2021). Descriptive statistics were calculated.
Results
A total of 179 patients were included in the analysis. Most (175 [97.8%]) had no allergic adverse event after receiving cefazolin. Two patients (1.1%) experienced immunoglobulin E (IgE)–mediated hives, and 2 patients (1.1%) experienced non–IgE-mediated rashes. No patients experienced anaphylaxis, and no patients with a documented history of anaphylaxis to penicillins experienced an allergic adverse event related to cefazolin. All 4 patients who experienced an allergic adverse event were discharged with no readmission to the study institution associated with the allergic adverse event.
Conclusion
No patients with a documented history of anaphylaxis to penicillin experienced an allergic adverse event upon receiving cefazolin. Cefazolin was safely given to pregnant people with a history of penicillin allergy.
KEYWORDS: cefazolin, obstetrics, pregnancy, penicillin allergy, β-lactam allergy
RÉSUMÉ
Contexte
La céfazoline est un antibiotique couramment utilisé pendant la grossesse comme prophylaxie de première intention pour les plaies opératoires de césarienne ou comme solution de rechange à la pénicilline pour la prophylaxie contre le streptocoque du groupe B. Environ 10 % des personnes déclarent être allergiques à la pénicilline et les cliniciens ont par le passé évité l’utilisation des β-lactamines (y compris la céfazoline) chez ces patients, préférant des antibiotiques non β-lactamines, ce qui peut entraîner des résultats maternels moins favorables.
Objectif
Évaluer l’innocuité de la céfazoline chez les patientes enceintes ayant des antécédents attestés d’allergie à la pénicilline.
Méthodologie
Cette étude de cohorte rétrospective incluait toutes les patientes enceintes ayant une allergie attestée à la pénicilline au moment de recevoir la première administration de céfazoline dans un grand hôpital tertiaire (de janvier 2016 à août 2021). Des statistiques descriptives ont été calculées.
Résultats
Un total de 179 patientes ont été incluses dans l’analyse. La plupart (175 [97,8 %]) n’avaient connu aucun antécédent d’événement indésirable allergique lié à la céfazoline. Deux patientes (1,1 %) ont eu de l’urticaire médiée par les immunoglobulines E (IgE), et des éruptions non médiées par les IgE se sont produites chez deux autres (1,1 %). Aucune patiente n’a connu d’anaphylaxie, et aucune ayant des antécédents attestés d’anaphylaxie aux pénicillines n’a connu d’événement allergique lié à la céfazoline. Les 4 patientes ayant connu un événement indésirable allergique ont quitté l’établissement étudié lié à cet événement sans y être réadmises.
Conclusion
Aucune patiente ayant des antécédents attestés d’anaphylaxie à la pénicilline n’a vécu d’événement indésirable allergique après avoir reçu de la céfazoline. La céfazoline a été administrée en toute sécurité aux personnes enceintes ayant une allergie à la pénicilline.
Mots-clés: céfazoline, obstétrique, grossesse, allergie à la pénicilline, allergie aux β-lactamines
Cefazolin is commonly prescribed during pregnancy for the treatment of infections, for surgical prophylaxis related to cesarean section, and as an alternative to penicillin for prophylaxis against group B Streptococcus (GBS). Penicillin allergy is one of the most commonly reported drug allergies, with up to 10% of individuals, including pregnant people, reporting a history of an allergic reaction.1–3 Historically, clinicians have avoided β-lactam antibiotics, including cefazolin, in penicillin-allergic patients and instead have used non-β-lactam antibiotics (e.g., clindamycin, vancomycin, or erythromycin) because of concerns regarding cross-reactivity. The use of non-β-lactam alternatives, such as clindamycin, has been associated with poorer maternal outcomes, including increased frequency of wound infections, increased length of hospital stay, and more adverse events.4,5
Historically, cross-reactivity between penicillins and cephalosporins, involving type I allergies (immunoglobulin E [IgE]–mediated), was attributed to the β-lactam ring; however, more recent data have suggested that cross-reactivity is associated with similarities in the side chains of the drug molecules.6,7 Cefazolin does not share any similar side chains with penicillins (or any β-lactam antibiotic); therefore, even patients with severe type I penicillin allergies (e.g., anaphylaxis) can safely receive cefazolin.7–12
A growing body of literature reports the safe use of cefazolin in patients with type I penicillin allergies. However, despite the data available from the nonpregnant population, there is a dearth of evidence describing the safe use of cefazolin in the pregnant population.13,14 Although there is no literature suggesting a difference in safety for the pregnant population, current guidelines from both the Society of Obstetricians and Gynaecologists of Canada and the American College of Obstetricians and Gynecologists recommend against the use of cefazolin in high-risk penicillin-allergic patients—defined as patients with a history of anaphylaxis, angioedema, respiratory distress, or significant urticaria—recommending instead the use of less effective non-β-lactam antibiotics.15–18
Extrapolating from the literature pertaining to nonpregnant patients (and anticipating no difference in safety due to pregnancy), our institution, a large tertiary care hospital in British Columbia, decided in November 2018 to use cefazolin in pregnant patients with documented penicillin allergies (including anaphylaxis) when it was indicated as first-line therapy or as an alternative to penicillin. At the time of our study, the impact of this practice change had not yet been assessed.
In this study, we aimed to assess this practice change and also address the gap in the literature by contributing relevant data from the pregnant population. The primary objective was to describe the safety of cefazolin in pregnant patients with a documented history of penicillin allergy. The secondary objectives were to describe the prevalence of allergic adverse events related to cefazolin in penicillin-allergic patients, to describe the outcomes for any patients who experienced an allergic adverse event after receiving cefazolin, and to describe nonallergic adverse drug events (ADEs) associated with cefazolin.
A retrospective cohort study was conducted. All pregnant patients with a documented penicillin allergy at the time of receiving their first dose of cefazolin at the study hospital, from January 1, 2016, to August 31, 2021, were included in the analysis. Patients were identified from the pharmacy database on the basis of documented allergy history and a prescription for cefazolin. Data were collected by a single investigator (C.C.) using a standardized electronic data collection form within the Research Electronic Data Capture (REDCap) tool.19 Quality assurance, to ensure the accuracy and integrity of data collection, was based on duplicate data extraction by a second investigator, working independently, for 10% of the health records. The association between cefazolin and nonallergic ADEs was assessed with the Naranjo scoring tool, where a score of 0 or below indicates a doubtful association, a score of 1 to 4 indicates a possible association, a score of 5 to 8 indicates a probable association, and a score of 9 or above indicates a definite association.20 All events with a Naranjo score of 1 or above were reported.
This study was approved by the institution’s research ethics board. The research was conducted in accordance with the ethical standards of the research ethics board and the Helsinki Declaration.
The following definitions were applied in this study.
Penicillin allergy was defined as an immunologically mediated response to a penicillin in a sensitized person.1 Signs and symptoms that characterize a penicillin allergy include the following21,22:
Anaphylaxis was defined by the occurrence of any one of the following 3 sets of criteria21:
Length of stay was defined as the time from administration of cefazolin to discharge (in hours). If the patient experienced a hypersensitivity reaction to cefazolin, the time from reaction to discharge was also collected.
Safe tolerability of cefazolin was defined, in the context of this study, as administration of cefazolin to pregnant people with documented penicillin allergy and no subsequent documented allergic adverse event.
Descriptive statistics were calculated, including the mean and standard deviation or median and range for continuous variables. Statistical analysis was performed using Excel spreadsheet software, version 16.82 (Microsoft Corporation).
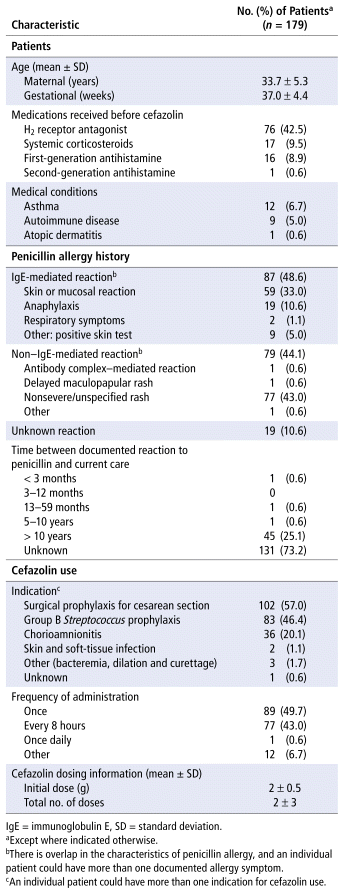
In total, 179 patients were included in the analysis (Table 1). Of the documented histories of penicillin allergy, most patients had a non–IgE-mediated, nonsevere/unspecified rash (43.0%). Nineteen patients (10.6%) had a documented history of anaphylaxis to penicillin, and 9 patients (5.0%) had a documented history of a positive skin test with penicillin. For most patients (73.2%), the timing of their penicillin allergic reaction was not documented; however, of the 48 patients with documentation of timing, most (n = 45, 93.8%) reported a childhood penicillin allergy.
TABLE 1 Characteristics of Patients, Prior Reactions, and Cefazolin Therapy

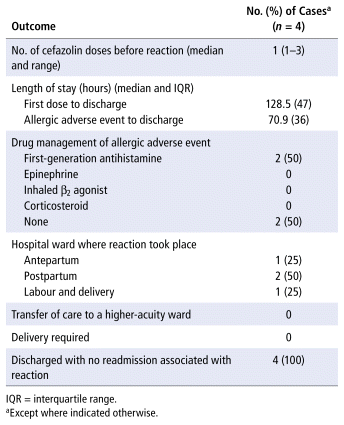
Most patients (175 [97.8%]) had no allergic adverse event after receiving cefazolin (Figure 1). No patients experienced anaphylaxis to cefazolin. Two patients experienced IgE-mediated hives, which were managed with a first-generation antihistamine. Two patients experienced mild non–IgE-mediated rashes. One of these patients experienced a delayed-onset (> 72 hours) rash after a single dose of cefazolin, which resolved with no drug therapy. The other patient experienced a red rash on one arm approximately 36 hours after receiving a single dose of cefazolin, which resolved without drug therapy. Of these 4 patients who experienced an allergic adverse event after receiving cefazolin, 2 patients (50%) had an unknown history of penicillin allergy, 1 patient (25%) had a documented history of hives after receiving penicillin, and 1 patient (25%) had a documented history of non–IgE-mediated rash. No patients with a documented history of anaphylaxis to penicillin experienced an allergic adverse event after receiving cefazolin. All 4 patients were discharged with no readmission to our institution associated with the allergic adverse event (Table 2).
|
| ||
|
FIGURE 1 Cefazolin safety outcomes (n = 179), presented as the proportion of pregnant patients with a history of penicillin allergy who safely tolerated cefazolin versus the proportion who experienced an allergic adverse event related to cefazolin. Among those who experienced an allergic adverse event, 2 (1.1%) experienced hives mediated by immunoglobulin E (IgE), and 2 (1.1%) experienced non–IgE-mediated rashes. | ||
TABLE 2 Outcomes of Allergic Adverse Events Related to Cefazolin
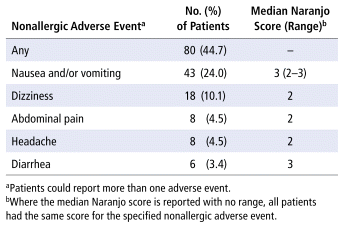
Nonallergic ADEs associated with cefazolin were frequent (Table 3). For all of the reported nonallergic ADEs, the associated Naranjo score was 3 or below, indicating a possible association between cefazolin and the adverse event. Of the 80 patients who experienced nonallergic ADEs, 7 (8.8%) reported isolated subjective pruritus during their admission, and the median Naranjo score for the association between subjective pruritus and cefazolin was 1. Of these 7 patients, 6 had received an epidural during their admission. A Naranjo score to characterize the association between the epidural and subjective pruritus was also calculated; the median Naranjo score for this association was 2 (range 2–3).
TABLE 3 Nonallergic Adverse Events Associated with Cefazolin (n = 179)
Most pregnant people with a documented history of penicillin allergy, including anaphylaxis, safely tolerated cefazolin with no allergic adverse event. Four patients experienced allergic adverse events after receiving cefazolin, all of which were mild reactions requiring minimal intervention. No patients experienced anaphylaxis secondary to cefazolin. There is an abundance of research characterizing the safe use of cefazolin in penicillin-allergic patients, specifically in the nonpregnant population. There are no data to suggest any differences in safety of cefazolin in the pregnant population. However, to our knowledge, this is the first study to characterize the safe use of cefazolin in the pregnant population regardless of the severity of the previously documented penicillin allergy.7,9,10,12
In this study, we included all patients with a documented history of any penicillin allergy, including a history of positive skin test or anaphylaxis. In a previous retrospective cohort study, published in 2011, Critchfield and others13 reported that non-β-lactam antibiotics were prescribed for GBS prophylaxis in 76% of patients with high-risk penicillin allergy, in accordance with national guidelines. However, the study sample included no patients with a history of anaphylaxis who received cefazolin. Another retrospective cohort study, published in 2016, described the safety of cefazolin for GBS prophylaxis in non-anaphylactic penicillin-allergic pregnant patients.14 Although the authors concluded that cephalosporins were underutilized in this population, with 44.2% of their patient population receiving cefazolin or penicillin, they did not describe the rate of allergic adverse events within this population or the safety of cefazolin in high-risk penicillin-allergic patients.14 Current guidelines of the Society of Obstetricians and Gynaecologists of Canada and the American College of Obstetricians and Gynecologists recommend against the use of cefazolin for high-risk penicillin-allergic pregnant patients.15–18 In our study, 10.6% of patients had a documented history of anaphylaxis and 5.0% had a documented history of a positive skin test with penicillin, all of whom were able to safely tolerate cefazolin. No patients with a documented history of anaphylaxis to penicillin had an allergic adverse event after receiving cefazolin.
Almost half of the patients received an H2 receptor antagonist before cefazolin, and others received first- or second-generation antihistamines or systemic corticosteroids. These medications were given as part of standard antenatal care, not as premedication or for the prevention of allergic adverse events. There is a lack of data to support the use of these drug therapies as premedications to prevent severe allergic adverse events, because they are not effective in preventing anaphylaxis and would not have masked the onset of a severe allergic adverse event in our study.21,23 Although these medications may be used to alleviate symptoms of urticaria or pruritus during an allergic adverse event or for the management of mild cutaneous symptoms, they are likely to contribute to delay of first-line therapies and are ineffective for the acute management of anaphylaxis or severe adverse allergic events.21,24,25
This study also described nonallergic ADEs and their association with cefazolin. In particular, we found that 44.7% of patients may have had nonallergic ADEs related to cefazolin. Previous retrospective studies examining cefazolin exposure in penicillin-allergic pregnant patients did not report nonallergic ADEs or their association with cefazolin exposure.13,14 For all events reported in our study, the associated Naranjo score was 3 or below, indicating a possible association between the event and exposure to cefazolin. A possible association is present when the reaction follows a temporal sequence after administration of a drug and may follow a recognized pattern for the suspected drug; however, there could be another equally likely explanation for the event and/or there is uncertainty or lack of information.20 We took a conservative approach and reported any adverse event; however, in doing so, we may have reported some adverse events with low probability of being associated with cefazolin. In general, cefazolin is a well-tolerated antibiotic with minimal adverse effects.26 The high proportion of nonallergic ADEs reported in our study could also be secondary to the patients’ labour and delivery. Nausea and/or vomiting, dizziness, abdominal pain, headache, and diarrhea are commonly reported events that may be associated with labour and delivery, postpartum complications, and other concomitant medications given antepartum.27,28
Among the 80 patients in this study who experienced nonallergic ADEs, almost 10% also experienced subjective pruritus during their admission. Subjective pruritus or neuraxial opioid-induced pruritus is a commonly reported adverse event that occurs secondary to an epidural and is reported in 60% to 100% of patients.27 Of the 7 patients in our study who experienced subjective pruritus, 6 had received an epidural during their admission. All 6 of these patients were treated effectively with nalbuphine, an opioid antagonist used for the management of epidural-induced subjective pruritus.29 The median Naranjo score for the association between subjective pruritus and epidural was higher than the Naranjo score for the association between subjective pruritus and cefazolin (2 vs. 1). It is unlikely that these cases of subjective pruritus were allergic adverse events related to cefazolin because they were characterized by pruritus alone, with no associated skin, mucosal, respiratory, gastrointestinal, or systemic allergic signs and symptoms. Nalbuphine is ineffective for the management of an allergic adverse event.
The limitations of our study include its retrospective nature and the small sample size. More specifically, the study population included only a limited number of patients with documented history of anaphylaxis to penicillin. Additionally, the nature of the penicillin allergy and detailed allergy histories were not consistently documented in patients’ medical records, and those that were documented were likely based on patient reporting and hence were not verifiable. We included all patients with unknown reactions to penicillin, as well as patients with documented nonspecific, unspecified rashes related to penicillin. This approach may have led to an overestimation of the number of true penicillin allergies captured within this patient population. Patients with unknown or childhood penicillin allergies were also included. By 5 years after the occurrence of an allergic event, approximately 50% of people will outgrow their penicillin allergy, and at 10 years, this proportion increases to 80%.1,30,31 Despite these limitations, we demonstrated the safe use of cefazolin among patients with a documented history of anaphylaxis to penicillin.
Another limitation was that this study captured only encounters at our own institution. Patients may have already been exposed to and tolerated cefazolin at other sites before admission to our institution. Also, we reviewed only readmissions to our institution related to allergic adverse events to cefazolin; patients who experienced delayed allergic adverse events related to cefazolin after discharge from our institution and were admitted to another institution would not have been captured in this study. Lastly, we were unable to assess neonatal outcomes for patients who had an allergic adverse event related to cefazolin; however, none of the allergic adverse events resulted in delivery, and all were considered mild, requiring minimal to no intervention.
Most pregnant people with a documented history of penicillin allergy who received cefazolin tolerated the drug with no subsequent allergic adverse event. None of the patients with a documented history of anaphylaxis to penicillin experienced an allergic adverse event related to cefazolin. Cefazolin may safely be given to pregnant people with a history of penicillin allergy.
1 Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105(4):259–73.
Crossref PubMed
2 Idsoe O, Guthe T, Willcox RR, de Weck AL. Nature and extent of penicillin side-reactions, with particular reference to fatalities from anaphylactic shock. Bull World Health Organ. 1968;38(2):159–88.
PubMed PMC
3 Solensky R. Hypersensitivity reactions to beta-lactam antibiotics. Clin Rev Allergy Immunol. 2003;24(3):201–19.
Crossref PubMed
4 Harris BS, Hopkins MK, Villers MS, Weber JM, Pieper C, Grotegut CA, et al. Efficacy of non-beta-lactam antibiotics for prevention of cesarean delivery surgical site infections. AJP Rep. 2019;9(2):e167–71.
Crossref PubMed PMC
5 Desai SH, Kaplan MS, Chen Q, Macy EM. Morbidity in pregnant women associated with unverified penicillin allergies, antibiotic use, and group B Streptococcus infections. Perm J. 2017;21:16–080.
Crossref PubMed PMC
6 Blanca M, Vega JM, Garcia J, Miranda A, Carmona MJ, Juarez C, et al. New aspects of allergic reactions to betalactams: crossreactions and unique specificities. Clin Exp Allergy. 1994;24(5):407–15.
Crossref PubMed
7 Miranda A, Blanca M, Vega JM, Moreno F, García JJ, Segurado E, et al. Cross-reactivity between a penicillin and a cephalosporin with the same side chain. J Allergy Clin Immunol. 1996;98(3):671–7.
Crossref PubMed
8 Trubiano JA, Stone CA, Grayson ML, Urbancic K, Slavin MA, Thursky KA, et al. The 3 Cs of antibiotic allergy—classification, cross-reactivity, and collaboration. J Allergy Clin Immunol Pract. 2017;5(6):1532–42.
Crossref PubMed PMC
9 Novalbos A, Sastre J, Cuesta J, De Las Heras M, Lluch-Bernal M, Bombín C, et al. Lack of allergic cross-reactivity to cephalosporins among patients allergic to penicillins. Clin Exp Allergy. 2001;31(3):438–43.
Crossref PubMed
10 Blanca M, Fernandez J, Miranda A, Terrados S, Torres MJ, Vega JM, et al. Cross-reactivity between penicillins and cephalosporins: clinical and immunologic studies. J Allergy Clin Immunol. 1989;83(2 Pt 1): 381–5.
Crossref PubMed
11 Sousa-Pinto B, Blumenthal KG, Courtney L, Mancini CM, Jeffres MN. Assessment of the frequency of dual allergy to penicillins and cefazolin: a systematic review and meta-analysis. JAMA Surg. 2021;156(4):e210021.
Crossref PubMed PMC
12 Campagna JD, Bond MC, Schabelman E, Hayes BD. The use of cephalosporins in penicillin-allergic patients: a literature review. J Emerg Med. 2012;42(5):612–20.
Crossref
13 Critchfield AS, Lievense SP, Raker CA, Matteson KA. Group B Streptococcus prophylaxis in patients who report a penicillin allergy: a follow-up study. Am J Obstet Gynecol. 2011;204(2):150.e1.
Crossref
14 Briody VA, Albright CM, Has P, Hughes BL. Use of cefazolin for group B streptococci prophylaxis in women reporting a penicillin allergy without anaphylaxis. Obstet Gynecol. 2016;127(3):577–83.
Crossref PubMed
15 Money D, Allen VM. The prevention of early-onset neonatal group B streptococcal disease. J Obstet Gynaecol Can. 2016;38(12 Suppl): S326–35.
Crossref
16 American College of Obstetricians and Gynecologists. Prevention of group B streptococcal early-onset disease in newborns. ACOG Committee Opinion Summary No. 782. Obstet Gynecol. 2019;134(1):1.
PubMed
17 American College of Obstetricians and Gynecologists, Committee on Practice Bulletins—Obstetrics. ACOG Practice Bulletin No. 199: Use of prophylactic antibiotics in labor and delivery. Obstet Gynecol. 2018;132(3):e103–19.
Crossref PubMed
18 van Schalkwyk J, Van Eyk N. No. 247–Antibiotic prophylaxis in obstetric procedures [practice guideline]. J Obstet Gynaecol Can. 2017; 39(9):e293–9.
Crossref PubMed
19 Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–81.
Crossref PMC
20 Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239–45.
Crossref PubMed
21 Shaker MS, Wallace DV, Golden DBK, Oppenheimer J, Bernstein JA, Campbell RL, et al. Anaphylaxis—a 2020 practice parameter update, systematic review, and Grading of Recommendations, Assessment, Development and Evaluation (GRADE) analysis. J Allergy Clin Immunol. 2020;145(4):1082–123.
Crossref PubMed
22 Coombs PR, Gell PG. Classification of allergic reactions responsible for clinical hypersensitivity and disease. In: Gell RR, editor. Clinical aspects of immunology. Oxford University Press; 1968. pp. 579–96.
23 Nurmatov UB, Rhatigan E, Simons FER, Sheikh A. H2-antihistamines for the treatment of anaphylaxis with and without shock: a systematic review. Ann Allergy Asthma Immunol. 2014;112(2):126–31.
Crossref PubMed
24 Sheikh A, Ten Broek V, Brown SGA, Simons FER. H1-antihistamines for the treatment of anaphylaxis: Cochrane systematic review. Allergy. 2007;62(8):830–7.
Crossref PubMed
25 Vanden Bussche G, Emanuel MB, Rombaut N. Clinical profile of astemizole. A survey of 50 double-blind trials. Ann Allergy. 1987;58(3): 184–8.
PubMed
26 Thompson JW, Jacobs RF. Adverse effects of newer cephalosporins. Drug Saf. 1993;9(2):132–42.
Crossref PubMed
27 Szarvas S, Harmon D, Murphy D. Neuraxial opioid-induced pruritus: a review. J Clin Anesth. 2003;15(3):234–9.
Crossref PubMed
28 Brousseau EC, Danilack V, Cai F, Matteson KA. Emergency department visits for postpartum complications. J Womens Health. 2017;27(3): 253–7.
Crossref
29 Jannuzzi RG. Nalbuphine for treatment of opioid-induced pruritus: a systematic review of literature. Clin J Pain. 2016;32(1):87–93.
Crossref
30 Blanca M, Torres MJ, García JJ, Romano A, Mayorga C, de Ramon E, et al. Natural evolution of skin test sensitivity in patients allergic to β-lactam antibiotics. J Allergy Clin Immunol. 1999;103(5):918–24.
Crossref PubMed
31 Sullivan TJ, Wedner HJ, Shatz GS, Yecies LD, Parker CW. Skin testing to detect penicillin allergy. J Allergy Clin Immunol. 1981;68(3):171–80.
Crossref PubMed
Address correspondence to: Dr Vanessa Paquette, Department of Pharmacy, Children’s & Women’s Health Centre of British Columbia, Lower Mainland Pharmacy Services, 4500 Oak Street, Room 0B7, Vancouver BC V6H 3N1, email: vpaquette@phsa.ca
Competing interests: For activities unrelated to the study reported here, Chelsea Elwood has received grants from the Public Health Agency of Canada and the Canadian Institutes of Health Research, and has also received payment from the College of Physicians and Surgeons of British Columbia for expert testimony. No other competing interests were declared.
Funding: None received.
Submitted: January 16, 2025
Accepted: June 29, 2025
Published: October 8, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 4, 2025