Shane Pawluk, Clara Jo Hoban, and Blair SeifertTo cite: Pawluk S, Hoban CJ, Seifert B. Role of pharmacy professionals in gene therapy based on adeno-associated viruses: treatment of hemophilia as a template of care. Can J Hosp Pharm. 2025;78(4):e3795. doi: 10.4212/cjhp.3795
ABSTRACT
Background
Gene therapy using adeno-associated viruses (AAVs) has emerged as a promising approach for the treatment of genetic disorders, including hemophilia, a rare bleeding disorder. Recently approved AAV-based treatments for hemophilia, such as etranacogene dezaparvovec and fidanacogene elaparvovec, could provide long-term benefits by targeting the genetic basis of the condition. Nonetheless, the application of gene therapies presents several challenges that necessitate a multidisciplinary approach.
Objectives
To explore the challenges associated with AAV-based gene therapy and the role of pharmacy professionals in ensuring its safe and effective application, using treatment of hemophilia as a care model.
Data Sources
Relevant literature was identified through searches in the PubMed, Embase, and Google Scholar databases, with a focus on publications related to AAV-based gene therapy, hemophilia, and the involvement of pharmacy professionals.
Study Selection and Data Extraction
Peer-reviewed articles, clinical guidelines, and regulatory documents were selected based on their relevance to the clinical, logistical, and ethical dimensions of AAV-based gene therapy.
Data Synthesis
Pharmacy professionals are integral to the continuum of care for AAV-based gene therapy. Their responsibilities include product handling and preparation, patient education, medication management, and long-term safety monitoring. Furthermore, pharmacy professionals can help address challenges such as financial obstacles, regulatory adherence, and ethical issues.
Conclusions
Using their expertise in medication management, patient education, and health system processes, pharmacy professionals can enhance the safety, effectiveness, and accessibility of AAV-based gene therapies for hemophilia, ultimately leading to better patient outcomes.
KEYWORDS: adeno-associated virus, gene therapy, hemophilia, pharmacy professionals
RÉSUMÉ
Contexte
La thérapie génique utilisant les virus adéno-associés (AAV) a émergé comme une approche prometteuse pour le traitement des maladies génétiques, notamment l’hémophilie, un trouble rare de la coagulation. Des traitements récents contre l’hémophilie, approuvés et reposant sur les AAV, tels que l’étranacogène dézaparvovec et le fidanacogène élaparvovec, pourraient offrir des bienfaits à long terme en ciblant la cause génétique de la maladie. Néanmoins, l’application de ces thérapies géniques s’accompagne de plusieurs défis nécessitant une approche multidisciplinaire.
Objectif
Étudier les défis associés à la thérapie génique basée sur les AAV, ainsi que le rôle des professionnels de la pharmacie pour s’assurer de son administration sécuritaire et efficace, en prenant le traitement contre l’hémophilie comme exemple de prise en charge.
Sources des données
La littérature pertinente a été identifiée à l’aide de recherches effectuées sur PubMed, Embase et Google Scholar, en se concentrant sur les publications portant sur la thérapie génique basée sur les AAV, l’hémophilie et l’implication des professionnels de la pharmacie.
Sélection des études et extraction des données
Des articles évalués par des pairs, des guides de pratique clinique et des documents réglementaires ont été sélectionnés selon leur pertinence par rapport aux dimensions cliniques, logistiques et éthiques de la thérapie génique basée sur les AAV.
Synthèse des données
Les professionnels de la pharmacie jouent un rôle essentiel dans le continuum des soins lié à la thérapie génique basée sur les AAV. Leurs responsabilités comprennent la manipulation et la préparation du produit, l’éducation des patients, la gestion des médicaments ainsi que le suivi de l’innocuité à long terme. De plus, ils peuvent contribuer à relever divers défis, tels que les obstacles financiers, le respect de la réglementation et les enjeux éthiques.
Conclusions
Grâce à leur expertise en gestion des médicaments, en éducation des patients et dans les processus des systèmes de santé, les professionnels de la pharmacie peuvent améliorer la sécurité, l’efficacité et l’accessibilité des thérapies géniques basées sur les AAV pour le traitement de l’hémophilie, contribuant ainsi à de meilleurs résultats pour la santé des patients.
Mots-clés: virus adéno-associé, AAV, thérapie génique, hémophilie, professionnels de la pharmacie
Gene therapy has emerged as a novel approach for the treatment of genetic disorders, offering the potential to address the root cause of inherited conditions.1 The concept of using gene therapy to correct genetic abnormalities was introduced in 1972 in an article in the journal Science.2 Since then, the field of gene therapy has evolved considerably, and various types of gene therapy have been explored.1
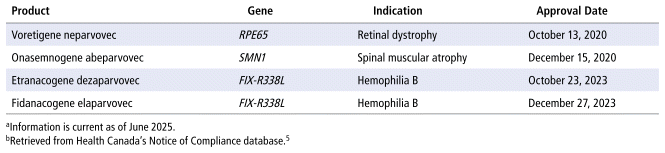
Gene therapy mediated through adeno-associated virus (AAV) has been studied for rare genetic disorders.3 AAV vectors are engineered viral particles that deliver therapeutic segments of genes derived from human DNA sequences. These recombinant AAV vectors have been studied for gene therapy applications because of their favourable properties, including their ability to infect both dividing and nondividing cells, their low pathogenicity, and their long-term gene expression, in addition to being nonreplicating vectors.3,4 As of June 2025, 4 AAV-based gene therapies had gained regulatory approval for use in Canada (Table 1): a pair of AAV-based therapies for hemophilia B (etranacogene dezaparvovec and fidanacogene elaparvovec), one therapy for retinal dystrophy (voretigene neparvovec), and one for spinal muscular atrophy (onasemnogene abeparvovec).5 More than 200 investigational AAV-based gene therapies are in various stages of clinical trials.6
TABLE 1 Gene Therapies Based on Adeno-Associated Virus Approved by Health Canadaa,b

Although the approval of AAV-based gene therapies provides renewed hope for the treatment of hemophilia and other rare inherited diseases, it also creates regulatory and ethical challenges and safety concerns.7 These challenges necessitate a new paradigm in health care delivery, one that emphasizes the importance of a multidisciplinary team approach. Practising at the intersection of medicine, science, and patient care, pharmacy professionals play an important role in ensuring the safe, effective, and efficient delivery of AAV-based gene therapy to patients with hemophilia.8,9 Pharmacy professionals are well positioned to facilitate the successful implementation of gene therapy across the continuum, from pretreatment preparation through treatment administration to post-treatment monitoring.8,9
In this review article, we discuss the critical role of pharmacy professionals in the successful implementation of AAV-mediated gene therapy, using hemophilia as a model. The article explores key considerations, challenges, and emerging trends, emphasizing the importance of pharmacy expertise across the continuum of care, including the implications of gene therapy approval for pharmacy professionals’ scope of practice.
The topic for this article was determined through a needs assessment performed for a program accredited by the Canadian Council on Continuing Education in Pharmacy (CCCEP), entitled “DECODED: Unraveling the Applications of Gene Therapy for Pharmacy Professionals”, which was developed for the Canadian Society of Hospital Pharmacists (now the Canadian Society of Healthcare-Systems Pharmacy) and sponsored by Pfizer Canada ULC.
A literature review was conducted to support the recommendations put forth through the educational program. Relevant literature was identified through searches in the PubMed, Embase, and Google Scholar databases, with a focus on publications related to AAV-based gene therapy, hemophilia, and the involvement of pharmacy professionals. Peer-reviewed articles, clinical guidelines, and regulatory documents were selected based on their relevance to the clinical, logistical, and ethical dimensions of AAV-based gene therapy.
This narrative review presents findings from the literature review and highlights some of the key recommendations from the educational program.
Hemophilia is a rare inherited bleeding disorder caused by mutations in the genes encoding clotting factor VIII (the F8 gene for hemophilia A) or IX (the F9 gene for hemophilia B). Without sufficient concentrations of these blood-clotting proteins, individuals with hemophilia experience prolonged bleeding episodes that can lead to serious complications and reduced quality of life.10 In Canada, hemophilia A affects approximately 1 in 5600 individuals, while hemophilia B affects approximately 1 in 25 600 individuals.11,12 Both forms of the disorder show an X-linked recessive pattern of inheritance and are thus more common in males.10
The current standard of care for patients with hemophilia involves factor replacement therapy with regular infusions of clotting factor concentrates. Although factor replacement therapy can be used to prevent and manage bleeding episodes, it imposes a considerable burden on patients and health care systems and necessitates lifelong treatment, because it does not address the underlying genetic cause.13–16 Additional limitations of factor replacement therapy include the need for frequent IV infusions, the risk of developing inhibitors against the infused factor, and the high cost.13–16
Bispecific antibodies, such as emicizumab and concizumab, are approved as routine prophylaxis to prevent bleeding or reduce the frequency of bleeding episodes in patients with hemophilia. However, these agents do not eliminate breakthrough bleeding, and repeated treatments are required.17,18 The frequency of administration of bispecific antibodies ranges from weekly to every 4 weeks, whereas prophylactic factor replacement therapy is typically administered 3 or 4 times per week for hemophilia A or 2 times per week for hemophilia B.
In addition, physical therapy, pain management, and joint treatments are used to prevent or manage complications associated with hemophilia; however, these represent only symptomatic management and further increase the treatment burden.13–15
Gene therapy has emerged as a novel approach to address the root cause of hemophilia and other genetic disorders. This approach, called gene replacement therapy, involves IV infusion of genetically modified AAV vectors that carry functional copies of the defective gene (in this context, the F8 gene for hemophilia A or the F9 gene for hemophilia B).19 Once in the body, the AAV vectors deliver the therapeutic gene into hepatocytes, which are responsible for the production of clotting factors. This process enables the patient’s hepatocytes to produce and secrete the missing clotting factor into the circulation, restoring normal hemostasis and preventing or reducing bleeding episodes.19 Currently, 2 AAV-based gene therapy products are approved by Health Canada for the treatment of hemophilia B: etranacogene dezaparvovec and fidanacogene elaparvovec (Table 1).
Etranacogene dezaparvovec is an AAV-based (serotype AAV5) gene therapy approved for the treatment of adults (≥ 18 years) with hemophilia B who rely on routine prophylactic therapies to prevent or reduce bleeding episodes.20 In the phase 3 HOPE-B trial (ClinicalTrials.gov identifier: NCT03569891), 54 adult males (≥ 18 years) with severe or moderately severe hemophilia B received a single infusion of etranacogene dezaparvovec.21 Compared with factor IX prophylaxis, infusion of etranacogene dezaparvovec resulted in a reduced annualized bleeding rate (relative risk [RR] 0.36, 95% confidence interval [CI] 0.20 to 0.64, p < 0.001), increased factor IX activity (by 34.3 [95% CI 29.5 to 39.1] percentage points, p < 0.001), and reduced usage of factor IX concentrate (mean decrease of 248 825 IU per year per participant in the post-treatment period, p < 0.001).21 No serious treatment-related adverse events or deaths were observed during the study period. The most common adverse event, occurring in 16.7% of participants, was increased alanine transaminase (ALT) necessitating supportive care with corticosteroids.21
Fidanacogene elaparvovec is an AAV-based (serotype AAVRh74) gene therapy approved for the treatment of adults (≥ 18 years) with moderately severe to severe hemophilia B who are negative for neutralizing antibodies to AAV serotype Rh74.22 In the phase 3 BENEGENE-2 trial (ClinicalTrials.gov identifier NCT03861273), 45 adult males (≥ 18 years) with moderately severe to severe hemophilia were given a single infusion of fidanacogene elaparvovec.22 Compared with factor IX prophylaxis, infusion of fidanacogene elaparvovec resulted in reduced annualized bleeding rate (treatment difference −2.34, 95% CI −4.97 to 0.29), reduced need for factor replacement therapy (treatment difference −54.37, 95% CI −63.64 to −45.10), and sustained expression of factor IX.22 The most common adverse events were increased serum transaminase concentration (in 46.6% of participants), nasopharyngitis (18.3%), and headache (16.6%).22
The therapeutic efficacy of AAV-based gene therapies for hemophilia depends not only on the vector delivery system but also on the regulatory elements that control transgene expression. Following IV administration, AAV vectors are distributed throughout the body and are taken up by multiple tissues, including the liver, skeletal muscle, and heart.23 However, both etranacogene dezaparvovec and fidanacogene elaparvovec use liver-specific promoter elements to direct selective expression of the factor IX transgene in hepatocytes, which are responsible for the production of clotting factors. Etranacogene dezaparvovec employs a liver-specific LP1 promoter, which is derived from the hepatitis B virus and provides robust, hepatocyte-targeted expression.20 Similarly, fidanacogene elaparvovec uses a liver-specific promoter, ApoE/hAAT, to ensure preferential transgene expression in hepatocytes, minimizing off-target expression in other tissues.22 This hepatocyte-specific targeting is crucial for achieving therapeutic factor IX levels while reducing the risk of immune responses that might occur with broader tissue expression patterns.24 Although AAV uptake occurs in multiple tissues, the off-target tissue distribution of these hemophilia B gene therapies has not been associated with significant clinical problems in clinical trials to date.20–22 The inclusion of liver-specific promoters minimizes transgene expression in nonhepatic tissues, thereby reducing the potential for adverse effects related to ectopic factor IX production. However, the biodistribution to multiple organs may contribute to immune responses against the AAV capsid, which can limit treatment efficacy and preclude repeat dosing with the same vector serotype.20–22
Despite promising results from clinical trials of AAV-mediated gene therapies for hemophilia, several challenges remain. Although gene therapy aims to provide long-lasting benefits, long-term data on its durability and potential adverse effects are still needed. Factors such as vector instability, immune responses, and epigenetic silencing may limit the long-term expression of the transgene, potentially necessitating complementary treatments such as factor IX prophylaxis.25
Gene-related therapies have other key challenges. Notably, they are very expensive. Beyond the financial challenges for health care facilities, paying agencies, and, potentially, the patient and family,26,27 in Canada, the lack of a well-defined regulatory framework for approval of these therapies, for post-marketing surveillance, and for cost–benefit monitoring is a consideration.26,28,29 Furthermore, various ethical and equity concerns require consideration.26,30 These challenges are discussed further in the section entitled “Need for a New Paradigm” (below).
Pre-existing immunity to AAV vectors or the transgene may render AAV-based gene therapy available to only a small portion of patients with hemophilia.31,32 In clinical trials for fidanacogene elaparvovec, trial eligibility criteria specified that all patients be screened for neutralizing antibodies to AAVRh74var using a cell-based antibody-mediated neutralization assay (transduction inhibition assay). In the global BeneGene-1 lead-in study, 61.1% of screened patients were positive for neutralizing antibodies (titer ≥ 1:1) and were therefore excluded.22 Moreover, potential immune responses triggered by the vector or transgene can limit the durability of gene therapy and make redosing with the same AAV vector impossible.31,32
Although no cases of treatment-emergent hepatocellular carcinoma have been reported in clinical trials of AAV-based gene therapy for hemophilia, the theoretical risk of insertional mutagenesis and carcinogenicity remains a concern. Studies in animal models have identified common integration sites for AAVs near active transcription units and CpG islands and have shown that preferences for integration in specific sites vary across different species.33 Despite evidence of near-random AAV integration in cell lines and animal models and evidence of AAV genotoxicity in neonatal mice studies, no confirmed genotoxic events have been reported in humans to date.33 Longer follow-up studies are needed to understand AAV integration in humans and its implications for patient safety and treatment efficacy.
Variability in transgene expression is another challenge in AAV-mediated gene therapy for hemophilia. A clinical trial of AAV5-based gene therapy for hemophilia A showed high variability in the therapeutic response and factor VIII activity. Median factor VIII activity at weeks 49 to 52 was 40 IU/dL or greater in 38% of patients, at least 5 IU/dL and less than 40 IU/dL in 50% of patients, less than 5 IU/dL in 12% of patients, and less than 3 IU/dL in 9% of patients.34 Although the causes of this variability in endogenous expression and clinical outcomes are not fully understood, factors such as vector dose, route of administration, and individual patient characteristics may influence the level and duration of transgene expression.34
Hepatotoxicity is a common complication of AAV-mediated gene therapy and increases the treatment burden for patients with hemophilia.35 Immune reactions after gene therapy could lead to hepatotoxicity and transaminitis and may influence the efficacy of gene therapy.36 This raises the need for frequent monitoring of ALT and aspartate aminotransferase (AST) levels and factor IX activity for several years after AAV-based gene therapy for hemophilia.20,22 Infusion-related effects are another common safety concern in patients with hemophilia who undergo AAV-based gene therapy.37 In clinical trials of etranacogene dezaparvovec infusion in patients with hemophilia B, 33.0% of patients experienced infusion-related reactions, which were the most common treatment-emergent adverse events.20
Pharmacy professionals lead the procurement, storage, quality control, preparation, and disposal of gene therapy products. Their responsibilities include coordinating with manufacturers, ensuring timely availability of the gene therapy product, and implementing measures to maintain vector integrity and stability before use.8,38,39 AAV vectors have specific storage requirements, such as ultra-low temperatures, to maintain their integrity and potency.40,41 Pharmacy professionals are responsible for ensuring that these storage requirements are met, as any deviations can reduce the integrity and efficacy of the gene therapy product.8,9
The preparation of AAV gene therapy products for administration involves aseptic preparation and reconstitution in a controlled, classified aseptic environment, to prevent contamination, with appropriate infrastructure to minimize staff members’ exposure to gene therapy products.8 Certified pharmacy professionals are trained in aseptic techniques, quality control processes, and proper handling of therapeutic or hazardous products. As such, they are well positioned to establish appropriate handling and preparation procedures, including the use of personal protective equipment (PPE), aseptic techniques, and biosafety cabinets.8,38,42 Pharmacy professionals are also key to establishing procedures for safe disposal of gene therapy–related materials to minimize exposure risks for pharmacy staff.8,42 Protocols for handling and preparing gene therapy products are needed to minimize the risk of occupational exposure, errors, or contamination that could compromise patient safety, therapeutic outcomes, and the safety of health care professionals.
Practical considerations for pharmacy departments include ensuring availability of the following: appropriate refrigeration and freezer units with backup power sources for product storage and thawing; frameworks for product ordering, reception, and transport of gene therapy products from the pharmacy to the preparation area and the administration site; necessary certifications for handling viral vectors; PPE, including gowns, gloves, and respiratory and eye protection (e.g., mask, goggles, and face shields); and spill kits and appropriate disposal containers.8,43 In terms of evaluating the site’s infrastructure, it is crucial to assess the capacity of the facility to accommodate the necessary clean room requirements, equipment, and storage units, as well as the ability to maintain the required environmental conditions for product storage and safe handling. This includes evaluating the availability of backup power sources to ensure continuous operation of refrigeration and freezer units. Because national guidelines and frameworks for the implementation of gene therapies are lacking, pharmacy professionals may play a crucial role in developing institutional protocols, policies, and standard operating procedures, as well as in establishing the appropriate infrastructure for the proper and safe preparation, handling, and disposal of gene therapy products, as well as for spill management and decontamination processes.
Pharmacy professionals contribute to patient selection and screening for AAV-based gene therapy through verification of key eligibility criteria, including patient age (≥ 18 years for both approved therapies), hemophilia B diagnosis with documented factor IX deficiency, and severity criteria (moderately severe to severe hemophilia).20,22 Pharmacy professionals can develop comprehensive screening checklists that include assessment for contraindications (such as active hepatitis B or C infection), significant liver disease (ALT and AST > 2 times the upper limit of normal), history of hypersensitivity reactions, and current immunosuppressive therapy.20,22,44,45 For neutralizing antibody screening, pharmacists coordinate with laboratories to ensure appropriate sample collection, storage, and transport for cell-based transduction inhibition assays, with specific protocols for AAV5 (etranacogene dezaparvovec) and AAVRh74 (fidanacogene elaparvovec) serotypes. Liver function assessment protocols should include baseline measurements of ALT, AST, total and direct bilirubin, alkaline phosphatase, and albumin.20,22,44,45
Effective patient and family education and counselling are essential for successfully implementing gene therapy and setting realistic patient expectations. In a survey of Canadians with hemophilia conducted by the Canadian Hemophilia Society, 43% of respondents indicated that a minimum factor VIII or IX expression of 40% to 100% of normal levels would convince them to be treated with gene therapy.46 Moreover, 40% of patients reported expecting that the desired factor concentration would last their entire lives.46 These findings suggest that many Canadians with hemophilia have unrealistic expectations for gene therapy, which reinforces the importance of pre-infusion counselling and the setting of realistic expectations. As part of the multidisciplinary care team preparing patients for AAV-based gene therapy, pharmacy professionals, with their expertise in medication management and patient communication, are well positioned to provide clear, comprehensive, and evidence-based counselling and education to patients with hemophilia and their family members.44,47
Patient and family education topics should include the mechanism of action, expected outcomes, anticipated benefits, potential risks, common adverse effects, monitoring requirements, vector shedding precautions, long-term follow-up considerations, necessary lifestyle changes (e.g., reduction of alcohol consumption, use of barrier contraception), and gene therapy access and coverage considerations.44,47 Pharmacy professionals can develop standardized patient education materials, including visual aids explaining the mechanism of AAV gene delivery, infographic comparisons of gene therapy and conventional treatment burdens, and realistic timeline expectations showing factor IX expression patterns over months to years. Effective counselling scripts should address common patient questions, such as “Will I ever need factor IX again?”, “Is this a cure?”, and “What are the chances it will work for me?” Informed consent enhancement strategies include using teach-back methods to confirm patient understanding, providing written summaries of key risks and benefits in plain language, offering family counselling sessions to address concerns about genetic implications, and implementing cooling-off periods between initial consultation and treatment decisions to allow adequate time for consideration.
In collaboration with other health care providers, pharmacy professionals can lead the development of patient support programs tailored to the needs and considerations of gene therapy recipients. Moreover, in preparing patients with hemophilia for gene therapy, pharmacy professionals collaborate with the team in the selection and correct dosing of prophylactic and supportive care medications associated with gene therapy, including immunosuppressants and adjunctive therapies.8,9,48
Pharmacy professionals can take the lead in developing standardized protocols and guidelines for the safe and effective administration of gene therapy, based on their expertise in medication management, dosing, and administration protocols.8,9,44 Pharmacy professionals can also provide guidance during the infusion of AAV gene therapy products, ensuring that therapy administration is carried out correctly and in accordance with established protocols.8,9,44 This may include monitoring and managing infusion-related adverse events and complications.8,9,44,49 They also play a key role in establishing and implementing protocols for post-infusion monitoring of concentrations of the clotting factor, liver enzymes, and total bilirubin, which are essential for assessing therapeutic response and risk of hepatotoxicity.8,9,44
The long-term safety of gene therapies and the durability of transgene expression are unknown, which emphasizes the importance of long-term monitoring of patients after AAV-based gene therapy.50 Long-term follow-up considerations include monitoring for adverse events, educating patients and families on potential long-term adverse effects, and monitoring adherence to post-infusion medications and lifestyle adaptations (e.g., use of barrier contraception and reduction of alcohol consumption). As part of the treatment team, pharmacy professionals may be involved with registries (e.g., gene therapy module of the Canadian Bleeding Disorders Registry, Gene Therapy Registry of the World Bleeding Disorders Registry) to document outcomes and routinely export data to provincial and national databases.8,48,51 Pharmacy professionals are well positioned to lead all these aspects of long-term follow-up of patients with hemophilia after AAV-based gene therapy.
In the immediate post-infusion period (first 12 weeks), patients require weekly monitoring of liver function tests (ALT, AST, total bilirubin) and factor IX activity levels to detect early hepatotoxicity and to assess initial therapeutic response.20,22 During the intermediate phase (3 months to 2 years), monitoring frequency can be reduced to monthly for the first 6 months, then every 3 months, with a focus on sustained factor IX expression, liver enzyme stability, and clinical bleeding outcomes. Long-term monitoring (beyond 2 years) should occur every 6 to 12 months and should include comprehensive assessments of factor IX activity, complete blood count, comprehensive metabolic panel, and screening for potential late-onset adverse events, such as hepatocellular carcinoma, through imaging studies and α-fetoprotein levels. Additional biomarkers of interest include anti-AAV antibodies, which indicate immune responses; inflammatory markers (C-reactive protein, interleukin-6), which are present during acute phases; and coagulation parameters beyond factor IX levels (such as activated partial thromboplastin time and thrombin generation assays), which can be measured to evaluate overall hemostatic function. Vector shedding could be monitored through polymerase chain reaction–based detection in bodily fluids during the first months after infusion, with contraceptive counselling during this period.
The duration of patient follow-up after AAV-based gene therapy should be long enough to allow monitoring of patients for potential delayed adverse events according to the product type, the route of administration, the product’s persistence and expression, the patient’s expected survival, and the background rates of the events of interest.52 Follow-up protocols should include a detailed plan for the schedule of patient visits, sampling methods, monitoring tests, clinical events of interest, detection and reporting of adverse events, causality analyses, and informed consent.52
Pharmacy professionals can also lead the establishment and implementation of institutional protocols, pharmacovigilance frameworks, and pharmacy checklists for patient-centric monitoring and for mitigating the long-term complications of AAV-mediated gene therapy. These frameworks should address the timing of monitoring and the parameters to be monitored, the management of potential immune responses and delayed adverse effects, monitoring of the impact of immune reactions on liver health and transgene expression, the use of immunosuppressive treatments (e.g., corticosteroids) when necessary, the patient’s adherence to recommended lifestyle changes, and the adjustment of immunization schedules after gene therapy in patients who are also undergoing immunosuppressive treatments.52,53
Although there have been reports of serious adverse events following AAV-based gene therapy, reporting and monitoring of these adverse events have not been consistent among trials and clinics.54,55 Pharmacy professionals are uniquely positioned to collaborate with regulatory agencies and manufacturers to report delayed therapy-related adverse events, to contribute to the evolving understanding of gene therapy safety profiles, to report any cases of hepatocellular carcinoma, to lead data collection and analysis efforts to assess real-world outcomes, and to inform future gene therapy development and utilization based on the collected data. Pharmacy professionals can also contribute to the long-term safety of gene therapy products by educating patients about the potential adverse events associated with gene therapy, the importance of prompt reporting of adverse events, and the need for ongoing monitoring and follow-up to assess the durability of gene therapy effects and potential late-onset adverse events.56,57
Gene therapy products are subject to stringent regulations and oversight by various agencies, such as Health Canada, the US Food and Drug Administration, and the European Medicines Agency.26,58,59 Integration of gene therapy into the Canadian health care system poses several regulatory challenges, including the lack of formal definition and classification of gene therapies, the limited capacity and expertise of regulatory bodies, the lack of pertinent legislation (or the presence of legislation that is not up to date with clinical practice), and insufficient post-market surveillance and data collection.26,28,29
Pharmacy professionals can ensure institutional preparedness and regulatory compliance by leading efforts to develop comprehensive guidelines and education programs for nursing, medical, and pharmacy staff and by collaborating with multidisciplinary teams to ensure a cohesive approach to gene therapy implementation.8,9 By staying informed about the evolving regulatory framework relating to gene therapy, pharmacy professionals can also contribute to developing and updating institutional policies and procedures for the handling and administration of gene therapy, as well as liaising with regulatory bodies and professional organizations to stay current on guidelines and best practices.
Gene therapy raises unique ethical and social concerns that need to be addressed. Although current gene therapies for hemophilia target somatic cells and do not introduce heritable genetic modifications, the potential for germline editing raises ethical concerns about altering the human genetic makeup and implications for future generations.26,30 There are also uncertainties regarding how to determine when there is sufficient preclinical evidence to initiate clinical trials and how to balance risks and benefits in dose selection, especially for irreversible and immunogenic gene therapies.26,30
Rigorous oversight and public discourse are needed to address concerns related to germline editing in patients undergoing gene therapy.60,61 Pharmacy professionals can enhance informed consent–related processes by ensuring that patients and their families understand the risks, benefits, and long-term implications of AAV-mediated gene therapy and addressing concerns about germline modifications.62 They must also collaborate with clinicians to develop fair and transparent patient selection criteria that consider both medical and ethical factors.63 In collaboration with bioethicists and other stakeholders, pharmacy professionals can develop policies and guidelines that balance the potential benefits of gene therapy with ethical principles and societal values.64,65
Gene therapy raises concerns about equitable access and coverage of the associated high costs.26,27 The high cost of gene therapies, particularly those targeting rare diseases, reflects the substantial investment required to develop and manufacture AAV-mediated gene therapy products.66,67 In Canada, etranacogene dezaparvovec and fidanacogene elaparvovec each cost approximately $4 million to $5 million per treatment.68,69 These one-time treatment costs must be considered in the context of the lifelong expenses associated with conventional factor IX prophylaxis, which can range from about $322 000 to $492 000 annually per patient, depending on disease severity and treatment regimen.68,69 Financial challenges associated with the integration of gene therapy into the Canadian health care system include limited coverage, budget constraints, lack of gene therapy–specific guidelines and frameworks from health technology assessment bodies, and uncertainties about the efficacy, long-term safety, and cost-effectiveness of AAV-based gene therapies.26,27
Pharmacy professionals can reduce the health care costs of gene therapy by conducting pharmacoeconomic analyses and evaluating the cost-effectiveness of gene therapy compared to traditional treatments.70 Factors to consider in pharmacoeconomic evaluations include the initial acquisition costs of gene therapy products, which can be substantial but may provide long-term benefits; the potential reductions in long-term health care costs associated with avoiding complications, hospitalizations, and ongoing traditional treatments; the value of improved quality of life and functional abilities for patients receiving gene therapy; and the broader economic implications of gene therapy, such as increased productivity and reduced caregiver burden.71–73
Pharmacy professionals can work with payers, health economists, health care administrators, patient advocacy groups, and other stakeholders to explore strategies for improving affordability and to develop reimbursement strategies to ensure that patients who could benefit from gene therapy have equitable opportunities to receive these treatments.74–76 Pharmacy professionals can also be vital in navigating the complex landscape of coverage and reimbursement for AAV-mediated gene therapy. This work includes collaborating with payers to develop appropriate coverage policies, advocating for fair reimbursement rates, and assisting patients in accessing available financial assistance programs.77,78 In addition, having trained pharmacists for on-site product preparation under aseptic conditions could enhance the ability of hospitals to deliver gene therapies and address some of the concerns about equitable access.79,80
The successful implementation of gene therapy requires close collaboration among various health care professionals, each contributing their unique expertise and perspective (Figure 1). Pharmacy professionals play a pivotal role in this multidisciplinary team, working closely with hematologists at infusion sites in patient selection, treatment planning, dose calculations, and monitoring of clinical outcomes, and with nurses and hematologists at referral or follow-up sites in coordination of care transitions, patient education (e.g., managing expectations of patients and caregivers), follow-up protocols (including adherence to lifestyle modifications), monitoring for complications of immunosuppression, assessment of eligibility and reimbursement requirements, and performance of post-infusion monitoring. In collaboration with hepatologists, pharmacy professionals can contribute to post-infusion monitoring and educating patients about long-term liver health. Pharmacy professionals can also liaise with regulatory and compliance experts to ensure adherence to relevant laws, regulations, and institutional policies governing gene therapy. Moreover, together with bioethicists and patient advocates, pharmacy professionals can help address ethical considerations, promote patient autonomy, and ensure equitable access to gene therapy.8,44,81
|
| ||
|
FIGURE 1 Multidisciplinary team approach in gene therapy for hemophilia. Diagram showcasing the close collaboration of pharmacy professionals with various health care professionals involved in gene therapy care. | ||
Patients require frequent and long-term follow-up after gene therapy for hemophilia, which can add to the burden of this disease for patients and their care partners.36,82 The preparation of patients and their families for gene therapy and the post-administration monitoring phase could be enhanced through the use of telehealth and remote monitoring platforms.83 Pharmacy professionals must be prepared to use these tools to facilitate patient follow-up, monitor adverse events, and optimize therapeutic outcomes.
The recent approval of AAV-mediated gene therapy is a significant advancement in hemophilia treatment. The adoption of these new therapies requires a multidisciplinary effort, with pharmacy professionals playing a vital role at various stages of the process. From ensuring regulatory compliance and ethical oversight to managing the handling, preparation, and administration of gene therapy products, pharmacy professionals contribute their expertise to optimize patient safety and therapeutic outcomes. Their involvement in patient education, counselling, and long-term monitoring is also important for managing expectations, promoting adherence, and evaluating the durability and potential adverse effects of gene therapy. Furthermore, pharmacy professionals’ contributions to performing pharmacoeconomic analyses, developing reimbursement strategies, and addressing access challenges are essential for navigating the complex financial landscape of gene therapies and ensuring equitable access for eligible patients.
Emerging developments in gene therapy are expected to expand the role of pharmacy professionals in the management of hemophilia. Next-generation AAV vectors with improved tissue specificity and reduced immunogenicity, redosing strategies enabled by immunomodulatory approaches, and advances in manufacturing, including point-of-care preparation technologies,84–86 will require pharmacy professionals to continuously adapt their protocols and infrastructure. The integration of digital health tools, artificial intelligence–assisted monitoring systems, and telemedicine platforms could enhance remote patient monitoring capabilities and predictive adverse event detection.87–89 In addition, advancements in potential CRISPR-based gene therapies90 necessitate that pharmacy professionals prepare for the increased complexity of the hemophilia treatment landscape through ongoing education, specialized training programs, and scalable operational frameworks.
The evolving regulatory landscape is likely to expand the responsibilities of pharmacy professionals, necessitating targeted education and training programs. Specialized training requirements could include certification programs covering AAV vector biology, aseptic handling of viral vectors, biosafety protocols (e.g., spill management), pharmacovigilance, and ethical aspects of gene therapy (e.g., equity considerations, germline modification concerns). Professional development opportunities such as workshops on gene therapy counselling techniques, webinars on emerging vector technologies, and hands-on training for specialized storage and preparation equipment would be essential for maintaining competency. To enhance their impact, pharmacy professionals should engage in continued collaboration with other health care professionals and stakeholders, pursue specialized education and training in gene therapy, and advocate for proactive guidance and leadership from pharmacy organizations.
1 Dunbar CE, High KA, Joung JK, Kohn DB, Ozawa K, Sadelain M. Gene therapy comes of age. Science. 2018;359(6372):eaan4672.
Crossref PubMed
2 Friedmann T, Roblin R. Gene therapy for human genetic disease? Science. 1972;175(4025):949–55.
Crossref PubMed
3 Wang JH, Gessler DJ, Zhan W, Gallagher TL, Gao G. Adeno-associated virus as a delivery vector for gene therapy of human diseases. Signal Transduct Target Ther. 2024;9(1):78.
Crossref PubMed PMC
4 Sayed N, Allawadhi P, Khurana A, Singh V, Navik U, Pasumarthi SK, et al. Gene therapy: comprehensive overview and therapeutic applications. Life Sci. 2022;294:120375.
Crossref PubMed
5 Notice of Compliance (NOC) search [database on Internet]. Health Canada; [cited 2024 Jul 19]. Available from: https://health-products.canada.ca/noc-ac/
6 ClinicalTrials.gov [database on Internet]. National Library of Medicine (US); [cited 2024 Jul 19]. Available from: https://clinicaltrials.gov/search?intr=AAV
7 Kohn DB, Chen YY, Spencer MJ. Successes and challenges in clinical gene therapy. Gene Ther. 2023;30(10–11):738–46.
Crossref PubMed PMC
8 Petrich J, Marchese D, Jenkins C, Storey M, Blind J. Gene replacement therapy: a primer for the health-system pharmacist. J Pharm Pract. 2020; 33(6):846–55.
Crossref PMC
9 Myers CJ. Preparing pharmacists to manage gene therapies. J Am Pharm Assoc (2003). 2021;61(3):e78–e82.
Crossref
10 Berntorp E, Fischer K, Hart DP, Mancuso ME, Stephensen D, Shapiro AD, et al. Haemophilia. Nat Rev Dis Primers. 2021;7(1):45.
Crossref PubMed
11 Factor FIX deficiency – 2021. Association of Hemophilia Clinic Directors of Canada; [cited 2024 Jul 15]. Available from: https://www.ahcdc.ca/storage/pdf/21/FIX-2021.pdf
12 Factor FVIII deficiency 2021. Association of Hemophilia Clinic Directors of Canada; [cited 2024 Jul 15]. Available from: https://www.ahcdc.ca/storage/pdf/21/FVIII-2021.pdf
13 Pratap R, Misra M, N V, Morampudi S, Patil A, Reddy J. The existing scenario of haemophilia care in Canada and China - a review. Hematol Transfus Cell Ther. 2020;42(4):356–64.
Crossref PMC
14 Okaygoun D, Oliveira DD, Soman S, Williams R. Advances in the management of haemophilia: emerging treatments and their mechanisms. J Biomed Sci. 2021;28(1):64.
Crossref PubMed PMC
15 Canadian integrated and comprehensive care standards for inherited bleeding disorders. 2nd ed. Canadian Inherited Bleeding Disorder Standards Working Group; [cited 2024 Jul 16]. Available from: https://www.hemophilia.ca/wp-content/uploads/2020/04/Standards-of-care-draft-5-06-04-2020-EN.pdf
16 Guidelines for emergency management of hemophilia and von Willebrand disease. Canadian Hemophilia Society; [cited 2024 Jul 16]. Available from: https://www.hemophilia.ca/wp-content/uploads/2020/10/FactorFirst-poster-January-2020-final.pdf
17 Hermans C, Gruel Y, Frenzel L, Krumb E. How to translate and implement the current science of gene therapy into haemophilia care? Ther Adv Hematol. 2023;14:20406207221145627.
Crossref PubMed PMC
18 Keam SJ. Concizumab: first approval. Drugs. 2023;83(11):1053–9.
Crossref PubMed
19 Castaman G, Di Minno G, De Cristofaro R, Peyvandi F. The arrival of gene therapy for patients with hemophilia A. Int J Mol Sci. 2022; 23(18):10228.
Crossref PubMed PMC
20 Hemgenix (etranacogene dezaparvovec) [product monograph]. CSL Behring Canada Inc; 2023 Oct 23.
21 Pipe SW, Leebeek FWG, Recht M, Key NS, Castaman G, Miesbach W, et al. Gene therapy with etranacogene dezaparvovec for hemophilia B. N Engl J Med. 2023;388(8):706–18.
Crossref PubMed
22 Beqvez (fidanacogene elaparvovec) [product monograph]. Pfizer Canada ULC; 2023 Dec 27.
23 Zhao J, Yue Y, Patel A, Wasala L, Karp JF, Zhang K, et al. High-resolution histological landscape of AAV DNA distribution in cellular compartments and tissues following local and systemic injection. Mol Ther Methods Clin Dev. 2020;18:856–68.
Crossref PubMed PMC
24 Becker J, Domenger C, Choksi P, Krämer C, Baumgartl C, Maiakovska O, et al. Identification of a robust promoter in mouse and human hepatocytes by in vivo biopanning of a barcoded AAV library. Mol Ther. 2025;33(8):3881–901.
Crossref PubMed PMC
25 Greig JA, Martins KM, Breton C, Lamontagne RJ, Zhu Y, He Z, et al. Integrated vector genomes may contribute to long-term expression in primate liver after AAV administration. Nat Biotechnol. 2023;42(8):1232–42.
Crossref PubMed PMC
26 Chisholm J, Ruff C, Viswanathan S. Current state of Health Canada regulation for cellular and gene therapy products: potential cures on the horizon. Cytotherapy. 2019;21(7):686–98.
Crossref PubMed
27 Gene therapy: international regulatory and health technology assessment (HTA) activities and reimbursement status. Canada’s Drug and Health Technology Agency (CADTH); 2018 Mar 23 [cited 2024 Jul 20]. Available from: https://www.cadth.ca/gene-therapy-international-regulatory-and-health-technology-assessment-hta-activities-and
28 Wang J, Griffiths E, Tounekti O, Nemec M, Deneault E, Lavoie JR, et al. Canadian regulatory framework and regulatory requirements for cell and gene therapy products. Adv Exp Med Biol. 2023;1430:91–116.
Crossref PubMed
29 Peyvandi F, Lillicrap D, Mahlangu J, McLintock C, Pasi KJ, Pipe SW, et al. Hemophilia gene therapy knowledge and perceptions: results of an international survey. Res Pract Thromb Haemost. 2020;4(4):644–51.
Crossref PubMed PMC
30 Iyer AA, Saade D, Bharucha-Goebel D, Foley AR, Averion GM, Paredes E, et al. Ethical challenges for a new generation of early-phase pediatric gene therapy trials. Genet Med. 2021;23(11):2057–66.
Crossref PubMed
31 Nidetz NF, McGee MC, Tse LV, Li C, Cong L, Li Y, et al. Adeno-associated viral vector-mediated immune responses: understanding barriers to gene delivery. Pharmacol Ther. 2020;207:107453.
Crossref PMC
32 Mingozzi F, High KA. Immune responses to AAV vectors: overcoming barriers to successful gene therapy. Blood. 2013;122(1):23–36.
Crossref PubMed PMC
33 Sabatino DE, Bushman FD, Chandler RJ, Crystal RG, Davidson BL, Dolmetsch R, et al. Evaluating the state of the science for adeno-associated virus integration: an integrated perspective. Mol Ther. 2022; 30(8):2646–63.
Crossref PubMed PMC
34 Ozelo MC, Mahlangu J, Pasi KJ, Giermasz A, Leavitt AD, Laffan M, et al. Valoctocogene roxaparvovec gene therapy for hemophilia A. N Engl J Med. 2022;386(11):1013–25.
Crossref PubMed
35 Ertl HCJ. Immunogenicity and toxicity of AAV gene therapy. Front Immunol. 2022;13:975803.
Crossref PubMed PMC
36 Pipe SW, Reddy KR, Chowdary P. Gene therapy: practical aspects of implementation. Haemophilia. 2022;28(Suppl 4):44–52.
Crossref PubMed PMC
37 Leebeek FWG, Miesbach W. Gene therapy for hemophilia: a review on clinical benefit, limitations, and remaining issues. Blood. 2021;138(11): 923–31.
Crossref PubMed
38 Shay B, Storey M. Gene therapy: practical considerations for clinical and operational pharmacy practice. Am J Health Syst Pharm. 2024; 81(12):479–82.
Crossref PubMed
39 Blind JE, McLeod EN, Campbell KJ. Viral-mediated gene therapy and genetically modified therapeutics: a primer on biosafety handling for the health-system pharmacist. Am J Health Syst Pharm. 2019; 76(11):795–802.
Crossref PubMed
40 Grossen P, Skaripa Koukelli I, van Haasteren J, Machado AHE, Dürr C. The ice age - a review on formulation of adeno-associated virus therapeutics. Eur J Pharm Biopharm. 2023;190:1–23.
Crossref PubMed
41 Doan TNK, Le MD, Bajrovic I, Celentano L, Krause C, Grooms H, et al. Thermostability and in vivo performance of AAV9 in a film matrix. Commun Med (Lond). 2022;2:148.
Crossref PubMed PMC
42 American Society of Health-System Pharmacists. ASHP guidelines on compounding sterile preparations. Am J Health Syst Pharm. 2014; 71(2):145–66.
Crossref PubMed
43 Astermark J, Baghaei F, Strandberg K, Toplican PG, Birkedal MF, Grahn EE, et al. Infrastructural considerations of implementing gene therapy for hemophilia in the Nordic context. Ther Adv Hematol. 2023; 14:20406207231202306.
Crossref PubMed PMC
44 Pipe S, Douglas K, Hwang N, Young G, Patel P, Fogarty P. Delivery of gene therapy in haemophilia treatment centres in the United States: practical aspects of preparedness and implementation. Haemophilia. 2023;29(6):1430–41.
Crossref PubMed
45 Mendell JR, Connolly AM, Lehman KJ, Griffin DA, Khan SZ, Dharia SD, et al. Testing preexisting antibodies prior to AAV gene transfer therapy: rationale, lessons and future considerations. Mol Ther Methods Clin Dev. 2022;25:74–83.
Crossref PubMed PMC
46 Submission to the Canadian Agency for Drugs and Technologies in Health (CADTH) by the Canadian Hemophilia Society on fidanacogene elaparvovec. Canadian Hemophilia Society; 2023 Aug 10 [cited 2024 Jul 24]. Available from: https://www.hemophilia.ca/wp-content/uploads/2023/10/CHS-submission-to-CADTH-for-fidanacogene-elaparvovec-August-10-2023.pdf
47 Charafi L, Campanella L, Shay B, Rabatin A, Hughes A, Kennerly-Shah J. Expanding pharmacy services to the hemophilia treatment center. Hosp Pharm. 2023;58(5):415–9.
Crossref PubMed PMC
48 Lozano-Bläzquez A, Makridaki D, Plesan CR, Kohl S; delegates of 52nd EAHP General Assembly. Advanced therapy medicinal products. Eur J Hosp Pharm. 2023;31(1):63–5.
Crossref PubMed PMC
49 Miesbach W, Mulders G, Breederveld D, Pinachyan K, Le Quellec S, Pabinger I. A 360-degree perspective on adeno-associated virus (AAV)-based gene therapy for haemophilia: insights from the physician, the nurse and the patient. Orphanet J Rare Dis. 2024;19(1):193.
Crossref PubMed PMC
50 Gene therapy needs a long-term approach [editorial]. Nat Med. 2021; 27(4):563.
Crossref
51 Hernandez JM. Biosafety considerations for viral vector gene therapy: an explanation and guide for the average everyday-hero pharmacist. J Pharm Pract. 2023;36(6):1532–9.
Crossref
52 Long-term follow-up after administration of human gene therapy products. Guidance for industry. US Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research; 2020 Jan [cited 2024 Jul 25]. Available from: https://www.fda.gov/media/113768/download
53 Committee for Medicinal Products for Human Use (CHMP). Guideline on follow-up of patients administered with gene therapy medicinal products. European Medicines Agency; 2009 Oct 22 [cited 2024 Jul 25]. Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-follow-patients-administered-gene-therapy-medicinal-products_en.pdf
54 Servais L, Horton R, Saade D, Bonnemann C, Muntoni F; 261st ENMC workshop study group. 261st ENMC international workshop: management of safety issues arising following AAV gene therapy. 17th–19th June 2022, Hoofddorp, The Netherlands. Neuromuscul Disord. 2023;33(11):884–96.
Crossref PubMed
55 Wills CA, Drago D, Pietrusko RG. Clinical holds for cell and gene therapy trials: risks, impact, and lessons learned. Mol Ther Methods Clin Dev. 2023;31:101125.
Crossref PubMed PMC
56 Miesbach W, O’Mahony B, Key NS, Makris M. How to discuss gene therapy for haemophilia? A patient and physician perspective. Haemophilia. 2019;25(4):545–57.
Crossref PubMed PMC
57 Petitpain N, Antoine ML, Beurrier M, Micallef J, Fresse A. Pharmacovigilance of gene therapy medicinal products. Rare Dis Orphan Drugs J. 2023;2(4):25.
Crossref
58 Takefman D, Bryan W. The state of gene therapies: the FDA perspective. Mol Ther. 2012;20(5):877–8.
Crossref PubMed PMC
59 Bachtarzi H, Farries T. The genetically modified organism medicinal framework in Europe, United States, and Japan: underlying scientific principles and considerations toward the development of gene therapy and genetically modified cell-based products. Hum Gene Ther Clin Dev. 2019;30(3):114–28.
Crossref PubMed
60 Meagher KM, Allyse MA, Master Z, Sharp RR. Reexamining the ethics of human germline editing in the wake of scandal. Mayo Clin Proc. 2020;95(2):330–8.
Crossref PubMed
61 Ormond KE, Mortlock DP, Scholes DT, Bombard Y, Brody LC, Faucett WA, et al. Human germline genome editing. Am J Hum Genet. 2017;101(2):167–76.
Crossref PubMed PMC
62 Woollard L, Gorman R, Rosenfelt DJ. Improving patient informed consent for haemophilia gene therapy: the case for change. Ther Adv Rare Dis. 2021;2:26330040211047244.
PubMed PMC
63 National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Sciences Policy; Forum on Regenerative Medicine. Understanding the complexities of patient selection, enrollment, and the consent process. In: Beachy SH, Alper J, Hackmann M, Addie S, editors. Exploring novel clinical trial designs for gene-based therapies: proceedings of a workshop. National Academies Press (US); 2020 Apr 9 [cited 2024 Jul 25]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK559964
64 Spink J, Geddes D. Gene therapy progress and prospects: bringing gene therapy into medical practice: the evolution of international ethics and the regulatory environment. Gene Ther. 2004;11(22):1611–6.
Crossref PubMed
65 Ansah EO. Ethical challenges and controversies in the practice and advancement of gene therapy. Adv Cell Gene Ther. 2022;2022:1015996.
Crossref
66 Hitchcock T. Manufacturing of AAV vectors: translational challenges from development to industrialisation. Emerg Top Life Sci. 2021;5(5): 725–8.
Crossref PubMed
67 van der Loo JCM, Wright JF. Progress and challenges in viral vector manufacturing. Hum Mol Genet. 2016;25(R1):R42–52.
Crossref PMC
68 CADTH reimbursement review: etranacogene dezaparvovec (Hemgenix). Can J Health Technol. 2024;4(9):158 pages.
69 CADTH reimbursement review: fidanacogene elaparvovec (Beqvez). Can J Health Technol. 2024;4(6):191 pages.
70 Dalton K, Byrne S. Role of the pharmacist in reducing healthcare costs: current insights. Integr Pharm Res Pract. 2017;6:37–46.
71 Gavan SP, Wright SJ, Thistlethwaite F, Payne K. Capturing the impact of constraints on the cost-effectiveness of cell and gene therapies: a systematic review. Pharmacoeconomics. 2023;41(6):675–92.
Crossref PubMed
72 Pochopień M, Qiu T, Aballea S, Clay E, Toumi M. Considering potential solutions for limitations and challenges in the health economic evaluation of gene therapies. Expert Rev Pharmacoecon Outcomes Res. 2021;21(6):1145–58.
Crossref
73 Toumi M, Dabbous O, Aballéa S, Drummond MF, von der Schulenburg JMG, Malone DC, et al. Recommendations for economic evaluations of cell and gene therapies: a systematic literature review with critical appraisal. Expert Rev Pharmacoecon Outcomes Res. 2023;23(5):483–97.
Crossref PubMed
74 Salzman R, Cook F, Hunt T, Malech HL, Reilly P, Foss-Campbell B, et al. Addressing the value of gene therapy and enhancing patient access to transformative treatments. Mol Ther. 2018;26(12):2717–26.
Crossref PubMed PMC
75 Hampson G, Towse A, Pearson SD, Dreitlein WB, Henshall C. Gene therapy: evidence, value and affordability in the US health care system. J Comp Eff Res. 2018;7(1):15–28.
Crossref
76 Adachi T, El-Hattab AW, Jain R, Nogales Crespo KA, Quirland Lazo CI, Scarpa M, et al. Enhancing equitable access to rare disease diagnosis and treatment around the world: a review of evidence, policies, and challenges. Int J Environ Res Public Health. 2023;20(6):4732.
Crossref PubMed PMC
77 Koleva-Kolarova R, Buchanan J, Vellekoop H, Huygens S, Versteegh M, Rutten-van Mölken M, et al.; HEcoPerMed Consortium. Financing and reimbursement models for personalised medicine: a systematic review to identify current models and future options. Appl Health Econ Health Policy. 2022;20(4):501–24.
Crossref PubMed PMC
78 Bolous NS, Bhatt N, Bhakta N, Neufeld EJ, Davidoff AM, Reiss UM. Gene therapy and hemophilia: where do we go from here? J Blood Med. 2022;13:559–80.
Crossref PubMed PMC
79 Eppink M, Wapenaar M, Crommelin D, et al. Therapeutic proteins and advanced therapy medicinal products. In: Le Brun P, Crauste-Manciet S, Krämer I, Smith J, Woerdenbag H, editors. Practical pharmaceutics: an international guideline for the preparation, care and use of medicinal products. Springer International Publishing; 2023. pp. 551–90.
Crossref
80 Singh S, Handschuh T. Building cell and gene therapy infrastructure in healthcare. Oliver Wyman; 2023 [cited 2024 Jul 20]. Available from: https://www.oliverwyman.com/our-expertise/insights/2023/jun/improving-cell-and-gene-therapy-infrastructure-in-healthcare.html
81 Lee D, Le AO, Meganck M, Chamberland S, Pai A. Adding a clinical hemophilia pharmacist to the hemophilia comprehensive care model improves health care-related outcomes and drug-related costs in an integrated health care system. Perm J. 2022;26(3):90–3.
Crossref PubMed PMC
82 Batty P, Lillicrap D. Advances and challenges for hemophilia gene therapy. Hum Mol Genet. 2019;28(R1):R95–R101.
Crossref PubMed
83 Saleh S, Dabbous O, Sullivan SD, Ankleshwaria D, Trombini D, Toumi M, et al. A practical approach for adoption of a hub and spoke model for cell and gene therapies in low- and middle-income countries: framework and case studies. Gene Ther. 2024;31(1–2):1–11.
Crossref PMC
84 Jiang Z, Dalby PA. Challenges in scaling up AAV-based gene therapy manufacturing. Trends Biotechnol. 2023;41(10):1268–81.
Crossref PubMed
85 Rodrigues GA, Shalaev E, Karami TK, Cunningham J, Slater NKH, Rivers HM. Pharmaceutical development of AAV-based gene therapy products for the eye. Pharm Res. 2018;36(2):29.
Crossref PubMed PMC
86 Nisanov AM, Rivera de Jesús JA, Schaffer DV. Advances in AAV capsid engineering: integrating rational design, directed evolution and machine learning. Mol Ther. 2025;33(5):1937–45.
Crossref PubMed PMC
87 Sharma S, Rawal R, Shah D. Addressing the challenges of AI-based telemedicine: best practices and lessons learned. J Educ Health Promot. 2023;12:338.
Crossref PubMed PMC
88 Haleem A, Javaid M, Singh RP, Suman R. Telemedicine for healthcare: capabilities, features, barriers, and applications. Sens Int. 2021;2:100117.
Crossref PubMed PMC
89 Perez K, Wisniewski D, Ari A, Lee K, Lieneck C, Ramamonjiarivelo Z. Investigation into application of AI and telemedicine in rural communities: a systematic literature review. Healthcare (Basel). 2025;13(3):324.
Crossref PubMed PMC
90 Chernyi N, Gavrilova D, Saruhanyan M, Oloruntimehin ES, Karabelsky A, Bezsonov E, et al. Recent advances in gene therapy for hemophilia: projecting the perspectives. Biomolecules. 2024;14(7):854.
Crossref PubMed PMC
Address correspondence to: Dr Shane Pawluk, Children’s & Women’s Health Centre of BC, Pharmacy, Room 0B7, 4500 Oak Street, Vancouver BC V6H 3N1, email: shane.pawluk@cw.bc.ca
Competing interests: For activities not directly related to the study reported here, Blair Seifert has participated as an advisory board member for Pfizer Global and has been a member of the organizing committee for educational programs and a speaker for Pfizer Canada; and Clara Jo Hoban has received an honorarium for conference participation from the Canadian Society of Healthcare-Systems Pharmacy, is a conference co-director with the Pharmacy Technician Society of Alberta, and is a volunteer panelist reviewer with the Canadian Council on Continuing Education in Pharmacy. No other competing interests were declared.
Funding: Funding for this project was provided by Pfizer Canada ULC. Pfizer Canada ULC had no role in performing the study, interpreting the results, or preparing the manuscript.
Acknowledgements: Editorial support during the pre-submission stages was provided by STA HealthCare Communications (Montréal, Quebec), with funding from Pfizer Canada ULC.
Submitted: March 19, 2025
Accepted: July 31, 2025
Published: November 12, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 4, 2025