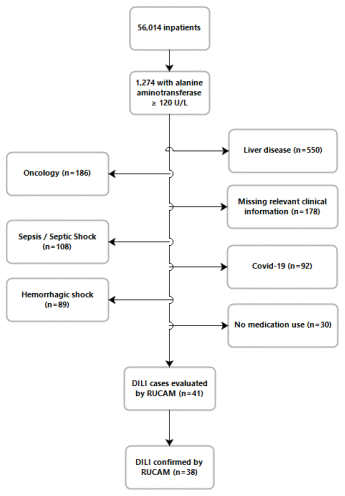
FIGURE 1 Evaluation and selection of suspected cases of drug-induced liver injury (DILI) for application of the Roussel Uclaf Causality Assessment Method (RUCAM).
Gabriela Xavier Ortiz, Karin Hepp Schwambach, Ana Helena Dias Pereira dos Santos Ulbrich, Henrique Dias Pereira dos Santos, Matheus William Becker, and Carine Raquel BlattTo cite: Ortiz GX, Schwambach KH, Dias Pereira dos Santos Ulbrich AH, Dias Pereira dos Santos H, Becker MW, Blatt CR. Pharmacist-led assessment of drug-induced liver injury in hospitalized patients. Can J Hosp Pharm. 2025;78(4):e3827. doi: 10.4212/cjhp.3827
ABSTRACT
Background
Drug-induced liver injury (DILI) is an important but underdiagnosed adverse drug reaction in hospitalized patients. The complexity of diagnosis, under-reporting, and a lack of standardized assessment tools contribute to underestimation of its prevalence.
Objectives
To estimate the prevalence of DILI in a tertiary care hospital and to discuss the role of clinical pharmacists in using the Roussel Uclaf Causality Assessment Method (RUCAM) to detect, assess, and manage DILI.
Methods
This cross-sectional study included patients who were admitted between January and June 2022 to a tertiary care hospital in Porto Alegre, Brazil. All hospitalized patients with alanine aminotransferase (ALT) levels at least 3 times the upper limit of normal at any point during the hospital stay were screened. Suspected cases of DILI were evaluated by trained pharmacists using the RUCAM. Data were collected retrospectively from electronic medical records and analyzed using descriptive and inferential statistics.
Results
Of 56 014 patients admitted to the hospital during the study period, 1274 had elevated ALT, of whom 38 were classified by the RUCAM as having DILI. Among these, 33 (87%) experienced development of DILI during the hospital stay. Most of the patients (n = 29, 76%) were in general hospital wards, and this factor showed a significant association with occurrence of DILI. Anti-infectives were the most commonly implicated drugs (69%), specifically the combination of rifampicin, isoniazid, pyrazinamide, and ethambutol and various β-lactams. Simvastatin was also commonly implicated. None of the DILI cases were reported to the institutional pharmacovigilance system.
Conclusions
DILI remains under-recognized and under-reported in hospital settings. This study reinforces the potential role of clinical pharmacists in active surveillance using RUCAM and highlights the need for institutional strategies prioritizing the monitoring of liver function in high-risk patients, particularly on general wards.
KEYWORDS: drug-induced liver injury, hospital pharmacy, patient safety, pharmacovigilance
RÉSUMÉ
Contexte
La lésion hépatique d’origine médicamenteuse (LHOM) est une réaction indésirable importante, mais souvent sous-diagnostiquée chez les patients hospitalisés. La complexité du diagnostic, la sous-déclaration des cas et le manque d’outils d’évaluation standardisés contribuent à la sous-estimation de sa prévalence.
Objectifs
Estimer la prévalence de la LHOM dans un hôpital de soins tertiaires et discuter du rôle des pharmaciens cliniciens dans la détection, l’évaluation et la prise en charge de la LHOM à l’aide de la méthode d’évaluation de la causalité Roussel Uclaf (échelle RUCAM).
Méthodologie
Cette étude transversale comprenait des patients admis de janvier à juin 2022 dans un établissement de soins tertiaires à Porto Alegre, au Brésil. Tous les patients hospitalisés dont le taux d’alanine aminotransférase (ALT) était supérieur ou égal à trois fois la limite supérieure de la normale à un moment quelconque de l’hospitalisation ont été sélectionnés. Les cas suspects de LHOM ont été évalués à l’aide de l’échelle RUCAM par des pharmaciens formés. Les données ont été recueillies rétrospectivement à partir des dossiers médicaux électroniques et analysées à l’aide de statistiques descriptives et inférentielles.
Résultats
Sur les 56 014 patients admis à l’hôpital pendant la période de l’étude, les taux d’ALT de 1274 patients étaient élevés, dont 38 qui ont été classés comme ayant une LHOM selon l’échelle RUCAM. Parmi ceux-ci, 33 patients (87 %) ont développé une LHOM au cours de leur séjour. La majorité des patients (n = 29, 76 %) se trouvaient dans des unités d’hospitalisation générale. Ce facteur a démontré une association significative avec la survenue de la LHOM. Les médicaments anti-infectieux étaient les plus fréquemment associés à cette dernière (69 %), en particulier la combinaison rifampicine, isoniazide, pyrazinamide et éthambutol, ainsi que divers β-lactamines. La simvastatine y était, elle aussi, fréquemment associée. Aucun des cas de LHOM n’a été signalé au système institutionnel de pharmacovigilance.
Conclusions
La LHOM demeure sous-reconnue et sous-déclarée en milieu hospitalier. Cette étude renforce le rôle potentiel des pharmaciens cliniciens dans la surveillance active à l’aide de l’échelle RUCAM et souligne la nécessité de stratégies institutionnelles visant à prioriser le suivi de la fonction hépatique chez les patients à haut risque, en particulier dans les unités d’hospitalisation générales.
Mots-clés: lésion hépatique d’origine médicamenteuse, pharmacie d’hôpital, sécurité du patient, pharmacovigilance
Adverse drug reactions (ADRs) are unintentionally harmful outcomes with negative effects on mortality, length of stay in hospital, and the costs of health care services. Drug-induced liver injury (DILI), caused by hepatotoxic medications, is the leading cause of withdrawal of drugs from the market.1 In the United States, DILI accounts for 13% of cases of acute liver failure.1 Although rare, DILI may be underestimated due to under-reporting and reliance on retrospective studies.2
The incidence of DILI among hospitalized patients varies by region and population: 6.6 per 1000 admissions per week in France, 3.1% in Turkey, and 1.4% in Switzerland.3–5 A Brazilian systematic review identified 32 cases, with nearly one-third involving drugs included on the Brazilian National List of Essential Medicines (also known as RENAME).6
DILI is classified as intrinsic (predictable, dose-dependent) or idiosyncratic (unpredictable), with hepatocellular, cholestatic, and mixed patterns.2 It may be asymptomatic or may present with symptoms such as jaundice, nausea, abdominal pain, and vomiting. Most patients recover spontaneously, although severe outcomes (including liver transplant or death) may occur.7,8 The diagnosis of DILI relies on exclusion of other liver diseases and consideration of drug exposure, treatment, symptom onset, and evolution of liver markers.2 The Roussel Uclaf Causality Assessment Method (RUCAM) is the tool recommended to assess liver injury from drugs or medicinal plants, as it shows greater sensitivity and specificity in this context than the Naranjo algorithm.9
Identifying DILI remains a challenge for health care professionals due to its complex clinical presentation and the requirement for a diagnosis of exclusion. Even when DILI is identified, the quality of evidence in published case reports is often questionable, as many lack a causality assessment with RUCAM, and many do not report the minimum data required for diagnosis.10,11
Substantial gaps remain in terms of detection, diagnosis, and treatment of DILI worldwide. In this study, we aimed to estimate the prevalence of DILI in a Brazilian tertiary care hospital using RUCAM and to discuss the role of clinical pharmacists in the detection, assessment, and management of DILI cases.
This retrospective cross-sectional study was based on a review of electronic medical records (EMRs). Data were collected retrospectively for patients admitted between January and June 2022 to a tertiary care hospital in Porto Alegre, Brazil. This 1300-bed facility offers specialized services, including internal medicine, surgery, intensive care, oncology, cardiology, and organ transplant. It is a recognized referral centre for the treatment of liver diseases and is affiliated with a federal university, serving as a teaching hospital for medical education. Services are provided to a primarily urban population, with more than 60% of the services delivered through Brazil’s public health system, known as the Sistema Único de Saúde or SUS.
All patients, regardless of age, with at least one alanine aminotransferase (ALT) result greater than or equal to 3 times the upper limit of normal (i.e., ≥ 120 U/L) were included, regardless of whether the ALT level subsequently returned to normal. This ALT cut-off was used to avoid missing patients with mild or asymptomatic DILI. For example, a threshold of ALT greater than 5 times the upper limit of normal accompanied by changes in total bilirubin and international normalized ratio (INR) could lead to selection bias. The 6-month study period was based on ensuring data availability during the research timeframe, and the included patients therefore represent a convenience sample.
Data extraction and RUCAM assessment were performed independently by 2 clinical pharmacists (G.X.O. and K.H.S.), who hold master’s and doctoral degrees in hepatology, respectively. Both received structured training on the standardized application of RUCAM, including case-based simulations. Disagreements were resolved by consensus. A third expert—an experienced pharmacy professor (C.R.B.) with a doctoral degree in pharmacy focused on hepatology and tutor of the clinical pharmacy residence program at the study hospital—was available for arbitration.
Patients with liver diseases unrelated to drug exposure (e.g., cirrhosis, viral hepatitis, alcoholic hepatitis, hepatocellular carcinoma, autoimmune hepatitis, cytomegalovirus infection, cholangitis, or choledocholithiasis) and those with conditions known to affect transaminase levels (e.g., COVID-19, hemorrhagic shock, septic shock, cardiogenic shock, or hemolytic disease) were excluded from RUCAM assessment. These clinical conditions, clearly documented in the EMR, were identified through the primary diagnostic code (International Statistical Classification of Diseases and Related Health Problems, 10th revision [ICD-10]) registered in the EMR. Oncology patients were excluded, as their treatment followed a distinct pharmacovigilance protocol for antineoplastics.
For patients with suspected DILI, the RUCAM was applied for each medication in use, regardless of whether the drug was initiated before or during the hospital stay. The RUCAM assesses 7 domains8,12: the interval between drug initiation and onset of liver injury, with specific scoring based on latency patterns; the course of liver enzymes—primarily ALT—following drug withdrawal, where a significant decline supports causality; the presence of specific risk factors that increase susceptibility to DILI, such as age 55 years or older and chronic alcohol use; concomitant use of other hepatotoxic drugs, which may decrease the attribution of causality; exclusion of alternative nondrug causes of liver injury; available literature or regulatory data supporting the hepatotoxicity of the drug; and response to unintentional or intentional re-exposure, which may confirm causality if a similar liver injury recurs upon rechallenge. This structured assessment allows differentiation between long-term medications having low temporal association with the liver injury and those more likely to cause such injury.
In addition, the RUCAM was not applied in cases with insufficient clinical data, which could compromise the reliability of the causality assessment. Specifically, a minimum of 3 domains were considered essential for valid application of the RUCAM: a complete medication history with documented start and stop dates in relation to onset of liver injury, laboratory evidence of elevated transaminases, and exclusion of alternative causes.8,9 Patients whose EMRs lacked data for any of these domains were excluded before application of the RUCAM. The remaining 4 domains (specifically hepatotoxicity of concomitant medications, risk factors, known hepatotoxicity of the drug, and recurrent injury upon re-exposure) were considered less critical, as their absence did not preclude assessment and they could be scored as zero.
The RUCAM score ranges from −9 to +14, with causality classified as follows: score ≤ 0, causality excluded; score 1 or 2, causality unlikely; score 3–5, causality possible; score 6–8, causality probable; and score > 8, causality highly probable. DILI was defined as a score of 3 or higher.11 The pattern of liver injury was classified as hepatocellular, cholestatic, or mixed according to the ratio of ALT to alkaline phosphatase (AP): ALT/AP ≥ 5, liver injury classified as hepatocellular; 2 ≤ ALT/AP, liver injury classified as cholestatic; 2 < ALT/AP < 5, liver injury classified as mixed.8
Data collected from the EMR included age, gender, hospital ward, medical history, symptoms, medications, hospital laboratory tests, imaging tests, self-reported alcohol consumption, and biopsy results (if available), as well as length of stay, time of recovery from DILI (if applicable), and final clinical outcomes. Drugs with no known hepatotoxicity (according to LiverTox13 or the medication label), used at standard doses, were excluded from the RUCAM, unless they were the sole drug used by the patient, with timing strongly suggestive of DILI.
Statistical analysis was performed using SPSS Statistics, version 25.0 (IBM). The nonparametric Mann–Whitney test was applied for variables with non-normal distribution, and the χ2 test with adjusted residuals was used for categorical variables. The study was approved by the Ethics Committee of the Hospital Nossa Senhora da Conceição, registered on the Brazilian study platform (number 4.763.390 CAAE 46652521.9.0000.5530) and conducted in accordance with the ethical principles outlined in the Declaration of Helsinki.
A total of 56 014 patients were admitted to the hospital from January to June 2022. Of these, 1274 patients had at least one instance of ALT at least 3 times the upper limit of normal (i.e., ≥ 120 U/L) at any time during the hospital stay (Figure 1). After exclusions, RUCAM was applied in 41 cases, with 38 patients classified as having possible, probable, or highly probable DILI for at least one of their medications.
|
| ||
|
FIGURE 1 Evaluation and selection of suspected cases of drug-induced liver injury (DILI) for application of the Roussel Uclaf Causality Assessment Method (RUCAM). | ||
The prevalence of DILI was 7 cases per 10 000 hospitalized patients. The median length of stay was 20 days (minimum 3 days, maximum 270 days) (Table 1); this extreme range reflects the presence of some outliers with prolonged length of stay. In most cases, peak ALT values ranged from 200 to 400 U/L, more than 5 times the upper limit of normal. Among the 38 patients with RUCAM-based DILI, 20 (53%) were male and 18 (47%) were older than 55 years of age (most between 65 and 75 years), which highlights the prominent presence of older adults in the cohort. Most patients were in the general hospital wards (n = 29, 76%), followed by the intensive care unit (ICU) (n = 6, 16%) and the emergency department (n = 3, 8%).
TABLE 1 Characteristics of Patients with Drug-Induced Liver Injury (n = 38)
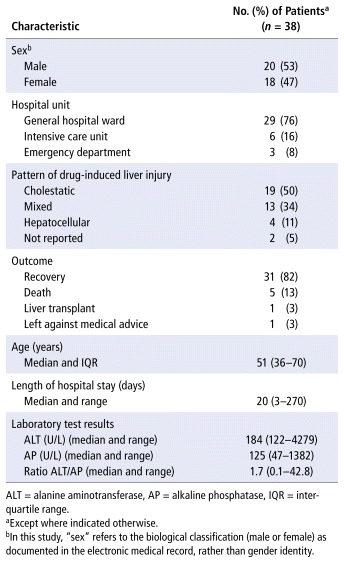
In 5 of the 38 cases, DILI was documented in the EMR as the reason for hospital admission, based on physicians’ clinical diagnosis, without formal application of the RUCAM. In the remaining cases, pharmacists inferred in-hospital DILI based on the clinical data and the RUCAM assessment. Most patients (n = 31, 82%) recovered after discontinuation of the suspected drug. One patient, whose DILI was attributed to paracetamol (acetaminophen) and codeine, needed a liver transplant. Five patients died, but their deaths were not directly linked to DILI in the EMR.
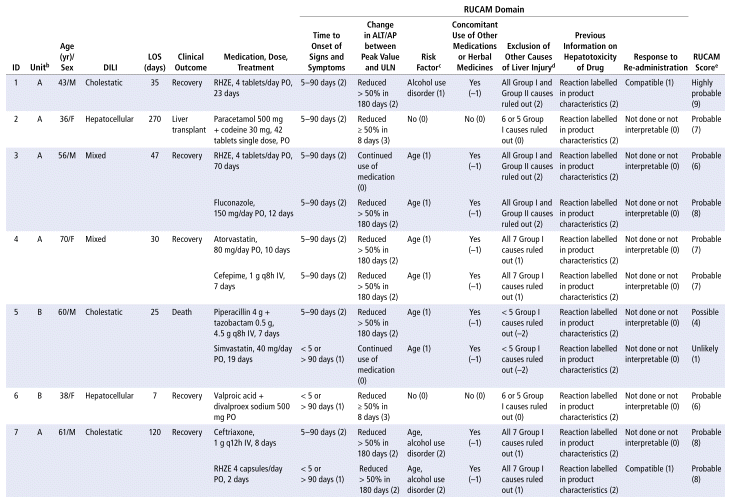
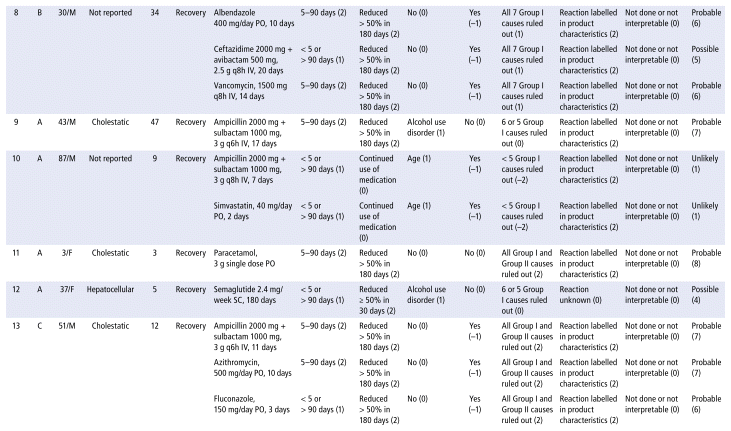
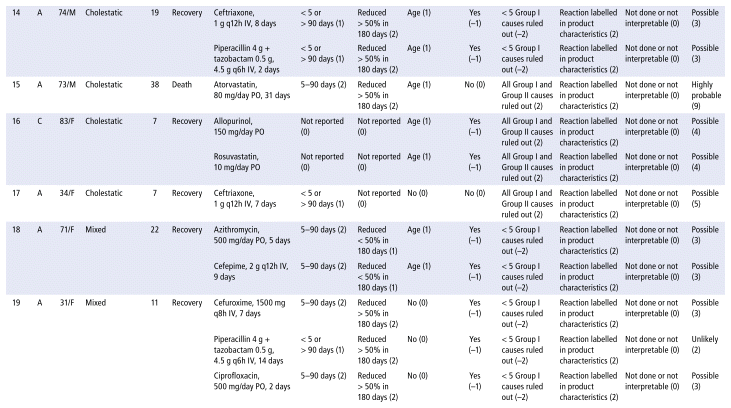
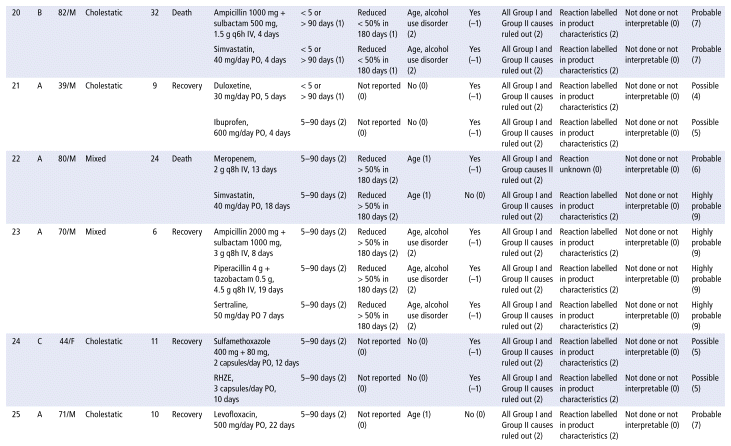
As shown in Table 2, the main drug classes involved in cases of DILI were anti-infectives (69%), followed by nervous system (14%) and cardiovascular agents (10%). The 5 most frequent drugs (or drug combinations) were ampicillin/sulbactam; piperacillin/tazobactam; the combination of rifampicin, isoniazid, pyrazinamide, and ethambutol (RHZE); ceftriaxone; and simvastatin. Receipt of care in the general hospital ward was significantly associated with DILI. Table 3 presents the results of the statistical association tests. The complete results of the causality assessment through RUCAM, showing drugs, doses, and outcomes, appear in Appendix 1.
TABLE 2 List of Medications by Therapeutic Class and Hepatotoxicity Classification for Patients with Drug-Induced Liver Injury in a Tertiary Care Hospital in Southern Brazil
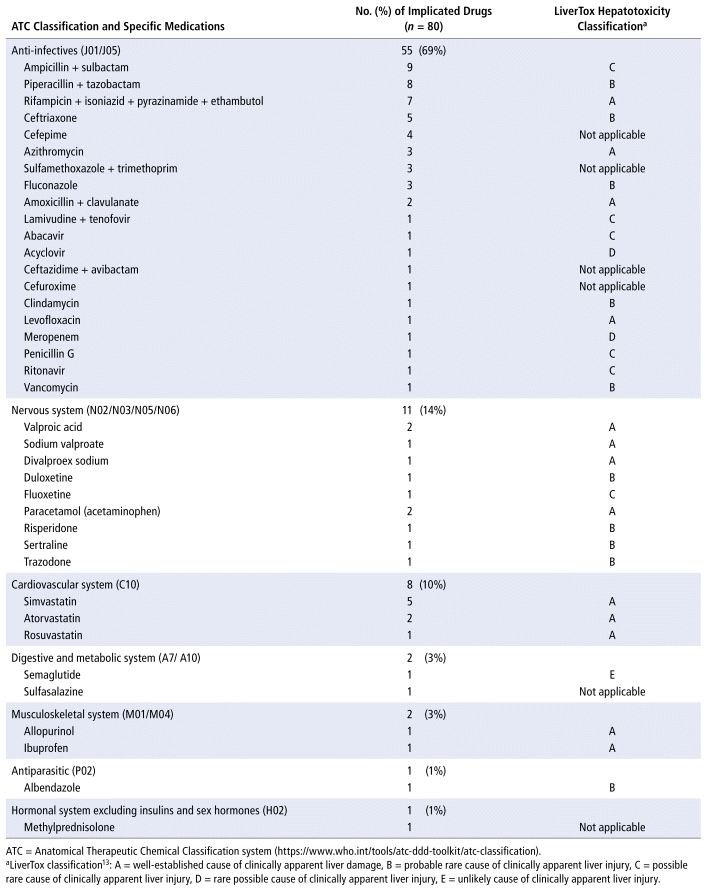
TABLE 3 Association of Drug-Induced Liver Injury with Selected Factors
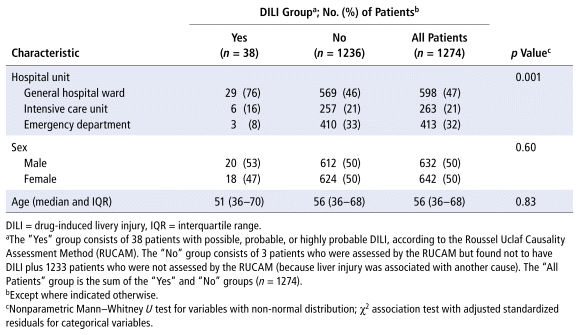
In this study, 41 cases of potential DILI were evaluated with the RUCAM, of which 38 were deemed to represent possible, probable, or highly probable DILI. Among these 38 cases, only 5 had DILI explicitly recorded in the EMR, though without formal RUCAM assessment at the time of care. In the remaining 33 cases (87%), DILI was identified retrospectively and likely developed during the hospital stay; this situation underscores that when DILI is not the reason for admission, it is rarely suspected by health care professionals. These findings highlight the need for institutional strategies that prioritize monitoring of liver function in patients receiving high-risk medications, particularly in hospital wards where such hepatotoxic agents are commonly used, and indicate an opportunity for pharmacist surveillance. A significant association was observed between DILI and receipt of care in general hospital wards (as opposed to the ICU or emergency department). This may reflect reduced clinical and laboratory monitoring in general wards compared to ICUs, despite similar or greater exposure to polypharmacy and hepatotoxic drugs. Patients in the ICU may also be excluded from RUCAM assessment or score lower on the RUCAM because of conditions like shock or sepsis, which independently cause liver injury and confound DILI attribution. The prevalence of 7 cases for every 10 000 hospitalized patients is lower than has been observed in other studies3,5; this may reflect our assessment of DILI only in cases with complete clinical information needed to apply the RUCAM.
Teschke and Danan10 highlighted the risk of DILI misdiagnosis in a review of 46 drugs listed in LiverTox. Key shortcomings included lack of a standardized DILI definition, absence of essential clinical data, and absence of validated causality assessment tools like the RUCAM. The result was diagnoses based on subjective clinical judgment. Lower methodological rigour in establishing causality may lead to overestimation of DILI prevalence, potentially increasing the number of false-positive cases and resulting in unnecessary clinical interventions and avoidable health care costs.14
The RUCAM is the most validated tool for DILI causality assessment and is endorsed by international guidelines such as those of the Council for International Organizations of Medical Sciences2 and the European Association for the Study of the Liver.7 In their 2020 review, Teschke and Danan12 reported use of RUCAM in 81 856 published cases, reinforcing its global acceptance. Its structured scoring system ensures standardization, reproducibility, and ease of use. Although some domains, such as exclusion of alternative causes and re-exposure, may be limited by information bias—especially in retrospective studies—RUCAM remains the preferred method. Therefore, we strongly recommend consistent application of RUCAM by hospital pharmacists when DILI is suspected.
No significant associations were found between occurrence of DILI and gender, age, or alcohol use, factors often cited as risks in other studies.2,5 This discrepancy may reflect population-specific characteristics, our sample size, or the multifactorial nature of DILI in hospitalized patients. Antibiotics such as ampicillin/sulbactam, piperacillin/tazobactam, RHZE, and ceftriaxone were the most commonly implicated agents, consistent with their known potential for cholestatic livery injury.15,16
In a review of 3312 cases of DILI evaluated by RUCAM, the predominant drugs were amoxicillin/clavulanate, atorvastatin, diclofenac, and simvastatin.17 Notably, IV amoxicillin/clavulanate—one of the agents most frequently implicated in DILI globally—was absent from our analysis, because it is not included in the hospital’s formulary. This does not diminish its therapeutic value but highlights how institutional formularies shape local DILI profiles and emphasizes the need for pharmacovigilance aligned with local prescribing patterns.
The RHZE regimen, widely used as first-line therapy for tuberculosis, ranks among the leading causes of DILI.18 The incidence of hepatotoxicity associated with the RHZE regimen has been reported to range from 2% to 28%, reflecting its well-established hepatotoxic potential.19 In our study, 7 (18%) of the 38 RUCAM-based DILI cases involved the RHZE regimen, which aligns with previously reported rates and reinforces the need for close monitoring of liver function during treatment. Guidelines recommend discontinuation of this regimen based on transaminase thresholds and symptomatology, thus addressing the risk–benefit of drug use.20 In our study, one patient required a liver transplant following paracetamol/codeine intoxication, with a peak ALT of 4279 U/L. Although predictable and widely recognized, this type of ADR still occurs.21 Pharmacists may play a role in promoting rational use of over-the-counter medication through patient education.21
Health care professionals should consider DILI in patients with unexplained liver injury.22 Elevated ALT remains one of the main alerts for detecting this ADR. Recent studies have suggested that liver markers, combined with structured assessments such as RUCAM, can support the development of automated screening systems in hospitals.23 While transaminase levels are useful predictors of DILI, they must be interpreted in the context of the patient’s medical history and overall clinical presentation to ensure diagnostic specificity and sensitivity.11,18 In other words, ALT should serve as a trigger for further investigation, not as an isolated marker.2 In clinical practice, pharmacists should also evaluate total bilirubin, INR, and AP to help define the pattern, severity, and possible alternative causes of liver injury.2,7
Polypharmacy and drug interactions are potential contributors to DILI, especially when agents metabolized by cytochrome P450 enzymes are involved,24,25 such as the cases in our study involving concomitant use of antiretroviral and tuberculostatic drugs. While the RUCAM method adjusts the DILI score downward when multiple drugs are implicated, the theoretical risk of synergistic hepatotoxicity justifies close monitoring in patients exposed to complex regimens.
Nearly half of the cases with elevated ALT were excluded because the DILI could have been due to pre-existing liver disease, a finding that would be expected in a hepatology referral centre. Moreover, the inability to assess 178 patients because of incomplete EMRs highlights a critical limitation of retrospective analyses such as this one. Missing data included prior medication use, alcohol consumption, and medical history (documented as free-text fields in the EMR, which may have limited completeness of the information). In addition, for many patients, viral hepatitis serology results were unavailable during the hospital stay, echoing challenges frequently reported in pharmacovigilance research.26
Under-reporting of confirmed DILI cases to the hospital’s pharmacovigilance system was observed, indicating an opportunity for pharmacist-led education and enhanced surveillance efforts. Factors such as diagnostic complexity, limited awareness of DILI among health care professionals, and passive reporting mechanisms may contribute to this situation.27,28 Strengthening pharmacists’ involvement in pharmacovigilance programs could improve case detection and reporting.
DILI is a rare but significant ADR with considerable implications for patients’ prognosis in the hospital setting. Over the 6-month period of this study, 38 RUCAM-based cases of DILI were observed, 33 of which were linked to medications commonly used in the hospital setting, underscoring the importance of this ADR for patient safety. Antibiotics, particularly the RHZE regimen, remained the most frequently implicated drugs in DILI within the Brazilian context. The absence of comprehensive clinical data in many EMRs hindered effective patient management and early ADR detection.
Clinical pharmacists can play a pivotal role in addressing this issue by promoting DILI education, conducting medication reconciliation, monitoring high-risk patients on the basis of prioritization scores and pharmaceutical consultations, providing support in choosing evidence-based treatment, and ensuring consistent use of RUCAM in suspected cases of DILI.27 In China, a university hospital implemented a pharmaceutical consultation service (known as PAC) for suspected cases of DILI. Clinical pharmacists applied a standardized protocol that included the RUCAM. After 180 days, recovery was higher in the PAC group than in the non-PAC group (96% vs. 86%), an improvement of 10 percentage points with the intervention.29 However, the approach demanded substantial manual effort for case identification.
To overcome this challenge, pharmacists must work on new forms of systematic detection of DILI using various technologies, including the EMR. Manual patient assessment is laborious and time-consuming, which makes it an impractical method, in hospitals with limited human and financial resources, for identifying ADRs that are considered rare. The use of artificial intelligence and machine learning could improve this scenario. It is possible to apply machine learning algorithms by automatically and prospectively tracking DILI in the EMR and hospital data sets through relevant laboratory tests, risk factors, ICD-10 codes for hepatic injury, and data mining.30,31
Francisco and others32 reported that inpatients with polypharmacy, prolonged length of stay, and use of systemic antibacterials were more likely to require pharmaceutical interventions. This finding highlights the importance of pharmacists in reviewing prescriptions, identifying potentially hepatotoxic drug combinations, and participating in multidisciplinary rounds to recommend timely adjustments.
This study had limitations due to its retrospective design and the incompleteness of EMR data, which may have affected RUCAM scores and the prevalence of DILI observed. The sample size and various hospital-specific factors limit generalizability. Cancer patients, who are at high risk for DILI, were not included because of operational constraints. ALT was used exclusively as the laboratory trigger for identifying DILI cases. The results of other laboratory tests, such as total bilirubin, INR, and AP, were not consistently available in patients’ EMRs and therefore were not used as triggers in this retrospective analysis. Critically ill patients may have been excluded or may have had lower RUCAM scores due to the multifactorial nature of liver injury (leading to potential underestimation of DILI). The high number of inpatient beds in the study facility may also have influenced case distribution, although this factor was not assessed quantitatively. Future analyses will compare elevated ALT among patients with and without DILI who are exposed to drugs suspected of causing this type of injury. Despite these limitations, this study advances understanding of DILI prevalence and highlights opportunities for detection and management by pharmacists.
In this study, 38 cases of DILI in a tertiary care hospital were confirmed by the RUCAM over a 6-month period, with most cases occurring during the hospital stay and none reported to the institutional pharmacovigilance system. These findings highlight significant under-reporting, and our insights reinforce the opportunity for active surveillance of DILI by clinical pharmacists using validated tools such as the RUCAM. The most frequently implicated drugs were commonly used antibiotics, especially the RHZE regimen and β-lactams. Given that 82% of patients recovered after discontinuation of the implicated drug, we conclude that timely detection can prevent complications. Institutional strategies should prioritize monitoring of liver function in patients who are receiving high-risk medications, particularly in hospital wards where such agents are commonly used.
1 Reuben A, Tillman H, Fontana RJ, Davern T, McGuire B, Stravitz RT, et al. Outcomes in adults with acute liver failure between 1998 and 2013: an observational cohort study. Ann Intern Med. 2016;164(11): 724–32.
Crossref PubMed PMC
2 Drug-induced liver injury (DILI): current status and future directions for drug development and the post-market setting. A consensus by a CIOMS working group. Council for International Organizations of Medical Sciences (CIOMS); 2020 [cited 2024 Jan 18]. Available from: https://cioms.ch/wp-content/uploads/2020/06/CIOMS_DILI_Web_16Jun2020.pdf
3 Bagheri H, Michel F, Lapeyre-Mestre M, Lagier E, Cambus JP, Valdiguié P, et al. Detection and incidence of drug-induced liver injuries in hospital: a prospective analysis from laboratory signals. Br J Clin Pharmacol. 2000;50(5):479–84.
Crossref PubMed PMC
4 Idilman R, Bektas M, Cinar K, Toruner M, Cerit ET, Doganay B, et al. The characteristics and clinical outcome of drug-induced liver injury: a single-center experience. J Clin Gastroenterol. 2010;44(6):e128–32.
Crossref PubMed
5 Meier Y, Cavallaro M, Roos M, Pauli-Magnus C, Folkers G, Meier PJ, et al. Incidence of drug-induced liver injury in medical inpatients. Eur J Clin Pharmacol. 2005;61(2):135–43.
Crossref PubMed
6 Becker MW, Lunardelli MJM, Tovo CV, Blatt CR. Drug and herb-induced liver injury: a critical review of Brazilian cases with proposals for the improvement of causality assessment using RUCAM. Ann Hepatol. 2019;18(5):742–50.
Crossref PubMed
7 European Association for the Study of the Liver; Andrade RJ, Aithal GP, Björnsson ES, Kaplowitz N, Kullak-Ublick GA, Larrey D, et al. EASL clinical practice guidelines: drug-induced liver injury. J Hepatol. 2019;70(6):1222–61.
Crossref
8 Danan G, Teschke R. RUCAM in drug and herb induced liver injury: the update. Int J Mol Sci. 2015;17(1):14.
Crossref PubMed PMC
9 Danan G, Teschke R. Roussel Uclaf Causality Assessment Method for drug-induced liver injury: present and future. Front Pharmacol. 2019; 10:853.
Crossref PubMed PMC
10 Teschke R, Danan G. The LiverTox paradox—gaps between promised data and reality check. Diagnostics (Basel). 2021;11(10):1754.
Crossref PubMed PMC
11 Danan G, Teschke R. Drug-induced liver injury: why is the Roussel Uclaf Causality Assessment Method (RUCAM) still used 25 years after its launch? Drug Saf. 2018;41(8):735–43.
Crossref PubMed
12 Teschke R, Danan G. Worldwide use of RUCAM for causality assessment in 81,856 idiosyncratic DILI and 14,029 HILI cases published 1993-mid 2020: a comprehensive analysis. Medicines (Basel). 2020; 7(10):62.
PubMed PMC
13 LiverTox: clinical and research information on drug-induced liver injury. National Institute of Diabetes and Digestive and Kidney Diseases (US); 2012-[cited 2025 Oct 14]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547852/?
14 Carroll AE. The high costs of unnecessary care. JAMA. 2017;318(18): 1748–9.
Crossref PubMed
15 Sundaram V, Björnsson ES. Drug-induced cholestasis. Hepatol Commun. 2017;1(8):726–35.
Crossref
16 Ampicillin-sulbactam. In: LiverTox: clinical and research information on drug-induced liver injury. National Institute of Diabetes and Digestive and Kidney Diseases (US); 2012-[updated 2020 Oct; cited 2024 Jan 18]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547872/
17 Teschke R. Top-ranking drugs out of 3312 drug-induced liver injury cases evaluated by the Roussel Uclaf Causality Assessment Method. Expert Opin Drug Metab Toxicol. 2018;14(11):1169–87.
PubMed
18 Becker MW, Schwambach KH, Lunardelli M, Blatt CR. Overview of drug induced liver injury in Brazil: what is the role of public health policy on the evidence? World J Gastrointest Pharmacol Ther. 2021;12(3):40–55.
Crossref PubMed PMC
19 Yew WW, Chang KC, Chan DP. Oxidative stress and first-line antituberculosis drug-induced hepatotoxicity. Antimicrob Agents Chemother. 2018;62(8):e02637–17.
PubMed PMC
20 Secretaria de Vigilância em Saúde, Departamento de Vigilância das doenças transmissíveis. Manual de recomendações para o controle da tuberculose no Brasil [Guidelines for tuberculosis control in Brazil]. Ministério da saúde (Brazil); 2019. Publication in Portuguese.
21 Rotundo L, Pyrsopoulos N. Liver injury induced by paracetamol and challenges associated with intentional and unintentional use. World J Hepatol. 2020;12(4):125–36.
Crossref PubMed PMC
22 Chalasani NP, Maddur H, Russo MW, Wong RJ, Reddy KR; Practice Parameters Committee of the American College of Gastroenterology. ACG clinical guideline: diagnosis and management of idiosyncratic drug-induced liver injury. Am J Gastroenterol. 2021;116(5):878–98.
Crossref PubMed
23 Tan EH, Ling ZJ, Ang PS, Sung C, Dan YY, Tai BC. Comparison of laboratory threshold criteria in drug-induced liver injury detection algorithms for use in pharmacovigilance. Pharmacoepidemiol Drug Saf. 2020;29(11):1480–8.
Crossref PubMed
24 Li M, Luo Q, Tao Y, Sun X, Liu C. Pharmacotherapies for drug-induced liver injury: a current literature review. Front Pharmacol. 2022; 12:806249.
Crossref PubMed PMC
25 Yu K, Geng X, Chen M, Zhang J, Wang B, Ilic K, et al. High daily dose and being a substrate of cytochrome P450 enzymes are two important predictors of drug-induced liver injury. Drug Metab Dispos. 2014; 42(4):744–50.
Crossref PubMed
26 Sun W, Cai Z, Li Y, Liu F, Fang S, Wang G. Data processing and text mining technologies on electronic medical records: a review. J Healthc Eng. 2018;2018:4302425.
Crossref PubMed PMC
27 Güner MD, Ekmekci PE. Healthcare professionals’ pharmacovigilance knowledge and adverse drug reaction reporting behavior and factors determining the reporting rates. J Drug Assess. 2019;8(1):13–20.
Crossref PMC
28 Aung AK, Walker S, Khu YL, Tang MJ, Lee JI, Graudins LV. Adverse drug reaction management in hospital settings: review on practice variations, quality indicators and education focus. Eur J Clin Pharmacol. 2022;78(5):781–91.
Crossref PubMed PMC
29 Li D, Dong J, Xi X, Huang G, Li W, Chen C, et al. Impact of pharmacist active consultation on clinical outcomes and quality of medical care in drug-induced liver injury inpatients in general hospital wards: a retrospective cohort study. Front Pharmacol. 2022;13:972800.
Crossref PubMed PMC
30 Minerali E, Foil DH, Zorn KM, Lane TR, Ekins S. Comparing machine learning algorithms for predicting drug-induced liver injury (DILI). Mol Pharm. 2020;17(7):2628–37.
Crossref PubMed PMC
31 Yeboah-Korang A, Louissaint J, Tsung I, Prabhu S, Fontana RJ. Utility of a computerized ICD-10 algorithm to identify idiosyncratic drug-induced liver injury cases in the electronic medical record. Drug Saf. 2020;43(4):371–7.
Crossref PubMed PMC
32 Francisco DB, Dal Paz K, Didone TVN. Patient factors associated with pharmaceutical interventions for inpatients at a Brazilian teaching hospital. Can J Hosp Pharm. 2021;74(3):211–8.
Crossref PubMed PMC
Address correspondence to: Gabriela Xavier Ortiz, Department of Pharmacy / Department of Pharmaceutical Sciences, Federal University of Health Sciences of Porto Alegre (UFCSPA), Rua Sarmento Leite, 245, Centro Histórico, Porto Alegre – RS, 90050-170, Brazil, email: gabiau92@gmail.com
Competing interests: None declared.
Funding: None received.
Acknowledgements: This study received various forms of nonfinancial support from the Federal University of Health Sciences of Porto Alegre and the Santa Casa Hospital (the university’s teaching hospital and the site of data collection).
Submitted: April 21, 2025
Accepted: July 31, 2025
Published: November 12, 2025
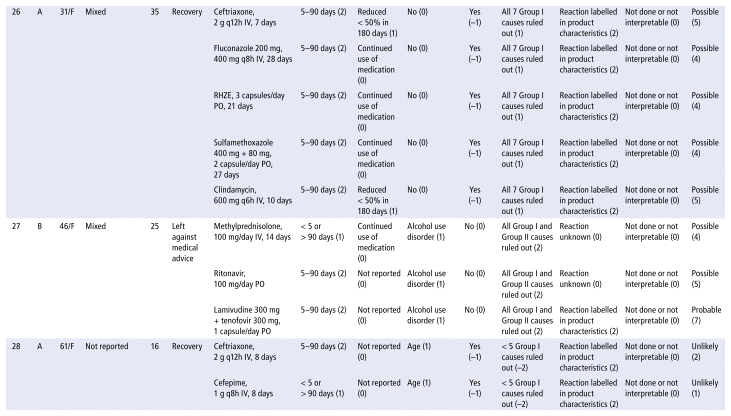
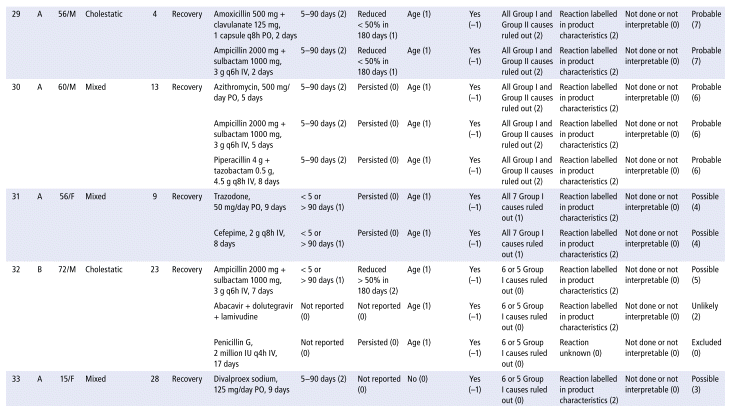
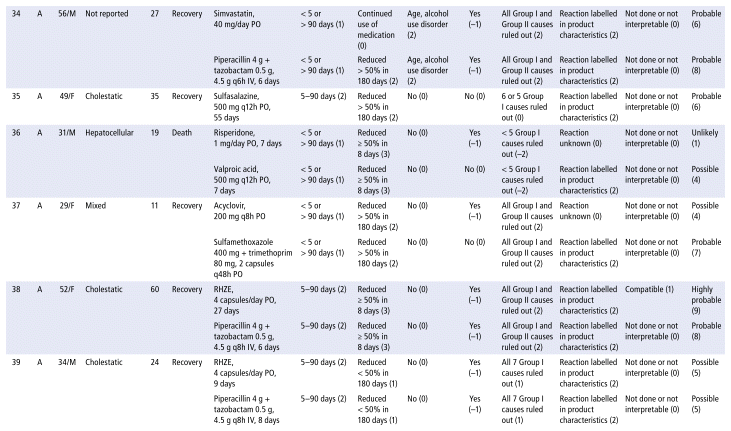
APPENDIX 1 Characteristics of the 41 cases evaluated for drug-induced liver injury (DILI), scores for the domains, and final RUCAM score.a

© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 4, 2025