Kristin Kaupp, Sarah Smit, Sarah Burgess, Emily Black, Osama Loubani, and Meghan MacKenzieTo cite: Kaupp K, Smit S, Burgess S, Black E, Loubani O, MacKenzie M. Antimicrobial administration delays for patients with suspected sepsis in a Canadian emergency department: prevalence and risk factors. Can J Hosp Pharm. 2026;79(2):e3830. doi: 10.4212/cjhp.3830
ABSTRACT
Background
For patients with sepsis, timely administration of antimicrobials is imperative, with evidence demonstrating that delays may be associated with an increase in mortality.
Objective
To determine whether antimicrobial administration is delayed among patients with sepsis admitted through the Queen Elizabeth II Health Sciences Centre Emergency Department (QEII-HSC-ED) in Halifax, Nova Scotia.
Methods
A single-centre, retrospective health records review was conducted for adult patients with sepsis admitted through the QEII-HSC-ED between January 1, 2021, and December 31, 2022. Differences between groups with and without antimicrobial administration delays were compared by univariate analysis, and an adjusted multivariate regression was completed to examine associations between risk factors and significant delays.
Results
A total of 275 patient encounters were included in the analysis, accounting for a total of 1208 antimicrobial doses: 275 first doses and 933 subsequent doses. Of the 275 patient encounters, 216 (78.5%) had at least one significant dose delay; 135 (49.1%) of the encounters had delay of the first dose, and 169 (61.5%) had delay of a subsequent dose. Of the 933 subsequent doses administered, 276 (29.6%) had a significant delay. Relative to patients admitted to a medicine service, surgical patients had reduced odds of experiencing significant delay of a subsequent dose (odds ratio 0.25, 95% confidence interval 0.08–0.77).
Conclusions
Many patients with sepsis admitted through the QEII-HSC-ED experienced significant delays in administration of first and/or subsequent doses of antimicrobial therapy, which may have increased their risk of negative outcomes. These results can be used to develop future initiatives aimed at improving time to administration of antimicrobials for patients with sepsis admitted to the study institution.
KEYWORDS: sepsis, septic shock, antimicrobials, antibiotics, timely administration
RÉSUMÉ
Contexte
Il est impératif d’administrer en temps voulu des antimicrobiens aux patients atteints d’une septicémie : des données probantes montrent en effet que les retards peuvent être associés à une augmentation de la mortalité.
Objectif
Déterminer si l’administration des antimicrobiens est retardée chez les patients atteints d’une septicémie admis à l’urgence du Queen Elizabeth II Health Sciences Centre à Halifax (Nouvelle-Écosse) (QEII-HSC).
Méthodologie
Une revue rétrospective des dossiers médicaux menée dans un centre unique a été réalisée auprès de patients adultes atteints d’une septicémie admis à l’urgence du QEII-HSC entre le 1er janvier 2021 et le 31 décembre 2022. Les différences entre les groupes chez qui les antimicrobiens avaient été administrés en retard et en temps voulu ont été comparées au moyen d’analyses univariées; une régression multivariée ajustée a ensuite été effectuée afin d’examiner les associations entre les facteurs de risque et les retards significatifs.
Résultats
Au total, 275 rencontres avec les patients ont été incluses dans l’analyse, représentant un total de 1208 doses d’antimicrobiens administrées : 275 premières doses et 933 doses subséquentes. Sur les 275 rencontres, 216 (78,5 %) se caractérisaient par au moins un retard d’administration significatif; 135 (49,1 %) se caractérisaient par un retard d’administration de la première dose et 169 (61,5 %) se caractérisaient par un retard d’administration d’une dose subséquente. Sur les 933 doses subséquentes administrées, 276 (29,6 %) l’ont été avec un retard significatif. Par rapport aux patients admis dans un service de médecine, la probabilité qu’une dose subséquente soit administrée aux patients chirurgicaux avec un retard significatif (rapport de cotes 0,25; intervalle de confiance à 95 % : 0,08–0,77) était moins élevée.
Conclusions
L’administration tant de la première dose que des doses subséquentes d’antimicrobiens à de nombreux patients atteints d’une septicémie admis à l’urgence du QEII-HSC se caractérisait par des retards significatifs, ce qui pourrait avoir accru le risque de résultats défavorables. Ces résultats peuvent servir à élaborer de futures initiatives visant à améliorer les délais d’administration des antimicrobiens aux patients atteints d’une septicémie admis dans l’établissement étudié.
Mots-clés: septicémie, choc septique, antimicrobiens, antibiotiques, administration rapide
Sepsis, defined as life-threatening organ dysfunction caused by a dysregulated host response to infection, affects millions globally and carries a significant risk of death.1 Timely administration of antimicrobials is imperative, with evidence demonstrating an increase in mortality associated with delay in antimicrobial administration.2,3 Research concerning time to administration of antimicrobials in patients with sepsis has focused primarily on the time to administration of the first dose.2 However, research also shows that delays in administration of second doses of antimicrobials in patients with sepsis are associated with increases in hospital mortality, length of stay, and need for mechanical ventilation.4,5 Various studies have found the following factors to be associated with first-to-second dose delays of antimicrobials: shorter administration intervals,4,6 inpatient boarding in the emergency department (ED),4,6,7 initial 3-hour bundle compliance,4 younger age,7 older age,4 receipt of 30 mL/kg of fluid in the first 3 hours,6 renal failure,6 high-acuity presentation,7 and presence of multiple medical comorbidities.7
Several systemic limitations in the Queen Elizabeth II Health Sciences Centre Emergency Department (QEII-HSC-ED) in Halifax, Nova Scotia, may affect timely medication administration, including lack of an electronic health record, lack of automated screening for sepsis, lack of formal medication administration records that include transcribed orders and future medication administration times, prolonged boarding in the ED, and overcrowding. Although commonly prescribed antimicrobials are readily accessible in the ED as ward stock, most in ready-to-administer preparations, we hypothesized that because of these limitations, patients with sepsis were likely experiencing significant delays in antimicrobial administration.
In this project, we aimed to determine whether adult patients with sepsis admitted through the QEII-HSC-ED were experiencing delays in the time to administration of antimicrobials. Secondarily, we aimed to determine whether selected patient and systemic factors were associated with antimicrobial administration delays at our institution.
This quality improvement project was a single-centre, retrospective review of health records for patients admitted between January 1, 2021, and December 31, 2022, through the ED of QEII-HSC, an adult, tertiary care teaching hospital affiliated with Dalhousie University and located in Halifax, Nova Scotia. This study was approved by the Nova Scotia Health Research and Ethics Board (no. 1029063).
All adult patients (16 years of age or older) with an admission diagnosis of sepsis, as provided by the ED physician, were included if they had received at least 2 doses of an antimicrobial regimen initiated in the QEII-HSC-ED. Patients who received only 1 dose of an antimicrobial, those whose antimicrobial therapy was intentionally discontinued and then reinitiated, those who were originally assessed at another hospital, and those with missing information about administration of antimicrobials were excluded.
The primary outcome was the prevalence of significant delays in administration of antimicrobials for the first and subsequent antimicrobial doses. The secondary outcome consisted of associations between patient or systemic factors and delays in antimicrobial administration.
Any antimicrobial dose that was administered after the recommended dosing interval was considered to have been delayed. Delays were calculated using the following formula: delay = actual interval – recommended interval. In cases of delay, the following formula was used to calculate the magnitude of the delay: (actual interval/recommended interval) × 100, expressed as a percentage. A first-dose delay was considered significant if the antimicrobial was administered more than 1 hour after it was prescribed. A first-dose delay of more than 1 hour was considered significant because guidelines recommend that patients receive antimicrobials within 1 hour of sepsis recognition, since delays may be associated with increased mortality.1–3 This definition of significant delay is also consistent with local institutional policy on medication administration, which specifies that antimicrobials are to be administered within 1 hour of an order being placed. Consistent with previous literature, delays for subsequent doses were considered significant either if the delay was greater than or equal to 125% of the recommended dosing interval4,7 (in cases where the antimicrobial remained the same or the antimicrobial was changed to an agent with a narrower spectrum of activity) or if the antimicrobial was administered more than 1 hour after being prescribed (in cases where a new antimicrobial was added or the antimicrobial was changed to an agent with a broader spectrum of activity). The recommended dosing interval for subsequent doses of an antimicrobial was defined as the prescribed interval. If there was no prescribed interval, the recommended interval was determined from local antimicrobial dosing recommendations for the indication and the patient’s estimated creatinine clearance, calculated using a modified Cockcroft–Gault equation standardized to 70 kg.8
Charts of patients with an admission diagnosis of sepsis provided by the ED physician during the study period were identified by Decision Support. These charts were then manually screened for inclusion by a research assistant (S.S.). The research assistant then collected data from the patient charts and the Nova Scotia Health intensive care unit (ICU) database using a standardized data collection form in the Research Electronic Data Capture (REDCap) application. To ensure accuracy, data from 10 charts were extracted independently by the research assistant and the principal investigator (K.K.) and compared, with discrepancies resolved by discussion and consensus.
For each patient, the data collected included medical record number, health card number, age, sex, weight, admission diagnosis, suspected primary site of infection, and vital signs at triage. Presenting serum creatinine and lactate, registration date and time, triage date and time, and Canadian Triage and Acuity Scale (CTAS) score, as well as date and time of first physician assessment, were also collected. Details of all antimicrobials administered, specifically medication name, dose, route, frequency, time ordered, and time of administration, were collected until one of the following occurred: the antimicrobial was administered on an inpatient unit, the patient was discharged home (directly from the ED), or the patient died.
The following information related to treatment was also collected: volume of IV fluids administered in the first 3 hours; use of separate orders for first and subsequent antimicrobial doses; use of the sepsis order set, which contains initial antimicrobial orders and timing recommendations; admitting service; date and time of admission; date and time of transfer to inpatient unit; and total length of time in the ED. The patient’s physical location (within the ED) at the time of each antimicrobial dose was also collected, because the level of monitoring and staffing ratios vary by location. Additional information collected included hospital length of stay, ICU utilization, ICU length of stay, need for and duration of vasopressors, need for and duration of mechanical ventilation, and need for new renal replacement therapy, as well as discharge location or date and the time and location of death (if the patient did not survive).
Patient characteristics and the primary outcome were analyzed using descriptive statistics. Differences between groups were compared using univariate analysis, with nonparametric methods for continuous variables and the χ2 or Fisher exact test for categorical variables. An adjusted multivariate regression was completed to examine associations between risk factors and significant delays. The data were analyzed using SAS software, version 9.4 (SAS Institute Inc).
All patients admitted between January 1, 2021, and December 31, 2022, who met the inclusion criteria were included. Previous research indicates that between 19.3% and 54% of patients with sepsis will experience a significant delay in the administration of the second dose of antimicrobial.4–7,9 Therefore, a minimum sample size of 200 patients was required to detect an event rate of 20% ± 10% with 90% confidence.
The data supporting the findings of this study may be available from the corresponding author upon reasonable request.
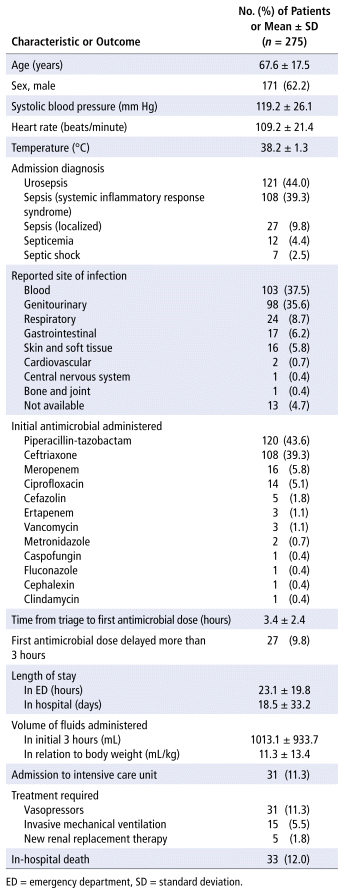
Of 319 patient encounters with an admission diagnosis of sepsis initially identified, 275 patient encounters were included in data analysis. Of the 44 encounters that were excluded, 35 involved initial assessment at another hospital, 5 involved patients who received only 1 dose of antimicrobial, and 4 had incomplete antimicrobial administration records. Patient characteristics are presented in Table 1.
TABLE 1 Patient Demographic Characteristics and Descriptive Outcomes

The 275 patient encounters were associated with a total of 1208 antimicrobial doses: 275 first doses and 933 subsequent doses. The sepsis order set was not used for any of the patient encounters. Most of the patients (n = 239, 86.9%) were triaged as having a CTAS score of 1 or 2. Most patients (n = 244, 88.7%) received initial broad-spectrum antimicrobial therapy with either piperacillin–tazobactam (n = 120, 43.6%), ceftriaxone (n = 108, 39.3%), or meropenem (n = 16, 5.8%). Of the 275 patient encounters included in the analysis, 216 (78.5%) had at least one significant dose delay: 135 (49.1%) had a first-dose delay, and 169 (61.5%) had delay of at least 1 subsequent dose. Of the 275 first doses, 27 (9.8%) were administered more than 3 hours after being ordered. Nearly one-third of subsequent doses were significantly delayed (276/933, 29.6%). The first dose received after transfer to an inpatient unit was significantly delayed in about one-quarter of cases (64/269, 23.8%).
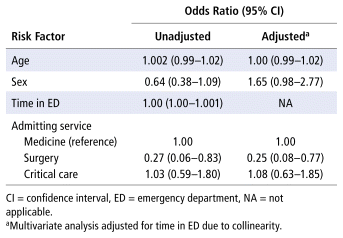
Patients who experienced a significant first-dose delay (Table 2) or a significant subsequent-dose delay (Table 3) had a statistically significant longer length of stay in the ED. Medicine patients had a longer median ED length of stay (1484 minutes) than surgical patients (708 minutes) or critical care patients (769 minutes). Admission to a surgical service reduced the odds of experiencing a significant subsequent-dose delay by 75% relative to admission to a medicine service (adjusted odds ratio [OR] 0.25, 95% confidence interval [CI] 0.08–0.77) (Table 4). No associations were found between significant first-dose delays and the first dose being a one-time order or patient location in the ED (Table 2). Patients who experienced a significant first-dose delay were not found to experience significant subsequent-dose delays more frequently or vice versa (Tables 2 and 3). Patients’ location within the ED was not associated with significant subsequent-dose delays (Table 3).
TABLE 2 Characteristics of Patients with and without Significant Delay of First Dose
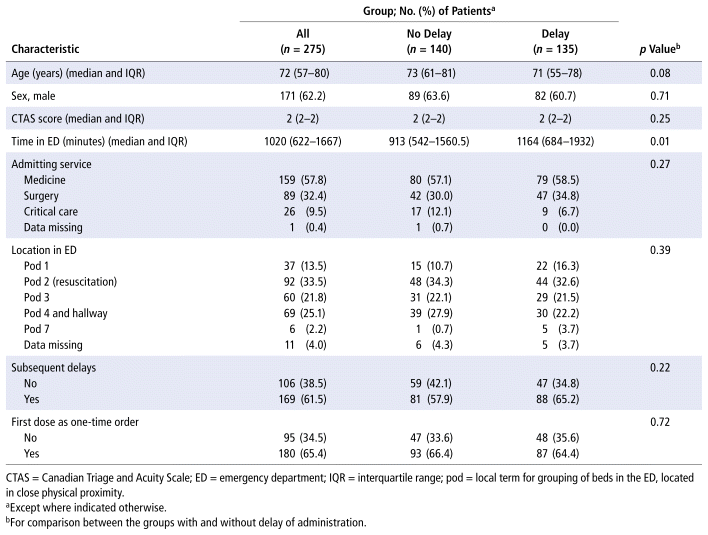
TABLE 3 Characteristics of Patients with and without Significant Delay of Subsequent Dose
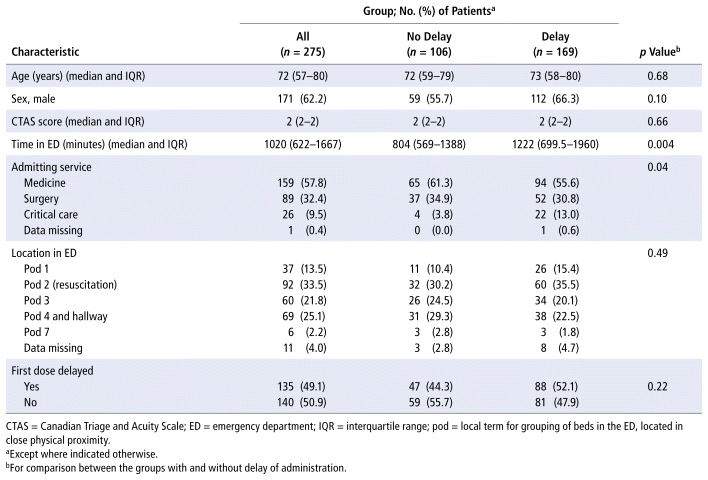
TABLE 4 Risk Factors for Significant Delay of Subsequent Doses
In this study, a concerning proportion of patients (78.5%) admitted with sepsis through the QEII-HSC-ED experienced significant delays in administration of both the first dose (49.1%) and subsequent doses (61.5%) of antimicrobials, which may have increased their risk of negative outcomes and is not in accordance with best practice. The majority of patients (86.9%) were triaged as having a CTAS score of 1 or 2, meaning they had “threats to life/limb requiring aggressive interventions” or “a potential threat to life, limb or function, requiring rapid medical intervention”,10 yet they did not receive potentially life-saving antimicrobials in a timely manner. We chose to include patients with a physician’s real-time admission diagnosis of sepsis, rather than retrospectively identifying patients with sepsis according to specific criteria, to allow examination of antimicrobial administration practices as they occurred in a real-world population of patients at our institution.
Patients who experienced a significant first-dose delay spent a median of 251 minutes longer in the ED, and those who experienced a significant subsequent-dose delay spent a median of 418 minutes longer in the ED, relative to those who did not experience such delays. Surgical patients had significantly reduced odds of experiencing a significant subsequent-dose delay. When ED length of stay (which was shorter for surgical patients) was removed from the regression analysis due to collinearity, surgical patients still had reduced odds of experiencing a significant subsequent-dose delay relative to medicine patients (OR 0.25, 95% CI 0.08–0.77). This difference in ED length of stay between patient populations may be due to the nature of their illness, as surgical patients would likely be transferred to the operating room quickly, while medicine patients may be boarded in the ED due to overcrowding. Antimicrobial administration delays could be due to several systemic limitations at the QEII-HSC-ED, such as the complete lack of use of the sepsis order set (despite the existence of such an order set), the need to rely on verbal communication to identify when new orders for antimicrobials are placed by a prescriber, and the lack of formal medication administration records in the ED to identify when subsequent doses of antimicrobials are due.
These findings are consistent with those of previous studies, which found that patients admitted with sepsis experienced significant delays in the administration of subsequent doses of antimicrobials.3–6,11 However, patients admitted with sepsis through the QEII-HSC-ED experienced significant subsequent-dose delays more frequently than patients in previous studies (61.5% vs 19.3%–54%),4–7,9 which supports the hypothesis that there may be local systemic factors at the QEII-HSC-ED contributing to this high rate of antimicrobial administration delays.
Previous studies have shown that inpatients boarded in the ED experience significant second-dose delays more frequently4,7 and that ED boarding is associated with increased odds of experiencing a significantly delayed second dose.6 The current study found that patients who experienced a significant subsequent-dose delay had a statistically significant longer ED length of stay, with patients who experienced a delay spending a median 418 minutes longer in the ED than those who did not experience a delay. Notably, the current analysis included all subsequent antimicrobial doses until the patient’s first dose on an inpatient unit, not only second doses, as in previous studies.
This study provides objective evidence that patients with sepsis admitted through the QEII-HSC-ED experienced significant antimicrobial administration delays. These results can be used to improve care for this vulnerable patient population at our institution. This study also provides a Canadian perspective, based on a publicly funded system, which may differ from studies conducted in the United States or in privately funded systems. Hospitals and health care providers working within publicly funded systems may have fewer financial or legal incentives to implement sepsis care bundles within a strict time frame; therefore, the results of previous studies investigating delays in administration of antimicrobials for patients with sepsis may not be representative of practices in publicly funded Canadian EDs.
This study had several limitations. It involved a single-centre health records review, and therefore the results may not be generalizable to other hospitals with different processes for identifying and managing patients with sepsis and for ordering and administering medications. This quality improvement project focused solely on time to administration of antimicrobials and did not examine associations between delays and patient outcomes, nor did it thoroughly review the appropriateness of antimicrobial spectrum or dosing. However, extensive literature shows that delays of antimicrobial administration in sepsis are associated with worse patient outcomes,1,2–5 and we found that most patients (88.7%) in this study had received broad-spectrum antimicrobials consistent with sepsis guidelines. Due to the retrospective design, we were limited to examining associations between patient/systemic factors and antimicrobial administration delays and could not conclude causation. The results of this study were dependent on the accuracy of documentation in handwritten patient records, and records with missing or illegible details related to antimicrobial administration had to be excluded. The study took place during the COVID-19 pandemic, and the potential effects of this context should also be considered. The volume of patients and nursing resources at the QEII-HSC-ED may have been altered during this period, and there may have been increased requirements for contact/airborne precautions. All of these factors may have affected the results of the study. However, the QEII-HSC-ED has frequently been required to operate at or above maximum capacity, with limited resources and precaution requirements. Therefore, we feel that these results are representative of and applicable to current practices at the QEII-HSC-ED.
The results of this study highlight an important patient safety issue at our institution. Timely administration of antimicrobials is imperative for patients with sepsis. Although there is conflicting evidence about the magnitude of the impact of antimicrobial administration delays in patients with sepsis, guidelines recommend that when a diagnosis of sepsis is definite or probable and the patient is in shock, antimicrobials should be administered within 1 hour of recognition.1,12,13 If the diagnosis of sepsis is less certain but concern for an infectious cause persists after initial assessment, guidelines recommend administering antimicrobials within 1 hour if shock is present or within 3 hours if shock is absent.1 Our findings demonstrate that the QEII-HSC-ED is not meeting these targets. To improve timely administration of antimicrobials, we recommend targeted education and a multidisciplinary re-rollout of the existing sepsis care directive and order set at QEII-HSC-ED, with the goal of administering the first dose of antimicrobials within 1 hour of sepsis recognition if the patient is in shock. Such initiatives have been shown to improve time to administration of antimicrobials and other important patient outcomes in patients with sepsis.11,13 We also recommend implementation of formal medication administration records in the ED to improve timely administration of subsequent doses of antimicrobials.
These results can be used as a baseline for future initiatives at our institution aimed at improving time to administration of antimicrobials in patients admitted with sepsis through the QEII-HSC-ED.11 Future quality improvement projects could also be designed to determine whether patients admitted with sepsis are experiencing delays in sepsis recognition (time from presentation to diagnosis of sepsis) and to evaluate whether patients are receiving appropriate antimicrobial treatment, given the suspected site of infection.
Patients with sepsis admitted through the QEII-HSC-ED experienced significant delays in antimicrobial administration. Notably, surgical patients were found to have reduced odds of experiencing a significant subsequent-dose delay. These findings highlight the need for implementation of improved processes and protocols, with the goal of improving timely administration of antimicrobials in patients with sepsis at the study institution.
1 Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47(11):1181–247.
Crossref PubMed PMC
2 Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34(6):1589–96.
Crossref PubMed
3 Tang F, Yuan H, Li X, Qiao L. Effect of delayed antibiotic use on mortality outcomes in patients with sepsis or septic shock: a systematic review and meta-analysis. Int Immunopharmacol. 2024;129:111616.
Crossref PubMed
4 Leisman D, Huang V, Zhou Q, Gribben J, Bianculli A, Bernshteyn M, et al. Delayed second dose antibiotics for patients admitted from the emergency department with sepsis: prevalence, risk factors, and outcomes. Crit Care Med. 2017;45(6):956–65.
Crossref PubMed
5 Taylor SP, Shah M, Kowalkowski MA, Taylor B, Chou SH. First-to-second antibiotic delay and hospital mortality among emergency department patients with suspected sepsis. Am J Emerg Med. 2021; 46:20–2.
Crossref PubMed
6 Lykins V JD, Kuttab HI, Rourke EM, Hughes MD, Keast EP, Kopec JAC, et al. The effect of delays in second-dose antibiotics on patients with severe sepsis and septic shock. Am J Emerg Med. 2021;47:80–5.
Crossref
7 Kemmler CB, Sangal RB, Rothenberg C, Li SX, Shofer FS, Abella BS, et al. Delays in antibiotic redosing: association with inpatient mortality and risk factors for delay. Am J Emerg Med. 2021;46:63–9.
Crossref PubMed
8 Dersch D, McCormack J. Estimating renal function for drug dosing: rewriting the gospel? Can J Hosp Pharm. 2008;61(2):138–43.
9 Randolph JL, Chan K, Albright A, Chen A. Delays in administration of the second antibiotic dose in patients with severe sepsis and septic shock. Hosp Pharm. 2021;56(4):247–51.
Crossref PubMed PMC
10 Beveridge R, Clarke B, Janes L, Savage N, Thompson J, Dodd G. Canadian emergency department triage and acuity scale: implementation guidelines. Can J Emerg Med. 1999;1(Suppl 3):S1–24.
11 McColl T, Gatien M, Calder L, Yadav K, Tam R, Ong M, et al. Implementation of an emergency department sepsis bundle and system redesign: a process improvement initiative. Can J Emerg Med. 2017;19(2):112–21.
Crossref
12 Cook ME, Schuler BR, Schontz MJ, McLaughlin KC, Lupi KE, DeGrado JR, et al. Prevalence, risk factors, and outcomes associated with delayed second doses of antibiotics in sepsis at a large academic medical center. Antimicrob Steward Healthc Epidemiol. 2023;3(1):e207.
Crossref PubMed PMC
13 Yealy DM, Mohr NM, Shapiro NI, Venkatesh A, Jones AE, Self WH. Early care of adults with suspected sepsis in the emergency department and out-of-hospital environment: a consensus-based task force report. Ann Emerg Med. 2021;78(1):1–19.
Crossref PubMed
Address correspondence to: Dr Kristin Kaupp, Department of Pharmacy, Nova Scotia Health, Halifax Infirmary Site, 1796 Summer Street, Halifax NS B3H 3A7, email: kristin2.kaupp@nshealth.ca
Competing interests: For research and activities not directly related to the study reported here, Emily Black has received grants or contracts from the Canadian Institutes of Health Research, Shoppers Drug Mart, AstraZeneca Canada Inc, GSK, Pfizer Canada, and the Drug Evaluation Alliance of Nova Scotia; speakers’ honoraria from the Pharmacy Association of Nova Scotia, the New Brunswick Pharmacists’ Association, Dalhousie Continuing Pharmacy Education, the Prince Edward Island Pharmacy Association, the Prince Edward Island College of Pharmacy, and the Ontario Pharmacists Association; and travel support from the Joint Programming Initiative on Antimicrobial Resistance (funded by ZonMw, the Netherlands Organisation for Health Research and Development); serves as an expert panelist for the Council of Canadian Academies and as research pillar lead for the Antimicrobial Resistance Steering Committee of the Nova Scotia Department of Health and Wellness; and has a role with the Nova Scotia Pharmacy Regulator. No other competing interests were declared.
Funding: This study was funded by a grant from the Dalhousie College of Pharmacy Endowment Fund.
Acknowledgement: The authors thank Dr Susan Bowles for her assistance with the statistical analysis.
Submitted: April 23, 2025
Accepted: December 5, 2025
Published: April 8, 2026
© 2026 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 79, NUMBER 2, 2026