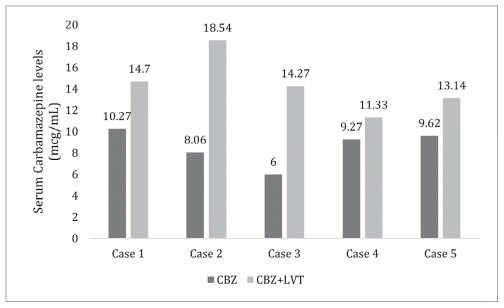
FIGURE 1 Trough serum carbamazepine levels observed for each patient with carbamazepine (CBZ) monotherapy and carbamazepine plus levetiracetam (CBZ + LEV) combination therapy.
Archana Mishra, Biswa Ranjan Mishra, Debadatta Mohapatra, Anand Srinivasan, and Rituparna MaitiTo cite: Mishra A, Mishra BR, Mohapatra D, Srinivasan A, Maiti R. Interaction between carbamazepine and concomitant levetiracetam therapy in patients with epilepsy. Can J Hosp Pharm. 2025;78(4):e3846. doi: 10.4212/cjhp.3846
Combination therapy is often recommended for the treatment of epilepsy when seizures are only partially controlled by monotherapy.1 This approach can potentially enhance seizure control if drugs with different mechanisms of action are selected for the combination therapy. Each medication in the combination can complement the others, addressing various aspects of seizure activity and reducing the overall frequency and intensity of the seizures.
Levetiracetam (LEV) was initially approved as an adjunctive treatment for focal-onset seizures with or without secondary generalizations; later, it was also prescribed as monotherapy.2 The efficacy of add-on LEV in the treatment of refractory seizures has been proven in randomized placebo-controlled trials.3 Known for its broad-spectrum activity and wide therapeutic index, LEV was initially thought to have minimal drug interactions, making it ideal for combination therapy.
Carbamazepine (CBZ), another drug used to treat epilepsy, is absorbed slowly and erratically from the gastrointestinal tract, typically reaching peak plasma concentrations 4 to 8 hours after administration. In contrast, LEV is rapidly and nearly completely absorbed (bioavailability > 95%), with peak concentrations achieved approximately 1.3 hours after ingestion. LEV is primarily eliminated through renal excretion (about 66% unchanged in urine) and undergoes minimal metabolism through enzymatic hydrolysis by plasma and tissue esterases, independent of the cytochrome P450 (CYP) enzyme system.
Unlike CBZ, which is extensively metabolized by CYP3A4, LEV does not induce or inhibit CYP enzymes, making pharmacokinetic interactions with CBZ unexpected.4 However, in the early 2000s, Sisodiya and others5 reported cases of CBZ toxicity—with symptoms such as unsteadiness, nausea, and double vision—when CBZ was combined with LEV, revealing an unexpected interaction. These authors suggested a pharmacodynamic interaction between LEV and CBZ, whereby symptoms of toxicity occurred even though CBZ level was not elevated. However, at our therapeutic drug monitoring clinic, we observed elevated CBZ levels and symptoms of toxicity, such as dizziness and diplopia, after LEV was added to patients’ regimens. The reference range for trough levels of CBZ is 4–12 μg/mL (approximately 17–51 μmol/L).6 Serum levels higher than 12 μg/mL have been associated with nystagmus and ataxia, which require supportive management; a specific antidote is not available.7 A case–control study reported a temporal association between LEV administration and toxic blood levels and toxic symptoms of CBZ, with an odds ratio of 16.65 and with the chances of toxicity being significantly higher when the dose ratio of LEV to CBZ was beyond 1.86.8 The best strategy to reduce symptoms of neurotoxicity and avoid elevation of trough CBZ levels beyond 12 μg/mL appears to be a reduction in the dose of CBZ.
Here, we report 5 cases in which a noticeable increase in trough levels of CBZ was observed, along with features of toxicity, after the addition of LEV. Concomitant administration of drugs (e.g., antifungal agents) or citrus juices (e.g., grapefruit juice) that might inhibit the metabolism of CBZ and increase its serum concentration were ruled out by direct questioning about medication history. The results of blood chemistry examinations were within normal limits, and adherence with therapy was confirmed by direct questioning of the patient or caregiver. All serum concentrations reported in the case summaries below are trough levels (i.e., samples obtained before the next dose was administered), measured at steady-state, whereby patients had been on a stable dose for at least 2 weeks before each measurement. The increase in trough CBZ levels after addition of LEV is shown in Figure 1.
|
| ||
|
FIGURE 1 Trough serum carbamazepine levels observed for each patient with carbamazepine (CBZ) monotherapy and carbamazepine plus levetiracetam (CBZ + LEV) combination therapy. | ||
The primary aim of this report is to alert clinicians to this potential interaction and to stimulate further prospective research to elucidate the precise mechanism. Analysis and reporting of these cases were approved by the Institute Ethics Committee, the data were collected following the principles of the Declaration of Helsinki, and written informed consent was obtained from all participants. All of the information was recorded without any linked identifiers, and confidentiality was maintained.
The 5 patients described in this case series were of Indian ethnicity. A thorough medication history confirmed that none of the patients were taking other concomitant antiseizure medications or known CYP3A4 inhibitors during the period of observation, with the exception of case 3, where zonisamide was added after tapering of LEV.
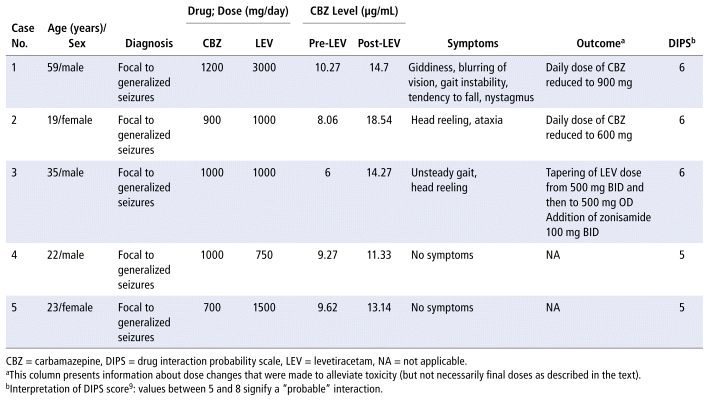
A 59-year-old man with a long history of focal-onset to generalized seizures presented with sleep-related attacks. Initially, he was receiving CBZ at a daily dose of 800 mg (400 mg twice daily). In an attempt to address the sleep-related attacks, the dose was increased to 1200 mg (600 mg twice daily) without any signs of toxicity, but the seizures persisted. The patient’s CBZ serum level was 10.27 μg/mL in January 2019. LEV was started at a dose of 1500 mg/day (750 mg twice daily) and titrated up to 3000 mg/day (1500 mg twice daily) over a period of 6 months, but the seizures remained uncontrolled. The patient experienced symptoms of toxicity, and the CBZ level increased to 14.7 μg/mL in June 2019. The CBZ dose was reduced to 900 mg (300 mg in the morning and 600 mg in the evening), which led to a CBZ level of 11.47 μg/mL in December 2023. Despite various permutations of the patient’s drug therapy, the seizures were not fully controlled until a regimen of CBZ 1000 mg and LEV 1000 mg twice daily was established. By October 2024, the patient had been seizure-free for 6 or 7 months without any adverse effects.
A 19-year-old woman with a history of focal-onset seizures since infancy and delayed developmental milestones had a partial response to CBZ monotherapy. While receiving CBZ 900 mg/day (300 mg in the morning and 600 mg in the evening), her serum level was 8.06 μg/mL in January 2024. Given that only partial seizure control was achieved, LEV 1000 mg/day was added. Although seizures were controlled with the combination therapy, she experienced symptoms of toxicity. The CBZ level rose to 18.54 μg/mL, which led to a dose reduction for CBZ to 600 mg/day (300 mg twice daily) and a dose increase for LEV to 1500 mg/day (750 mg twice daily). The subsequent CBZ level was 14.17 μg/mL, and the toxicity symptoms subsided. CBZ was further reduced to 500 mg/day, and by May 2024, her serum level was 9.88 μg/mL, with no seizures or symptoms of toxicity in the preceding 4 months.
A 35-year-old man with focal-onset to generalized seizures since childhood had been receiving controlled-release CBZ 1000 mg (400 mg in morning and 600 mg in evening) for 10 years, with partial seizure control. His serum CBZ level was 6 μg/mL in January 2024. LEV 1000 mg/day was added, and a month later, he experienced symptoms of toxicity, with the CBZ level rising to 14.27 μg/mL in March 2024. The LEV was gradually tapered, and zonisamide was added. By May 2024, the CBZ level was 12.67 μg/mL, with no signs of toxicity and no seizures during the previous month. Although the CBZ level seemed higher than the documented average population range, control of the patient’s seizures and toxicity symptoms were achieved after withdrawal of LEV.
A 22-year-old man with focal-onset to generalized seizures since childhood was started on CBZ 1000 mg/day (400 mg in the morning and 600 mg in the evening) in November 2023. Serum levels of CBZ were measured to corroborate the lack of efficacy, with a few abortive seizures in January 2024; at that time, the serum level was 9.27 μg/mL. LEV 750 mg/day was added, and 4 months later, the CBZ level was 11.33 μg/mL. The patient remained seizure-free with no symptoms of toxicity, and no changes were made to the regimen.
A 23-year-old woman who had been receiving CBZ 700 mg/day (300 mg in the morning and 400 mg in the evening) for the past 4 months had partial seizure control, with serum level of 9.62 μg/mL in October 2023. LEV 1500 mg was added, following which the CBZ level increased to 13.14 μg/mL with a report of diplopia in January 2024. The CBZ dose was reduced to 500 mg, and serum levels dropped below the toxic range. The patient’s seizures remained well controlled.
We applied the drug interaction probability scale (DIPS)9 for each individual case, determining a score of 5 or 6 for each one. This score indicates a suspicion of drug interaction (where the object drug is CBZ, and the precipitant is LEV).9 According to the DIPS criteria, a score between 5 and 8 signifies a “probable” interaction, supporting a causal relationship between the addition of LEV and the observed changes in CBZ levels and/or toxicity. The 5 cases are summarized in Table 1.
TABLE 1 Summary of Cases of Interaction between Carbamazepine and Levetiracetam
We have presented 5 cases of a possible pharmacokinetic interaction between CBZ and LEV, characterized by elevation of CBZ levels following the addition of LEV to patients’ drug regimens. CBZ is the preferred drug for focal seizures, and the incidence of adverse events is reportedly higher with addition of LEV than with CBZ alone.1,10 However, when physicians encounter elevated CBZ levels and symptoms of toxicity, it seems logical to down-titrate the CBZ dose. The lack of a known significant effect of LEV on CYP enzymes emphasizes the safety of LEV, which further supports down-titration of CBZ without clinicians having any second thoughts.
Sisodiya and others5 have previously reported a pharmacodynamic interaction between these 2 drugs. More specifically, they documented an increase in the incidence of dizziness, drowsiness, and confusion when the drugs were taken together. An animal study was conducted to evaluate the neurotoxic effect of antiseizure medications in terms of motor incoordination, showing that LEV potentiates the neurotoxicity of CBZ and topiramate.11 An isobolographic analysis of the micro-electroshock seizure model also showed a synergistic effect of the LEV–CBZ interaction.12 In that study, the levels of CBZ were increased after the addition of LEV, but the change was not statistically significant. In another study, hydrolysis of CBZ-epoxide, a toxic, pharmacologically active metabolite of CBZ, was inhibited by brivaracetam (a drug belonging to the same class as LEV).13 In that study, CBZ concentration seemed to decrease slightly after day 21, but that may have been due to the autoinduction properties of CBZ. Although to date there is no known significant pharmacokinetic interaction between the 2 drugs, we observed that serum levels of CBZ increased with co-administration of LEV in our patients. When we checked the interaction between CBZ and LEV using an online tool (DDInter checker 2.0, available from: http://ddinter2.scbdd.com/inter-checker/), we found some minor substrate–substrate, inhibitor–inhibitor, and substrate–inhibitor interactions between the 2 drugs for various CYP enzymes (Figure 2).
|
| ||
|
FIGURE 2 Bar plot generated from an online drug interaction checker tool (DDInter checker 2.0, available from: http://ddinter2.scbdd.com/inter-checker/), illustrating the predicted inhibitory potential of carbamazepine (blue) and levetiracetam (green) on various cytochrome P450 (CYP) isoenzymes. The y axis represents a predicted interaction score from 0 to 1, where higher values indicate stronger potential for that drug to act as an inhibitor of the specified enzyme (e.g., CYP1A2-inh). Calculated scores greater than 0.75, which may represent clinical significance, are shown. This figure illustrates that whereas carbamazepine is predicted to have significant inhibitory interactions, the predicted inhibitory potential of levetiracetam across these enzymes is low, which aligns with its known pharmacokinetic profile. For each of the 5 CYP enzymes shown across the horizontal axis, the first pair of bars represent each drug as inhibitor for the specified enzyme, whereas the second pair of bars show each drug as substrate for the enzyme. | ||
Although we did not measure levels of the primary active metabolite of CBZ (carbamazepine-10,11-epoxide), its reference range is not well established, and therefore correlation of efficacy or toxicity with serum CBZ levels appeared more reliable. We do not yet have any definitive explanation for this pharmacokinetic interaction; however, it might be explained by increased absorption of CBZ in the presence of LEV, pharmacogenetic variability, or ethnic variability, which could affect the pharmacokinetics of both CBZ and LEV. While it is known that LEV is not metabolized by CYP enzymes, this drug may influence CBZ levels through indirect mechanisms, such as transporter interactions (e.g., P-glycoprotein [P-gp], organic anion transporters) or allosteric modulation of metabolic enzymes. A plausible hypothesis for this interaction involves the modulation of drug transporters in the gastrointestinal tract. CBZ is a known substrate of the P-gp efflux transporter. If LEV exerts even a mild inhibitory effect on P-gp, it could decrease the efflux of CBZ from enterocytes back into the gut lumen, thereby increasing its net absorption and overall bioavailability. This would lead to higher serum concentrations, as observed in our patients. The increase may also be due to the effect of 2 uncharacterized metabolites of LEV. This could be a cause for concern in patients whose focal seizures are not fully controlled by CBZ and who may be considered for combination therapy. However, this hypothesis needs to be confirmed by robust, well-designed prospective studies that account for other confounding factors.
Given the findings reported here, we suggest that clinicians consider monitoring serum CBZ levels within 2–4 weeks after initiating LEV as add-on therapy, especially if patients report symptoms of toxicity. This may allow for early detection and timely dose adjustments, optimizing both safety and seizure control.
1 Brodie MJ, Perucca E, Ryvlin P, Ben-Menachem E, Meencke HJ; Levetiracetam Monotherapy Study Group. Comparison of levetiracetam and controlled-release carbamazepine in newly diagnosed epilepsy. Neurology. 2007;68(6):402–8.
Crossref PubMed
2 Lyseng-Williamson KA. Spotlight on levetiracetam in epilepsy. CNS Drugs. 2011;25(10):901–5.
Crossref PubMed
3 Nazarbaghi S, Hosseinpour H, Mirzaie S, Sadighpour T. Comparative effect of levetiracetam and carbamazepine versus levetiracetam and sodium valproate on seizure frequency in patients with epilepsy. J Phys Med Rehabil. 2022;4(1):13–8.
4 Patsalos PN. Clinical pharmacokinetics of levetiracetam. Clin Pharmacokinet. 2004;43(11):707–24.
Crossref PubMed
5 Sisodiya SM, Sander JWAS, Patsalos PN. Carbamazepine toxicity during combination therapy with levetiracetam: a pharmacodynamic interaction. Epilepsy Res. 2002;48(3):217–9.
Crossref PubMed
6 Grześk G, Stolarek W, Kasprzak M, Grześk E, Rogowicz D, Wiciński M, et al. Therapeutic drug monitoring of carbamazepine: a 20-year observational study. J Clin Med. 2021;10(22):5396.
Crossref
7 Schmidt S, Schmitz-Buhl M. Signs and symptoms of carbamazepine overdose. J Neurol 1995;242(3):169–73.
Crossref PubMed
8 Mishra A, Mishra BR, Mohapatra D, Srinivasan A, Maiti R, Hota D. Carbamazepine toxicity with concomitant levetiracetam therapy in patients with epilepsy. Int J Neurosci. 2025:1–7. doi: 10.1080/00207454.2025.2489701.
Crossref PubMed
9 Horn JR, Hansten PD, Chan LN. Proposal for a new tool to evaluate drug interaction cases. Ann Pharmacother. 2007;41(4):674–80.
Crossref PubMed
10 Trinka E, Marson AG, Van Paesschen W, Kälviäinen R, Marovac J, Duncan B, et al.; KOMET Study Group. KOMET: an unblinded, randomised, two parallel-group, stratified trial comparing the effectiveness of levetiracetam with controlled-release carbamazepine and extended-release sodium valproate as monotherapy in patients with newly diagnosed epilepsy. J Neurol Neurosurg Psychiatry. 2013; 84(10):1138–47.
Crossref
11 Luszczki JJ, Andres MM, Czuczwar P, Cioczek-Czuczwar A, Wojcik-Cwikla J, Ratnaraj N, et al. Levetiracetam selectively potentiates the acute neurotoxic effects of topiramate and carbamazepine in the rotarod test in mice. Eur Neuropsychopharmacol. 2005;15(6):609–16.
Crossref PubMed
12 Luszczki JJ, Andres MM, Czuczwar P, Cioczek-Czuczwar A, Ratnaraj N, Patsalos PN, et al. Pharmacodynamic and pharmacokinetic characterization of interactions between levetiracetam and numerous antiepileptic drugs in the mouse maximal electroshock seizure model: an isobolographic analysis. Epilepsia. 2006;47(1):10–20.
Crossref PubMed
13 Stockis A, Chanteux H, Rosa M, Rolan P. Brivaracetam and carbamazepine interaction in healthy subjects and in vitro. Epilepsy Res. 2015;113:19–27.
Crossref PubMed
Address correspondence to: Dr Rituparna Maiti, Department of Pharmacology, 1st Floor, Academic Block, All India Institute of Medical Sciences (AIIMS), Bhubaneswar (Odisha), India, Pin-751019, email: pharm_rituparna@aiimsbhubaneswar.edu.in
Competing interests: None declared.
Funding: None received.
Submitted: June 11, 2025
Accepted: August 28, 2025
Published: December 10, 2025
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 4, 2025