doi: 10.4212/cjhp.3850
Facilitated Poster Sessions: Discussions of original research, pharmacy practice projects, and case reports.
Urslak R1,2, Ladhar S3, Gauthier G2, Sajwani S1,2, Kanji S1,2,4, Pammett R3,5, Landry C2
1The Ottawa Hospital, Ottawa, ON
2School of Pharmaceutical Sciences, University of Ottawa, Ottawa, ON
3Faculty of Pharmaceutical Sciences, University of British Columbia, Vancouver, BC
4The Ottawa Hospital Research Institute, Ottawa, ON
5Northern Health, Prince George, BC
Climate change threatens humanity, biodiversity, and global health, with the healthcare system already experiencing its effects. Pharmacy teams must act to increase environmental sustainability through education and action, yet there is a dearth of literature on how to incorporate this content into pharmacy education. Despite increased awareness and agreement of importance of the topic, resources on integrating this content into pharmacy curricula remain limited.
To summarize and describe the extent, range, and nature of research and implementation of planetary health content within pharmacy education globally.
A scoping review was conducted using the Arksey and O’Malley framework and the PRISMA-ScR checklist. Nine databases were searched using well-defined inclusion and exclusion criteria. Dual independent reviewers screened and assessed articles published up to March 2024 for eligibility, and data was charted to analyze results.
The search yielded 1,827 articles, with 16 selected for analysis. These articles provided 28 examples of planetary health content integrated into 25 pharmacy schools across 12 countries. The content was organized using a novel framework with four main categories: the impact of environmental disruptions on health, the impact of health on the environment, mitigation strategies, and adaptation strategies. Integration varied, occurring across curricula, in mandatory courses, or through electives and extracurricular activities. Teaching methods also varied, with individualized and didactic learning being the most common.
While efforts to integrate planetary health into pharmacy education remain limited, internationally, progress is evident. This review offers a framework for understanding the diverse perspectives and topics within this emerging field. It aims to support discussions on developing prioritized competencies for inclusion in pharmacy curricula.
Mutin J1, Hafizin M-L1, El-Jammal V1, Morel C1, Leca L1, Bergeron M2, Bussières J-F3,4
1Étudiant en pharmacie, Unité de Recherche en Pratique Pharmaceutique, Département de pharmacie, Centre hospitalier universitaire Sainte-Justine, Montréal, QC
2Pharmacien, Coordonnateur régional des systèmes informatiques en pharmacie (SIP), Département clinique de pharmacie, Centre intégré de santé et de services sociaux des Laurentides, Hôpital de Saint-Jérôme, Saint-Jérôme, QC
3Responsable, Unité de Recherche en Pratique Pharmaceutique, Département de pharmacie, Centre hospitalier universitaire Sainte-Justine, Montréal, QC
4Professeur de clinique, Faculté de pharmacie, Université de Montréal, Montréal, QC
Generative Artificial Intelligence (AI), particularly conversational agents like ChatGPT, can be used to automate data extraction. However, it remains to be determined if these tools can extract scientific data with accuracy comparable to humans, especially in the field of pharmacy.
To assess the consistency of data extracted by ChatGPT 4.0 compared to Impact Pharmacie entries.
This is a descriptive comparative study. A random selection of 100 articles entered on Impact Pharmacie in 2024 was identified. ChatGPT 4.0 was programmed with a series of instructions and queried with 18 questions per article to extract targeted data. The responses were compared to data previously entered into the Impact Pharmacie platform by pharmacy students following a standardized operating procedure. Consistency was assessed across 23 criteria (e.g., study type description, pharmaceutical intervention description, limitations) and categorized into three levels: consistent, partially consistent, and inconsistent. The number and nature of AI hallucinations were also recorded.
The consistency of data extracted by ChatGPT compared to student-entered data averaged 77 ± 19%. However, AI hallucinations were observed in 23 out of 100 articles, and 30% of parameters (n = 138/449) were missing from the extraction.
ChatGPT 4.0 is a promising tool to support data extraction from pharmaceutical scientific literature, but human validation remains essential to ensure the accuracy of the information.
Keywords: Generative artificial intelligence, data extraction, pharmacists, impact
Marceau N1, Hung M1,2
1Association des pharmaciens des établissements de santé du Québec (A.P.E.S.), Montréal, QC
2Department of Pharmacy, St. Mary’s Hospital Center, Montreal West Island Integrated University Health and Social Services Center, Montréal, QC
Since January 2021, Quebec pharmacists can practice under collaborative practice agreements (CPA) with physicians and nurse practitioners. To support their implementation, the Association des pharmaciens des établissements de santé du Québec developed general and specific CPA templates. General CPA apply to most or all admitted patients, whereas specific CPA apply to patients in defined areas of clinical practice.
To measure the proportion of CPA implemented in Quebec hospitals 2 and 3 years after their enactment. Secondary objectives included to identify barriers and facilitators to CPA implementation and to assess pharmacy leadership perceptions on autonomous pharmacist prescribing.
A cross-sectional, descriptive study using 2 electronic surveys was conducted among directors of pharmacy in Quebec hospitals. The 2023 survey contained 17 multiple choice, closed, and open-ended questions. The 2024 survey comprised of 5 closed and open-ended questions. Descriptive statistics were conducted.
Data were collected between March 25 and April 11, 2023 and between April 19 and 22, 2024. Response rates were, respectively, 78.3% (29/37) and 91.9% (34/37). In 2023, 20.7% of responding health care facilities had implemented a general CPA. This increased to 38.2% in 2024. Regarding specific CPA, 19 respondents (65.5%) had implemented specific CPA in 2023. This increased to 20 respondents in 2024. Clinical practice areas with the greatest implementation of specific CPA were ambulatory oncology care and long-term care. The most cited barriers to CPA implementation included variations in pharmacists’ perceptions on autonomous prescribing and increased workload. Facilitators included collaborative pharmacist-physician relationships and physician receptivity to pharmacist prescribing. There were 58.6% of respondents who reported daily or near-daily autonomous prescribing by pharmacists.
General and specific CPA have been implemented in approximately 40% and 60% of Quebec hospitals, respectively. Pharmacists in Quebec appear to embrace their expanded scope and regularly engage in autonomous prescribing activities.
Newman P1, Dhaliwall S1, Polyakova O1, McDonald K1
1Northwest Telepharmacy Solutions, The North West Company, Winnipeg, MB
A clinical pharmacy key performance indicator (cpKPI) is a quantifiable measure of clinical pharmacy care provided by pharmacists. CpKPIs are aimed at improving the quality of patient care and advancing the practice of pharmacy. Implementation of cpKPIs and collecting data over time enables continuous quality improvement. Patient care activities provided by telepharmacists servicing health care facilities across Canada is unknown.
To describe the current state of cpKPI implementation and documentation in Canadian hospitals supported by telepharmacists.
Eligible participants were client site-lead telepharmacists for Canadian hospitals. Participants were asked to report the following for all 8 cpKPI’s at their sites: cpKPI expectations, documentation in the pharmacy software system, in the healthcare record, and workload. Awareness of prioritization of pharmacist clinical activities based on cpKPI’s, presence of computer prescriber order entry (CPOE) and person(s) responsible for the best possible medication history were requested. Data was gathered using Microsoft Office Forms. Descriptive statistics were applied, continuous data reported by central tendency, and categorical data as numbers and percentage.
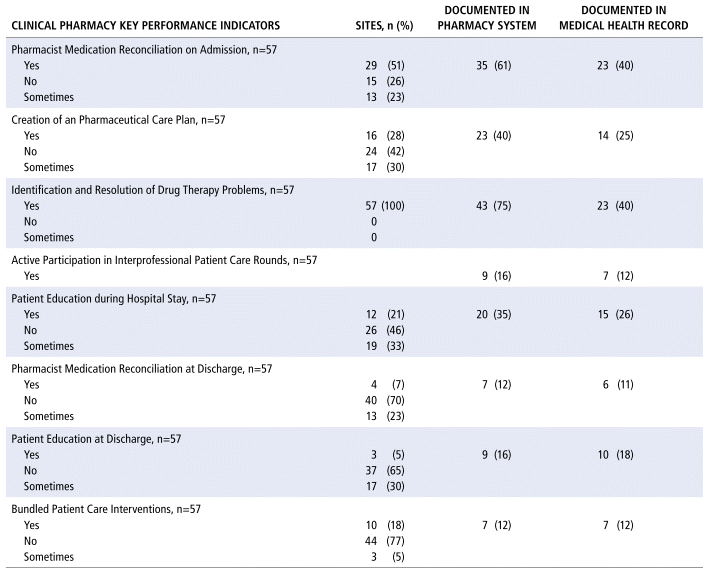
Forty-two site leads completed the questionnaire representing 57 hospitals from August to November 2024. Facility size ranged from 8–500 beds, supported in part, or in full, by telepharmacist services. Pharmacy software systems included Meditech (Magic DOS, Expanse, Client-Window), EPIC, Cerner and BDM. CPOE was employed in 65% of sites. Prioritization of clinical pharmacy services based on cpKPI’s was known in 16% of sites. Facility cpKPI activities and documentation are described in Table 1.
There is an ongoing need to progress in prioritizing cpKPI care activities and documentation integral to advancing the practice of pharmacy and improving patient outcomes.
For the table that belongs with this abstract, please see Appendix 1.
Mutin J1,2, El-Jammal V1,2, Bussières JF1,3
1Unité de recherche en pratique pharmaceutique, Département de pharmacie, CHU Sainte-Justine, Montréal, QC
2Faculté de Pharmacie de Lyon, Université Claude Bernard Lyon 1, Lyon, France
3Faculté de pharmacie, Université de Montréal, Montréal, QC
The scientific abstracts published in Pharmactuel are the basis for communications presented at the annual APES congress, playing a key role in disseminating work in hospital pharmacy.
To describe and evaluate the quality of these abstracts published in Pharmactuel from 2007 to 2023.
Retrospective descriptive study. All abstracts published from January 1, 2007, to December 31, 2023, were included. The abstracts were evaluated based on a grid of 10 descriptive criteria and 16 quality criteria. A compliance rate per criterion was calculated.
515 abstracts were published and analyzed. Pharmactuel publishes an average of 31 abstracts per year. On average, abstracts included 4.6±2.8 authors, an increase since 2018. The first authors were primarily pharmacists (38.4%) or pharmacy students (31.7%). Improvements were observed after 2007 in the description of context (39.1% to 96.7%) and the clarity of objectives (91.3% to 99.5%). In contrast, the description of evaluation criteria regressed (87.0% to 59.0%) and other criteria showed no differences. In total, 8/16 criteria showed a compliance rate >70%.
The annual number of abstracts published by Pharmactuel has remained constant since 2007. The update of guidelines in 2008 is associated with an improvement in the compliance of three criteria. An increase in the number of allowed words and the dissemination of compliant abstracts could contribute to improving quality.
Keywords: Pharmacy, abstract, scientific publication, quality
Roy C1, Johnson K1, Zerr E1, Panchuk K1, Iroh N1
1Department of Pharmacy Services, Saskatchewan Health Authority (SHA) – Regina, Regina, SK
Intravenous (IV) medications carry significant environmental burden (due to stringent sterility requirements, packaging, storage, and administration) and patient burden (site infections, prolonged hospitalization, and reduced mobility) compared to oral (PO). A local IV to oral antimicrobial stepdown prescriptive authority policy exists; however, its application is unknown.
To describe pharmacist awareness and application of the IV to PO stepdown policy.
Firstly, an electronic survey of 77 SHA-Regina pharmacists included five questions regarding awareness and application of the policy and barriers and opportunities for its use. Secondly, a one-day audit of acute care adult patients was conducted. Patients were identified for inclusion via a BDM Pharmacy report of active orders for eligible IV antimicrobials. Data was collected from patients’ electronic health records and included demographics and IV to PO stepdown eligibility (diagnosis is compatible with oral therapy, no evidence of abnormal oral absorption, and clinical stability). Descriptive statistical analysis was conducted via Microsoft Excel.
The survey included 34 pharmacists (44% response rate). All were aware of the policy, however, 72% do not apply in practice. Common barriers were a preference for interdisciplinary collaboration rather than prescriptive authority and concern with physician pushback. Opportunities included enhancing awareness for physicians and expanding eligible medications and dosages. The audit included 27 patients – 15 female (56%), median age 67 (range 16–88), and most commonly prescribed metronidazole (12, 44%) and ciprofloxacin (6, 22%). One-third (9) were eligible for IV to PO stepdown. Clinical instability (8, 30%) most commonly resulted in ineligibility.
Despite awareness of the IV to PO stepdown pharmacist policy and significant opportunity for its application, pharmacists do not routinely apply it. A preference for interdisciplinary collaboration requires broader engagement to address barriers and opportunities. These results will inform local initiatives, including a multi-pronged, multi-disciplinary approach of educational and system-level efforts.
Tangedal K1, Semchuk W1
1Saskatchewan Health Authority, Regina, SK
Climate change is the biggest health threat facing humanity, and healthcare professionals play an important role adaptation and mitigation. Given the importance of reducing provincial healthcare emissions, this study sought to determine the views of Saskatchewan Health Authority (SHA) employees on climate change and health.
An electronic survey was distributed to SHA employees from June 3rd – 28th 2024. Inclusion criteria was anyone employed by or practicing in SHA.
There were 179 participants that completed at least one question. The majority believed climate change is happening (158/179, 88%) and is caused by human activity (107/179, 60%). Over half felt climate change would harm them personally, however only 11% felt that this would a great deal. For future generations, 67% of respondents felt they would be effected a great deal. The majority agreed healthcare workers are responsible to bring climate change to the attention of policy makers (62%) and the public (58%). However, they did not feel empowered to take action (52%). Common reasons for not communicating about climate change were a lack of time, lack of effect, and lack of knowledge.
Survey respondents felt climate change is happening due to human activity and is likely to affect health, even more significantly for future generations. SHA employees were less convinced of climate change than multinational results, demonstrating a need for education.
Goldstein S1, Jaheinsha Y1, Nguyen M1, Shea A1
1Department of Pharmacy, The Ottawa Hospital, Ottawa, ON
Hospitalizations and mortality associated with opioid use disorder (OUD) are increasing and require patient-centered interventions to maximize the impact of OUD treatment. It is important to continue OUD pharmacotherapy in hospitalized patients to ensure continuity of care. Unaddressed OUD can lead to withdrawal, difficult behaviours, self-directed discharges, and distress among healthcare providers.
This study aimed to analyze outcomes associated with continuity of care for patients receiving OUD pharmacotherapy. The primary objective was to identify the proportion of any OUD pharmacotherapy administered greater than 24 hours after hospital presentation. Secondary objectives included outcomes related to continuity of OUD pharmacotherapy during transitions of care and initiation of new OUD pharmacotherapy for patients not previously on therapy.
A retrospective chart review was completed for 100 adult medicine patients with OUD who were receiving OUD pharmacotherapy in the community or were newly initiated on opioid agonist therapy in hospital, between November 2023 and April 2024.
Overall, 86.5% of patients on OUD pharmacotherapy prior to admission received a dose of any OUD pharmacotherapy within 24 hours of hospital presentation. Also, 73.3% of patients were not ordered their full home OUD regimens on admission, and 27.0% of changes to home regimens were either unintentional or without a documented reason. Intentional changes to home regimens on admission were commonly due to opioid overdose, illicit opioid withdrawal, and altered level of consciousness. Deleterious outcomes commonly occurred and included self-directed discharges, code whites, and violent behaviour.
The majority of patients admitted on existing OUD pharmacotherapy receive doses of any OUD pharmacotherapy within 24 hours of presentation to hospital. Gaps in care were identified in the continuity of home OUD pharmacotherapy regimens, especially among patients on complicated regimens prior to admission. Further research is required to determine reasons for unintentional discrepancies to improve patient outcomes.
Kean E1, d’Entremont-Harris M1, Callaghan A1, Peltekian S1, Ramsey TD1,2
1Pharmacy Department, Nova Scotia Health, Halifax, NS
2Dalhousie University, Halifax, NS
Vaccination rates remain below target in Canada, and hospital admissions are an underutilized opportunity for immunization. Leveraging hospitalization to assess, prescribe, and administer vaccines can increase immunization rates.
This study aimed to compare the rate of vaccine history collection, eligibility assessment, administration, education, and follow-up activities before and after implementing a best possible vaccine history and vaccine eligibility tool (BPVHVET), personalized for pharmacy professional use, for publicly funded vaccines (PFVs).
A before and after, single-centre, retrospective chart review was utilized to characterize inpatient vaccine-related activities in adult patients admitted to internal medicine after implementing a BPVHVET. Patient demographics, vaccine eligibility factors, and immunization activities were collected.
Thirty-four and 51 participants were included in the before and after periods, respectively. Participants had risk factors for vaccine-preventable diseases in both study periods, including heart disease (37.6%), diabetes (30.6%), and chronic lung disease (29.4%). In the before period, no best possible vaccine histories, vaccine eligibility assessments, immunizations, education sessions, or plans for outpatient follow-up were completed. In the after period, 9 (17.6%) best possible vaccine histories and vaccine eligibility assessments were completed, all using the BPVHVET. Additionally, 4 (7.8%) patients received vaccines, 8 (15.7%) had education sessions, and 5 (9.8%) received outpatient vaccine follow-up plans. Of the 9 patients with vaccine eligibility assessments, 8 (88.9%) received 4 or more eligible vaccines.
Implementation of a BPVHVET that encompassed all adult PFVs led to increased completion of best possible vaccine histories, eligibility assessments, immunizations, education sessions, and outpatient follow-up plans.
Xiao Z1, Ho C1,2
1Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, ON
2Department of Psychiatry, Temerty Faculty of Medicine, University of Toronto, Toronto, ON
A culture of safety is important for improving patient safety and patient care. Safety climate questionnaires, which provide a snapshot of safety culture, have not been reported in the literature for hospital pharmacy settings in Ontario, Canada.
To perform a descriptive, cross-sectional exploration of Ontario hospital pharmacy patient safety climate in Canada.
This study was conducted using the Agency for Healthcare Research and Quality (AHRQ) Hospital Survey on Patient Safety Culture (HSOPSC) 2.0, consisting of 10 composite measures (from Teamwork to Communication Openness). The online questionnaire was posted on LinkedIn, Facebook, and X, from March 4 to April 7, 2024, and engaged with by the Ontario College of Pharmacists and the Canadian Society of Healthcare-Systems Pharmacists Ontario Branch, on the same social media platforms, for hospital pharmacy professionals’ anonymous and voluntary participation. Descriptive statistics were used to analyze quantitative data, and thematic analysis was applied to free-text responses accordingly.
Sixty-three responses (75% pharmacists, 25% pharmacy technicians) were received with the majority (54%) from the Toronto Region. An overall positive patient safety culture was perceived by more than 70% of respondents. Of the 10 composite measures, “Teamwork” and “Supervisor, Manager, or Clinical Leader Support for Patient Safety” were the top two, while “Handoffs and Information Exchange” was the lowest. Staffing, compensation, and ease of error reporting were identified as important facilitators for advancing patient safety climate.
Ontario hospital pharmacy patient safety culture was generally positively perceived by pharmacy professionals. Teamwork, peer, and leader collaboration could be leveraged to address patient safety gaps at transition points of care, such as handoffs. Patient safety climate could be explored in future studies using qualitative research methods for further insight, followed by development of potential interventions.
El-Jammal V1, Mutin J1, Morel C1, Hafizin M-L1, Leca L1, Deranlot T1, Charbonneau J1, Martel Côté N1, Bussières J-F1,2
1Unité de recherche en pratique pharmaceutique, CHU Sainte-Justine, Montréal, QC
2Faculté de pharmacie, Université de Montréal, Montréal, QC
Since 2012, the ImpactPharmacie platform (http://impactpharmacie.org) has been cataloging articles addressing the roles and impacts of pharmacy practice. A pedagogical activity involving pharmacy students could contribute to updating this platform.
To evaluate the feasibility and effectiveness of involving third-year pharmacy students (Université de Montréal, UdEM) in extracting scientific literature data on the roles and impacts of pharmacy practice to update the ImpactPharmacie platform.
This is a retrospective descriptive study. A total of 160 articles were assigned to third-year students as part of a pedagogical activity, with training provided via video. Each student read, analyzed, and completed a data extraction grid (MS Word). The extracted data were compared to those entered into ImpactPharmacie based on 25 criteria, with conformity rated using four statuses: compliant, incorrect, incomplete, or absent. The severity of non-compliant criteria was assessed using two levels: minor or major.
On average, 76% of the data extracted by students matched those entered into ImpactPharmacie. Conformity ranged from 45% to 100%, depending on the criteria. 58% of all non-compliant criteria were classified as major (479/826).
This pilot study demonstrates the feasibility of engaging third-year PharmD students in analyzing and extracting data on the roles and impacts of pharmacy practice within a pedagogical framework to update the ImpactPharmacie platform. For the next cohort, the training video will be improved to include an overview of non-conformities identified in the pilot phase.
Keywords: Data extraction, Impact, Pharmacy
Ahmed W1, Sørensen J2, Pollock RF1
1Covalence Research Ltd, Harpenden, UK
2Pharmacosmos A/S, Holbæk, Denmark
For patients living with iron deficiency anemia (IDA) in British Columbia (BC), intravenous (IV) iron is preferred when oral iron supplementation is ineffective, poorly tolerated, or contraindicated. Iron sucrose (IS, Venofer®) and ferric derisomaltose (FDI, Monoferric®) are two commonly-used IV iron formulations in BC, however, patients receiving IS require more infusions per treatment course to correct iron deficiency than patients receiving high-dose FDI.
To measure the impacts of differences in infusion frequencies for FDI and IS on annualized travel times, travel distances, and carbon dioxide (CO2) emissions for patients living with IDA in BC.
A list of public medical facilities administering IV iron in BC was obtained. The mean Haversine distance from each municipality to the closest medical facility was calculated, adjusted using a detour index, and weighted by population. Transport modality and CO2 emissions data were obtained from Canadian Government sources. Published studies were used to calculate mean infusion numbers per treatment course for IS and FDI, CO2 emissions arising from infusion material usage, and patient population estimates. These inputs were incorporated within the underlying transport model to calculate changes in annualized patient travel times, travel distances, and CO2 emissions associated with IS or FDI use, exclusively, over 1,000 model iterations.
Compared to IS, FDI reduced annual travel distances for 31,407 patients by 8,608,845 km (standard deviation [SD]: 1,676,428), from 10,498,592 km (SD: 2,043,402) to 1,889,747 km (SD: 367,812), reducing patient travel times by 171,152 hours (SD: 33,529) and lowering subsequent CO2 emissions by 1,344,059 kg (SD: 265,007). FDI also reduced annual CO2 emissions associated with infusion material usage by 53,889 kg compared to IS.
Relative to IS, FDI significantly reduced IV iron infusion requirements, yielding subsequent reductions in annual travel times, travel distances, and CO2 emissions for patients living with IDA in BC.
Pryjma M1, Buchkowsky S2, Feng J1, Masoudi S1, Barry A1,3
1Jim Pattison Outpatient Care and Surgery Centre, Lower Mainland Pharmacy Services, Surrey, BC
2Surrey Memorial Hospital, Lower Mainland Pharmacy Services, Surrey, BC
3Faculty of Pharmaceutical Sciences, University of British Columbia, Vancouver, BC
Intravenous iron supplementation reduced heart failure (HF) hospitalizations in patients with acute HF with reduced ejection fraction (HFrEF) and iron deficiency in the AFFIRM-AHF trial. The purpose of this study was to determine the rate of iron deficiency among patients with HFrEF at our institution, and evaluate whether the hospitalization rate was sufficiently high to warrant implementation of a routine intravenous iron repletion program.
To identify what proportion of patients with HFrEF also had iron deficiency, and compare the HF hospitalization rate among these patients to AFFIRM-AHF.
This was a retrospective cohort study at the Heart Function Clinic at the Jim Pattison Outpatient Care and Surgery Centre in Surrey, British Columbia. Included were ambulatory patients with HFrEF (left ventricular ejection fraction ≤40%) who had a serum ferritin measured <12 months and were attached to the clinic for ≥4 months between February 2021 and August 2022. The primary objective was proportion of patients with iron deficiency, defined as ferritin <100 mg/L or 100–299 mg/L with a transferrin saturation of <20%.
Two hundred and eighty-four patients were screened and 100 met the inclusion criteria. Of those, 73% had iron deficiency. Patients with iron deficiency were older (68 vs 66 years) and more were women (33% vs 19%) compared to those without. Among patients with iron deficiency, the rate of HF hospitalizations after clinic attachment was 21.6 per 100 patient-years, compared to 43.2 per 100 patient-years in patients who did not receive iron supplementation in AFFIRM-AHF.
Almost three-quarters of subacute HFrEF patients met the criteria for iron deficiency at our clinic. However, the HF hospitalization rate was noticeably lower than what was observed in AFFIRM-AHF. Therefore, it is questionable whether routine intravenous iron repletion would be effective at reducing HF hospitalizations among our patients with iron deficiency.
Kwan C1, Hinse A2, Lacombe J-G1, Powell G1, Lavoie L-P1, Plotnick L1,3, Bank I1,3
1Emergency Department, Montreal Children’s Hospital, McGill University Health Centre, Montréal, QC
2Pharmacy Department, Montreal Children’s Hospital, McGill University Health Centre, Montréal, QC
3Department of Pediatrics, Faculty of Medicine and Health Sciences, McGill University, Montréal, QC
Pediatric trauma medication preparation in emergency departments (EDs) is error-prone due to weight-based dosing, cognitive demands, and limited pharmacy support. These challenges delay care and risk patient safety. NurEx, a dosing software, was developed to streamline workflow sand integrate pharmacist oversight through a novel pharmacy module.
To evaluate NurEx’s impact on medication preparation efficiency for nurses and assess the pharmacy module’s potential to enhance pharmacist workflows and interdisciplinary collaboration.
A two-phase crossover trial was conducted at Montreal Children’s Hospital. Phase I involved healthcare professionals (N = 14) completing timed simulations under standard practice and NurEx conditions for non-acute and acute trauma medication tasks. Primary endpoints were time-to-calculate (order-to-dose) and time-to-patient (order-to-readiness). Secondary endpoints included clinician confidence and ease of use (7-point Likert scale). Phase II will test the pharmacy module with pharmacists, focusing on medication tracking, verification efficiency, and error interception. Statistical analysis used Wilcoxon signed rank tests.
In Phase I, NurEx significantly improved efficiency. For non-acute cases, NurEx decreased time-to-calculate by 70.7% and time-to-patient by 50.1% compared to standard practice (both P < 0.05). In acute trauma care, NurEx reduced time-to-calculate by 87.0% and time-to-patient by 42.6% compared to standard practice (both P < 0.05). High cognitive load medications saw time-to-calculate decrease by 83.1% (P < 0.001). For Phase II, the pharmacy module is projected to reduce pharmacist verification time by 60%, reduce medication preparation time by 30%, and improve interdisciplinary communication.
NurEx enhances medication safety and efficiency in pediatric EDs by reducing clinician cognitive load and automating dose calculations. The pharmacy module has the potential to optimize pharmacist workflows and strengthen interdisciplinary collaboration. These findings support NurEx as a scalable tool to address systemic challenges in emergency medication preparation. Future studies will assess broader applicability across care settings.
Roy C1, Kinsley B1, Vanin B2, Anthony E2, Saboni A2, Randhawa A1, Galey E2, Heilman L2, Lett R3, Semchuk B1
1Pharmacy Services, Saskatchewan Health Authority, Regina, SK
2Pharmacy Services, Saskatchewan Health Authority, Saskatoon, SK
3Department of Anesthesiology, Saskatchewan Health Authority, Regina, SK
Implementation of a universal outpatient intravenous (IV) iron program in Saskatchewan necessitated an analysis of oral iron usage in those utilizing IV iron to ensure appropriate iron deficiency anemia management.
To describe prior oral iron utilization in patients receiving outpatient IV iron therapy.
This survey included adults who were receiving an iron infusion at a Saskatchewan Health Authority outpatient infusion site June 17 to November 15, 2024. Patients receiving hemodialysis or in first trimester of pregnancy were excluded. Patients were invited to participate by a nurse or a poster at the infusion clinic, and had the option of completing a printed paper version, electronic version (QR code on the poster), or being interviewed by a researcher. The questionnaire comprised 20 questions, including demographics, history of oral iron use, patient understanding of indication for IV iron, and additional comments. Microsoft Excel was utilized for descriptive data analysis.
A total of 149 survey responses were collected. Among them, 110 (73.8%) were female. The majority of respondents were aged 35–44 years (34, 22.8%). Three-quarters (112/149, 75.2%) of respondents had trialed oral iron, in which oral iron was stopped due to ineffectiveness (40/112, 35.7%) or intolerance (33/112, 29.5%), commonly reported as constipation, nausea, or abdominal pain. The most common oral iron formulations trialed were ferrous gluconate (42, 26.9%) and polysaccharide-iron complex (38, 24.4%) at various dosage regimens. Over half of patients took oral iron with food or with interacting food or medications.
While most patients reported having trialed oral iron in the past, their usage was suboptimal for absorption and tolerability. Oral iron is a cost-effective treatment option and should be adequately trialed prior to commencing IV iron. Opportunities exist to optimize oral iron through education of healthcare providers and patients to increase tolerability and promote effective dosing.
Ye S1, Wei W1, Ahamed A1, Garabedian K1, Choi A1, Hashemi-Sabet F1, Dakkak T1, Ho C1
1Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, ON
Numerous studies have demonstrated the benefits of utilizing mobile health applications (m-health apps), to improve patient care. However, no guidelines exist as a reference for pharmacists when making recommendations of m-health apps to patients.
The objective of this project was to characterize the factors that pharmacists and patients prioritize when assessing m-health apps. These factors may help determine pharmacists’ knowledge, attitudes, and confidence in recommending m-health apps to patients.
A literature search was conducted in MEDLINE and EMBASE. Ten studies were reviewed and analyzed for factors related to using/recommending m-health apps, positive/negative attitudes on m-health app use/recommendations, and confidence/readiness in recommending a m-health app to a patient.
A factor that influenced pharmacists’ knowledge on m-health apps was age. Pharmacists less than 35 years old have better perceptions of apps and their use in patient care. Factors that promote positive attitudes in app recommendation included features such as notifications or reminders, of which their use have improved patient adherence to medication use. Factors that prompted negative attitudes included lack of scientific evidence behind information provided within the app. Other barriers involved a lack of usability, unfamiliarity with using mobile apps, and security concerns. Factors that improved confidence in making app recommendations included whether the app was accessible in various app stores, visually appealing, easy-to-use, and presentation of information in a patient-friendly manner.
From being aesthetically pleasing and user-friendly to being able to improve medication adherence and patient communication, all studies illustrated similar preferred factors in m-health apps. While further studies are necessary to elucidate factors influencing pharmacists’ recommendations of m-health apps, what we learned from the literature can serve as a starting point for future guideline development and continuing professional development.
Nguyen A1, Lee J1, Hashemi-Sabet F1, Huynh V1, Zhang Y1, Ho C1
1Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, ON
Recognizing the concerns raised about standards for quality control, assurance, and formulation consistency of compounded products, the National Association of Pharmacy Regulatory Authorities (NAPRA) published the NAPRA Model Standards for Pharmacy Compounding. These standards were adopted and implemented by the Ontario College of Pharmacists.
This project intends to assess pharmacy professionals’ perception of best practices and challenges in fulfilling the NAPRA Model Standards for Pharmacy Compounding in Ontario.
We surveyed pharmacy professionals in Ontario through an online questionnaire, including both quantitative and qualitative open-ended items, from August to October 2024. Quantitative and qualitative data were analyzed using descriptive statistics and thematic analysis, respectively.
We received 50 responses, of which 60% reported participation in in-house pharmaceutical compounding practices. Most (77%) respondents were familiar with and aware of the NAPRA Model Standards for Pharmacy Compounding for Non-Sterile Compounding, even if they did not participate in compounding (70%). Many (81%) believed that standardizing compounding practices would improve patient safety. However, 52% respondents found it challenging for pharmacies to implement the NAPRA standards, and 44% commented that maintaining compliance with the standards was also challenging. Limitations in implementing the NAPRA Model Standards for Pharmacy Compounding were reported to be associated with financial (64%), lack of space (56%), and human resources (52%).
While most respondents believe standardizing compounding practices would improve patient safety, challenges in implementing and maintaining compounding standards exist. Future investigations should explore avenues to improve compliance to compounding standards/guidelines, which may also encourage uptake of compounding practices.
Lynch R1, Brandt A1, Polyakova O1, Dhaliwall S1, McDonald K1, Newman P1, Tivy T1
1Northwest Telepharmacy Solutions, The North West Company, Winnipeg, MB
Medication reconciliation (Medrec) at transitions of care is key for medication safety. The best possible medication history (BPMH), creation of an accurate and complete list of a patient’s home medications, is an integral first step to the Medrec process. The usability of an electronic tool employing a standardized algorithm to guide the BPMH interview was tested.
To test tool usability and refine content based pharmacy technician (RPhT) end-user feedback. To describe nurse created BPMH discrepancies identified by the RPhT using the BPMH tool. To determine RPhT time requirements for preparation, conduction and documentation of a BPMH using the tool.
This was a non-interventional, single-center cross-sectional study that compared nurse created BPMHs to remote RPhT created BPMHs using an electronic tool. Those included were adult patients admitted to a 30 bed rural hospital through the emergency room. Data collected comprised of RPhT time to compile and complete a BPMH, number and type of medications per patient, nurse derived BPMH medication deviations and their associated risk. Descriptive analysis of the data was performed.
Three RPhTs created 154 patient BPMHs using the tool between February–March 2024. Tool algorithm questions were refined on a continuum throughout the study period. The median time for RPhT creation of a BPMH was 85 (42–181) minutes. Patients were on a median of 10 (5–15) medications. Nurse created BPMHs had deviations identified by the tool ranged from 1–6: 86% (1), 71% (2), 50% (3), 10% (4), 8% (5), and 3% (6) respectively. Most deviations involved scheduled prescription medications (35 %), and drug omissions (69%). Thirty per cent of discrepancies had the potential to cause moderate or severe patient discomfort or deterioration.
The electronic tool tested for the creation of BPMHs by RPhTs remotely was refined, deemed efficient, user-friendly, and improved the accuracy a BPMH.
Cameron J1,2, McNally D3, Blanc A1,2
1Department of Pharmacy, CHEO, Ottawa, ON
2Research Institute, CHEO, Ottawa, ON
3Department of Pediatrics, CHEO, Ottawa, ON
Respiratory syncytial virus (RSV) is the leading cause of bronchiolitis in infants, resulting in high hospitalization rates. The rate of superimposed bacterial infections in RSV positive (+) children who have been admitted to general pediatric wards is relatively low, typically below 2%.
A retrospective chart review was conducted to determine if the prevalence of RSV and bacterial co-infections in children ≤ 90 days old admitted to a pediatric intensive care unit (PICU) is similarly low to those seen in RSV+ children admitted to general wards.
Cases were identified using a Canadian academic pediatric hospital electronic health records (Epic®) reports were generated from October 1st, 2017, to April 1st, 2022. Data collected were variables related to demographics, clinical characteristics, antimicrobial use, bacterial sample collection and culture results.
Of the 197 RSV+ patients included for analysis, 32 were admitted to the PICU and 165 were admitted to general pediatric wards. Seventy-five percent of PICU and 60% of general ward patients had at least one cerebrospinal fluid (CSF), blood, or urine sample collected. Twenty-two percent, 56%, and 71% of PICU, and 13%, 46% and 53% of general ward patients had CSF, blood and urine samples collected, respectively. Seventy-five percent of PICU and 27% of general ward patients had antimicrobials started. Culture positivity rates for CSF, blood and urine were 0%, 0% and 5%, respectively in PICU patients, and 0%, 1% and 6%, respectively in general ward patients.
The rates of bacterial sample collection and antimicrobial use were higher in patients admitted to the PICU. The rates of culture positivity were similar between both wards and were low. In an Antimicrobial stewardship perspective opportunities to limit antimicrobial use and bacterial sample collection in the PICU should be considered.
Strongman C1, Bowles S1, Rioux J2, DiCostanzo T1, Marcoux C3, Minard LV1
1Pharmacy Department, Nova Scotia Health, Central Zone, QEII Health Sciences Centre, Halifax, NS
2Nova Scotia Health Cancer Care Program, Halifax, NS
3Division of Hematology and Hematologic Oncology, QEII Health Sciences Centre and Dalhousie University, Halifax, NS
Chemotherapy-induced nausea and vomiting (CINV) is commonly experienced by patients receiving chemotherapy. Since it is easier to prevent rather than to treat CINV, guidelines recommend that prophylactic antiemetics be used depending on the emetogenicity of the chemotherapy regimen.
The objectives of this study were to 1) characterize CINV in patients who received autologous hematopoietic stem cell transplant (HSCT) in Nova Scotia, and 2) analyze how CINV is managed and whether this management adheres to antiemetic guidelines.
This retrospective chart review included adult patients who received autologous HSCT in Nova Scotia in 2023. Data collected included patient demographics, treatment details (e.g., conditioning chemotherapy regimen) and the presence, grade, onset, and duration of nausea/vomiting. Guideline adherence was defined as ‘managed according to guidelines’ if antiemetic prescribing aligned with any one of three published guidelines.
All 56 patients who underwent autologous HSCT received highly emetogenic chemotherapy and a 3-drug antiemetic regimen (NK1-receptor antagonist, 5HT3 receptor antagonist, dexamethasone) for the prophylaxis of CINV. All patients experienced chemotherapy-induced nausea, and 80.4% experienced chemotherapy-induced vomiting. The median number of days patients experienced nausea or vomiting was 10 (range 1–19) and 2 (range 0–15), respectively. The most common grade of nausea and vomiting was grade 2. Antiemetics were not prescribed in accordance with published guidelines in 100% of patients: 96.3% did not receive post-chemotherapy dexamethasone and 100% received ondansetron for a longer than recommended duration.
Characterization of CINV in patients who received autologous HSCT revealed a high rate of CINV (100%) and no adherence to antiemetic guidelines (0%). Our results can increase awareness of CINV in patients receiving HSCT. Incorporating guideline-recommended antiemetics into standardized order sets, facilitated by oncology pharmacists working in collaboration with the multi-disciplinary team, could better support prescribers in providing optimal care to patients receiving HSCT.
Pozdirca M1(supervised by Khoday A)
1Faculty of Law, University of Manitoba, Winnipeg, MB
Given shifting abortion laws in the U.S., some healthcare professionals (“HCP”) may feel conscience-bound to cross these legal boundaries. Canada has similar insufficiencies in legal protection of conscience-based actions. However, conscientious objection (“CO”) is allowed through policies. Exploring these can show how policy can also create protections for conscience-based provision of healthcare services.
A search was conducted for CO policies on each regulator’s website and publicly-available search engines. Policy clauses were compared and categorized as weak, moderate, strong, or unclear. Legal decisions were then intertwined to provide narrative and analysis around identified themes.
Four themes were identified: avoiding persuasion, continuity of care, advance planning, and reducing harm in emergencies. Six physician regulators strongly limit persuasion, 1 has a moderate limitation, and 2 have weak limitations. Three pharmacist regulators have strong limitations against persuasion, 2 have moderate limitations, and 2 are unclear. An Ontario disciplinary decision shows the boundary between medication counselling and persuasion. Regarding continuity of care, 2 physician regulators have strong provisions for effective referral, 6 have moderate provisions, 1 has a weak provision, and 2 are unclear. Two pharmacist regulators have strong provisions, 3 have moderate provisions, 5 have weak provisions, and 1 is unclear. Pharmacist policies also require immediate, advance notice of CO to employers. Three physician and 3 pharmacist policies require that, in an emergency, the HCP may need to set aside their CO. Only pharmacist policies define “emergency”.
A review of CO policies and legal decisions show four key themes, demonstrating valid limits that may be applied to policies on CO and conscientious provision of services.
Orser S1, Patel D1, Kalia K1,2
1Trillium Health Partners (THP), Mississauga, ON
2University Toronto, Toronto, ON
A large acute care hospital, THP, implemented an electronic Health Information System (HIS) in 2020 supporting quality patient care, service delivery and clinician efficiency. Feedback from pharmacists at THP identified an increase in alert fatigue and workload associated with the volume of medication warnings generated by the system, particularly those considered inaccurate or clinically irrelevant.
This quality improvement project aimed to alleviate alert fatigue and increase time for patient care by reducing unnecessary medication warnings for inpatient pharmacists.
The project was conducted in two phases. The first phase involved analyzing the volume, types and responses to medication warnings presented to pharmacists using HIS data. The second phase focused on clinical analysis of the most frequently overridden warnings, and gathering feedback from a working group of pharmacists across clinical specialities on the clinical value of these warnings. Decisions to modify warnings were made based on leading practices and clinical consensus within the working group. An iterative, pilot approach was used to mitigate potential risks.
Since the implementation of the medication warning modifications, there has been a significant 79% reduction in the number of warnings presented to pharmacists, from 18,900 to 4,000 per week. These adjustments also led to a notable decrease of 25 hours per week in the time pharmacists spend reviewing warnings. Since the project’s initiation, no patient harm has been reported related to reduced warnings. Furthermore, feedback from pharmacists has been overwhelmingly positive, highlighting the effectiveness and safety of the intervention.
A comprehensive clinical analysis, including pharmacist engagement, of medication warnings can effectively reduce alert volumes and pharmacist time spent reviewing unnecessary medication warnings, without negatively impacting patient care. Active engagement of pharmacists was crucial in identifying alert fatigue as a priority issue and in implementing medication warning changes to improve project outcomes.
Boatswain G1, Bhajan S1, Bodley T1, John S1, Lee L1, McRiner D1, Yeung E1
1Scarborough Health Network, Toronto, ON
Unnecessary use of intravenous (IV) rather than enteral (PO) medications increases hospital length-of-stay, nursing administration time, drug cost, and environmental footprint. Choosing Wisely Canada recommends using enteral formulations when they are equally safe and effective.
Scarborough Health Network (SHN) targeted an IV to PO stewardship program for levetiracetam and pantoprazole.
The study included three SHN hospital sites; pre-intervention June 2022 to February 2023 and intervention March 2024 to November 2024. Interventions to enhance appropriate IV to enteral formulations included 1) education and awareness of evidence-base practices through e-mail and medicine rounds, 2) development of prescribing criteria for IV use by Drugs and Therapeutics Committee, 3) electronic health record decision support tools that recommend PO stepdown when IV is ordered, and 4) pharmacist interventions to recommend IV to PO stepdown. The primary outcome measure was intravenous medication doses per 1000 patient days. Statistical process control charts were used to establish statistical significance. Secondary measures included enteral formulation dispenses per 1000 patient days. Balancing measures included a survey of provider awareness/satisfaction.
A total of 832,915 patient-days were included; 334,404 pre- and 498,511 post-intervention. Levetiracetam IV dispenses decreased from 26.5 to 22 per 1000 patient days (17% reduction, special cause variation June 2024). Pantoprazole IV dispenses decreased from 107.8 to 76.6 per 1000 patient days (29% reduction, special cause variation December 2023). There was decrease in the IV/PO ratio by 23% for levetiracetam and 29% for proton pump inhibitors, indicating increased usage of enteral options.
Inter-disciplinary collaboration with prescribers and utilization of electronic decision support tools are both necessary to facilitate IV to PO stewardship. Future analysis of provider satisfaction, cost savings, and carbon footprint reduction are underway to evaluate economic and ecological impact.
Newman P1, Polyakova O1, McDonald K1, Dhaliwall S1, Lamoure J1
1Northwest Telepharmacy Solutions, The North West Company, Winnipeg, MB
Pharmacists in family health teams (FHT) play an integral role in improving patient care by providing medication management, counselling and ensuring safe optimal medication use. In collaboration with the patient and health care professionals, pharmacists optimize treatment plans, monitor therapies, educate patients on proper use of medications and help manage chronic disease to enhance health outcomes and improve patient safety. Although the involvement of pharmacists in FHT’s has been increasing, there is a paucity of research available for pharmacists supporting rural FHTs.
Quantify pharmacist activities, their associated time requirements, for patients serviced by telepharmacists in remote family health teams (FHT) in rural Ontario, Canada.
This prospective, multicenter cohort study took place in 4 FHTs. Five pharmacists documented their time requirements for patient specific tasks: appointment set up, interview preparation, patient interview, documentation, care provider communication and follow up visits. The number of drug therapy problems (DTP) per patient were captured. Central tendencies were reported for quantitative values and percentages for categorical variables. Inferential statistics was used to identify correlation.
A total of 181 patients received 155 hours of direct patient care from pharmacists at from Sep 2023–July 2024. Mean patient age was 61 (56–78). Overall, the proportion of time spent on patient specific tasks: patient interview (36%), documentation (29%), interview preparation (21%), appointment set up (9%) and communication to heath care team members (5%). Median time for each patient interaction was 27 (15–43) min. There was a strong, positive monotonic correlation between total spent time and identified DTPs (rs= .73. p=.46).
This study provided valuable insights into the amount of time required for remote pharmacists dedicated to FHT patient care activities helping to enhance understanding of the resources required to support pharmacist involvement in FHTs, both at the individual level and across multiple FHT’s.
Métras M-É1, Blanc A2,3, Cameron J2,3
1Department of Pharmacy, Centre Hospitalier Universitaire Sainte-Justine, Montréal, QC
2Department of Pharmacy, CHEO, Ottawa, ON
3Research Institute, CHEO, Ottawa, ON
Managing sedation and analgesia in critically ill pediatric patients is essential for ensuring patient comfort and compliance with mechanical ventilation. Identifying the precise level of sedation needed is challenging, as either under or oversedation can have negative effects. However, variability in practices exists across different pediatric intensive care units (PICUs), and standardized protocols are often lacking.
Our main objective was to review all drugs to manage pain and/or anesthesia, for critically ill, mechanically ventilated, children.
We conducted a retrospective chart review of mechanically ventilated pediatric patients admitted to the PICU (13 budgeted beds), of a Canadian academic pediatric hospital, between April 1, 2022 and March 31, 2023. Data on drug type, dosage, and duration of administration were collected, along with patient demographics and intubation characteristics.
A total of 121 patients were included, with respiratory failure (48.2%) being the leading cause of intubation. Dexmedetomidine, used in 82% of patients, had a median infusion duration of 78.9 hours (IQR: 45.06–147.34) at a mean dose of 0.7 mcg/kg/h. Fentanyl, administered to 84.2% of patients, had a median infusion duration of 70.3 hours (IQR: 36.83–135.63) at a mean dose of 2.4 mcg/kg/h. Younger patients (<1 year) received higher mean doses of fentanyl (2.4 mcg/kg/h) and dexmedetomidine (0.7 mcg/kg/h) than older children (1.67mcg/kg/h and 0.52mcg/kg/h, respectively). Patients intubated for prolonged periods (>7 days) had higher mean doses of fentanyl (2.9 mcg/kg/h) and dexmedetomidine (78 mcg/kg/h). Prolonged intubation was associated with increased dosages of dexmedetomidine and fentanyl.
This study highlights variations in treatment choices where age and duration of intubation influenced drug selection and dosing, emphasizing challenges with sedation protocols. Future research with comparative data from other pediatric hospitals is needed to better describe sedation protocols and understand the impact of variability on patient outcomes.
Anthony E1, Roy C1, Saboni A1
1Department of Pharmacy Services, Saskatchewan Health Authority, Regina, SK
Artificial Intelligence (AI) uses statistical methods to identify patterns in previous cases and applying this data to new cases. Application of AI may be useful in streamlining Pharmacy Practice Support activities.
To review AI application opportunities to improve efficiency within pharmacy project management.
A literature search via MEDLINE and Google Scholar was conducted. Search terms included artificial intelligence, pharmacy, project management, healthcare, and policy and procedure. The tasks performed by Pharmacy Practice Support (staff/student/resident development, research, formulary support, project coordination, and residency program coordination) and local healthcare system readiness were compared to the literature regarding relative cost to savings, system support for AI integration, and barriers.
Overall, used appropriately, AI can increase outputs, improve efficiencies, and reduce costs. An organization-specific AI tool using internal data could improve policy and procedure development. AI could also generate patient cases along with human clinical review. However, a majority of the tasks performed by the team require clinical knowledge not currently replaceable by AI. Limitations of AI include bias, as AI may incorrectly learn patterns, compounded by “black boxes” (not explaining the decision-making process). Additionally, AI cannot currently assess for clinical significance. Lack of routine updates leads to risk of outdated answers. Also, ethical considerations in healthcare use exist (i.e., patient privacy and confidentiality). Before implementing AI in a healthcare setting, ISMP recommends: establishing a multidisciplinary committee for AI governance, performing a risk assessment, implementing a vendor assessment process, and instituting a post-implementation quality control monitoring process. These currently do not exist in the local organization.
AI is capable of being both useful and harmful. Meaningful implementation of AI would be costly and require a significant learning curve for the local Pharmacy Practice Support team. However, identifying sources of inefficiencies can identify areas for future AI use.
d’Entremont-Harris M1, Spencer K1, McInnis S1, Slayter K1,2,3,4, Isenor JE3,5, Bowles SK1,2, Ramsey TD1,5
1Pharmacy Department, Nova Scotia Health, Halifax, NS
2Department of Medicine, Dalhousie University, Halifax, NS
3Canadian Center for Vaccinology, IWK Health, Halifax, NS
4Pharmacy Department, IWK Health, Halifax, NS
5College of Pharmacy, Dalhousie University, Halifax, NS
Vaccinations are essential public health tools to reduce the burden of vaccine-preventable diseases; however, Canada’s national adult vaccination goals have not been met. Community pharmacists have been identified as key enablers of adult vaccination, especially during the COVID-19 pandemic; however, the involvement of Canadian hospital pharmacists in vaccine advocacy activities is not well quantified.
This study aimed to describe the number of hospital pharmacists who participated in vaccine education, facilitation, or administration, and to assess practice change.
Electronic questionnaires were distributed to hospital pharmacists caring for adult patients practicing at tertiary care academic centres across Canada. The questionnaire was distributed in 2019 and 2023, and the results were compared.
A total of 607 completed questionnaires were included in the analysis (375 from 2019 and 234 from 2023). All nine provinces with tertiary care academic centres were represented in both time points. Only 17% (39/234) of pharmacists felt adult immunization rates were adequate. The proportion of pharmacists participating in all vaccine advocacy roles increased significantly from 2019 to 2023. In 2019, 87% of respondents identified as educators, 84% identified as facilitators, and 41% identified as administrators, compared to 90%, 93%, and 52%, respectively, in 2023. Further, in 2023 significantly more pharmacists had completed injection training (from 51% to 69%) and certification (38% to 58%).
The proportion of Canadian hospital pharmacists engaging in vaccine advocacy increased from 2019 to 2023, but only approximately one-half of hospital pharmacists administered vaccines, missing opportunities to enhance vaccination rates.
Blanc A1,2, Liu KW3, Fournel F1, Vasseur C1, Cameron J1,2
1Department of Pharmacy, CHEO, Ottawa, ON
2Research Institute, CHEO, Ottawa, ON
3Canadian Society of Healthcare-Systems Pharmacy (Ontario Branch), Ottawa, ON
The pharmacy sector faces critical challenges in recruitment and retention. The Hospital Pharmacy in Canada 2020–21 Survey reported an average vacancy rate for hospital pharmacists between 3.7 to 14%. The 2022 Canadian Pharmacists Association survey highlighted 28% of respondents are likely/somewhat likely to leave the pharmacy sector, thus understanding employment preferences and job satisfaction factors is vital.
To identify key features of career needs and aspirations of the future pharmacy workforce and help inform recruitment and retention strategies across pharmacy sectors in Canada.
A cross-sectional survey was conducted from April 19 to May 31, 2024, using the REDCap platform. Multiple choice responding provided data on demographics, work-hour preferences, salary expectations, and job satisfaction factors analyzed using descriptive statistics and subgroup comparisons.
Of the 807 respondents, 546 were pharmacy employees (388 pharmacists, 127 pharmacy technicians, and 31 pharmacy assistants) and 261 were students (36 science graduate students, 43 pharmacy students, 28 pharmacy technician students, and 7 pharmacist residents). Full-time employment was the most preferred by employees and students (67.4% and 59.0%, respectively). However, a considerable portion of employees and students desired to only work either 10–20 hours (36.9% and 47.5%, respectively) or 20–30 hours (41.7% and 20.3%, respectively) per week. Furthermore, work-life balance was the most selected factor contributing to short-term job satisfaction for employees, who prioritized stability and career advancement in the long run. In contrast, students valued role variety in the short term and sought mentorship in the workplace in the long term. Finally, employees had a higher salary expectation than students ($57.19/hour vs. $45.32/hour).
Pharmacy professionals currently in the workforce have different employment preferences and job satisfaction drivers compared to students. Tailored recruitment and retention strategies are essential to meet varying personal and professional goals and, in turn, ensure workforce sustainability in pharmacy.
Law S1, Wyllie A2, Ma NH1
1Department of Pharmacy, Sunnybrook Health Sciences Centre, Toronto, ON
2Department of Pharmacy, Sinai Health, Toronto, ON
Carbetocin is a long-acting oxytocin analogue used to prevent postpartum hemorrhage. Juno Pharmaceuticals’ carbetocin is formulated as a 100mcg/1mL prefilled syringe (PFS) and requires storage under refrigerated conditions (2–8°C). The ability to store Juno-Carbetocin at room temperature would allow stock to be located at the point of care without the need for a refrigerator and minimize wastage from syringes erroneously left at room temperature.
To evaluate the chemical stability of Juno-Carbetocin 100mcg/1mL PFS at room temperature (25°C) over 31 days.
On study day 0, 21 prefilled syringes were removed from the refrigerator and stored at room temperature with protection from light. Concentration analysis was completed on study days 0, 1, 3, 7, 14, 28, and 31 using a validated, stability-indicating liquid chromatographic method with UV detection. The chemical stability was determined by calculating the intersection of the lower limit of the 95% confidence interval of the observed degradation and the time to achieve 90% of the initial concentration.
The analytical method separated carbetocin from its degradation products such that the concentration of carbetocin was measured specifically, accurately (deviations from known averaged 1.32%), and reproducibly (within day variation averaged 0.44% and between day variation averaged 1.11%). During the study, all syringes retained their initial concentration and remained clear and colourless. The calculated time to achieve 90% of the initial concentration with 95% confidence exceeded the 31 day study duration.
We conclude that Juno-Carbetocin 100mcg/1mL PFS are stable for at least 31 days when stored at room temperature with protection from light.
Riss V1, Hook R1, Neault A1, Scharrer D1, Law S2, Ma NH2
1Department of Pharmacy, Hospital for Sick Children, Toronto, ON
2Department of Pharmacy, Sunnybrook Health Sciences Centre, Toronto, ON
While previous studies have demonstrated the stability of ketamine 1mg/mL, data for ketamine diluted in 0.9% sodium chloride (NS) to concentrations of 2 and 10mg/mL are not available.
To evaluate the chemical stability of ketamine diluted with 0.9% NS to concentrations of 2 and 10mg/mL stored in polypropylene syringes for 90 days at both room temperature (25°C) and in the refrigerator (4°C).
On study day 0, eight polypropylene syringes containing ketamine 2mg/mL diluted in 0.9% NS and an additional eight polypropylene syringes containing ketamine 10mg/mL diluted in 0.9% NS were prepared. Four syringes of each concentration were stored at room temperature and four syringes were stored in the refrigerator. Three syringes were used to determine the concentration of ketamine on each study day and the fourth syringe was used to evaluate physical stability. The concentration was determined and physical stability evaluated on study days 0,1,7,14,21,28,42,56,72,90 using a validated, stability-indicating liquid chromatography method with UV detection. Chemical stability was determined by calculating the intersect of the lower limit of the 95% confidence interval of the degradation rate and time to achieve 90% of the initial concentration.
The analytical method separated degradation products from ketamine such that the concentration was measured specifically, accurately (deviations from known averaged 1.35%) and reproducibly (within day replicate error averaged 0.50% and between day replicate error averaged 1.22%). Linear regression identified concentration (p<0.01) as a predictor of percent remaining but not study day (p=0.63) or temperature (p=0.38). The calculated time to 90% of the initial concentration with 95% confidence exceeded the 90 day study period for both concentrations and both storage temperatures.
We conclude that ketamine 2 and 10mg/mL solutions diluted with 0.9% sodium chloride stored in polypropylene syringes is stable for at least 90 days when stored at 4°C and 25°C.
Gill JK1, Gessner B1, Natsheh C2,3, Bruchet N1,4, Bolt J1,4
1Pharmacy Department, Interior Health Authority, Kelowna, BC
2Department of Pharmacy, University Health Network, Toronto, ON
3Leslie Dan Faculty of Pharmacy, University of Toronto, Toronto, ON
4Faculty of Pharmaceutical Sciences, University of British Columbia, Vancouver, BC
Pharmacy residents are at risk of burnout, which can impact wellbeing, performance, and reduce sense of satisfaction. Support interventions, specifically peer support, mentorship, and connectedness may help alleviate stress and improve well-being.
To describe the current opportunities for and perceptions of available peer support, mentorship, and connectedness amongst Canadian Post-Graduate year 1 (PGY1) pharmacy residents, and to identify interest and desire for additional support opportunities.
A cross-sectional national online survey of Canadian PGY1 residents from the 2021–2022, 2022–2023, and 2023–2024 residency years was conducted. A subset of survey respondents participated in an online focus group designed to further explore themes identified in the survey.
There were 109 participants in the survey and 7 participants in the focus group. Support was available to participants in the forms of mentorship (90.8%), wellness or self-care opportunities (41.3%), and peer connection (86.2%). Mentorship and peer connection were highly valued, yet just over half of pharmacy residents were satisfied with the level of mentorship and peer connection received. Identified opportunities for improvement included formalizing mentorship programs with clear roles, protected time, intentional matching, and paid training for mentors, enhancing access to health and wellness resources, and designating site support for residents.
Mentorship and peer connection are the most commonly provided and highly valued supports for Canadian PGY1 pharmacy residents. Opportunities exist to enhance all forms of support offered to PGY1 pharmacy residents, which ultimately may help to reduce resident burnout and improve resident wellbeing.
Cameron J1,2, Bournival V3, Lavoie C4, Blanc A1,2
1Department of Pharmacy, CHEO, Ottawa, ON
2Research Institute, CHEO, Ottawa, ON
3Department of Pediatrics, Northern Ontario School of Medicine, Sudbury, ON
4Department of Pediatrics, CHEO, Ottawa, ON
Ipratropium bromide is a key adjunct therapy in treating acute asthma exacerbations in pediatric emergency departments (EDs). While clinically effective, its delivery via metered-dose inhalers (MDIs) often leads to significant wastage, contributing to financial costs and environmental harm due to the release of hydrofluorocarbon (HFC) propellants, measured in kilograms of C02 (C02e). Just one MDI can generate the CO2e of travelling by car from Toronto to Niagara.
This study aimed to investigate the administration patterns of ipratropium bromide in a pediatric ED, quantify wastage, and assess its environmental and financial impacts.
A retrospective chart review was conducted at a Canadian academic pediatric hospital for patients aged 0–18 years who received ipratropium bromide in the ED between January 1, 2023, and December 31, 2023. Demographics, Pediatric Respiratory Assessment Measure (PRAM) scores, medication use, and MDI wastage were extracted from the electronic medical record system (Epic®). Descriptive statistics were used to assess medication use and wastage, while C02e was calculated to estimate environmental impact of wasted doses.
A total of 1,027 patients (1,232 ED visits) were included, with a mean PRAM score was 7.6±2, indicating that most patients presented with moderate-to-severe asthma exacerbations. However, 7.3% of ipratropium bromide administrations occurred in patients with presenting PRAM scores below 6, suggesting overuse in mild cases. Of the 246,400 dispensed doses, only 3,644 were administered (1.5%), resulting in a wastage rate of 98.5% (242,756 doses). This wastage contributed to 17,700 kg of CO2e emissions and a financial cost more than $43,500 CAD.
This study highlights significant wastage of ipratropium bromide in a ED, contributing to environmental and financial burdens. Strengthened adherence to PRAM-based guidelines could reduce un-necessary usage, while maintaining clinical efficacy. Implementing sustainable alternatives, such as dry powder inhalers or recycling programs, may further minimize the carbon footprint.
Mutin J1,2, Leclerc V3,4, Guénette L5,6, Métras M-E1,7, Bondurant-David K8, Nguyen P9,10, Poulin M-C11,12, Bergeron L5,6,13, Méthot J3,4,5, Dupont C11, Bussières J-F1,7
1Unité de recherche en pratique pharmaceutique et Département de pharmacie, Centre hospitalier universitaire Sainte-Justine, Montréal, QC
2Faculté de Pharmacie de Lyon, Université Claude Bernard Lyon 1, Lyon, France
3Institut universitaire de cardiologie et de pneumologie de Québec-Université Laval, Québec, QC
4Centre de recherche de l’Institut universitaire de cardiologie et de pneumologie de Québec-Université Laval, Québec, QC
5Faculté de pharmacie, Université Laval, Québec, QC
6Centre de recherche du CHU de Québec-Université Laval, Axe santé des populations et pratiques optimales en santé, Québec, QC
7Faculté de pharmacie, Université de Montréal, Montréal, QC
8Centre intégré universitaire de santé et de services sociaux de l’Ouest-de-l’Île-de-Montréal, Institut universitaire en santé mentale Douglas et Centre Hospitalier St-Mary’s, Montréal, QC
9Centre hospitalier universitaire de Montréal, Montréal, QC
10Centre de recherche du Centre hospitalier universitaire de Montréal, Montréal, QC
11Centre hospitalier universitaire de santé McGill, Montréal, QC
12Association des pharmaciens de établissements de santé du Québec, Montréal, QC
13CHU de Québec-Université Laval, Québec, QC
Generative artificial intelligence (GAI) has rapidly developed since its launch in 2022. From image analysis to drug design and scientific research and writing, its use in healthcare is continuously expanding. The editorial board of Pharmactuel has investigated pharmacists’ exposure to GAI in scientific publishing.
To provide an overview of the knowledge and use of GAI by pharmacists in Quebec healthcare institutions.
A retrospective descriptive study. An online questionnaire (Forms, Microsoft, Seattle, WA, USA) containing 11 questions was sent to the professional emails of 2,074 hospitals pharmacists members of the Association des pharmaciens des établissements de santé du Québec in December 2024. Respondents were informed that the aggregated results would be used for posted and written communication purposes.
Out of 2,074 pharmacists solicited, 336 responded (response rate of 16%). Fifty-one percent reported having used GAI, with 85% accessing it through free platforms. Twenty-eight percent had already read an article about the use of GAI in healthcare, with a median of 3 articles (min: 1, max: 50). The most cited uses were translation assistance (84%) and grammatical and syntactical corrections (75%). The main concerns were data confidentiality breaches and the generation of inaccurate information. Lastly, 83% expressed interest in training on the use of GAI in scientific publishing.
GAI is now part of the scientific landscape, and half of the responding pharmacists are already using it. All pharmacists should monitor its developments, familiarize themselves with its benefits in their practices, and assess its risks and challenges.
Keywords: Generative artificial intelligence, scientific publishing, hospital pharmacists
Augustyniak M1Ariano R*2, Bonnar P*3, Dixon A*4, Sheth P*5, Péloquin S1, Lazure P1
*Equal contributions
1AXDEV Group Inc., Montréal, QC
2Department of Pharmacy, St. Boniface Hospital, Winnipeg, MB
3Division of Infectious Diseases, Department of Medicine, Dalhousie University, Halifax, NS
4Department of Pediatrics, Faculty of Medicine & Dentistry, University of Alberta, Edmonton, AB
5Department of Pathology and Molecular Medicine, Queen’s University, Kingston, ON
Pharmacists are vital in the interdisciplinary management of infections in concordance with antimicrobial stewardship (AMS) principles and should be supported in demonstrating leadership in this space.
To identify where and how Canadian pharmacists working in hospital (Pharm-H) and retail (Pharm-R) settings can improve their AMS competencies and leadership.
A mixed-methods needs assessment combined a thematic analysis of qualitative interviews (n=28) with a subsequent inferential analysis of quantitative surveys (n=140) that included subjective (self-report of current and expected levels of knowledge/skill) and objective (multiple-choice questions) assessment techniques. Methods and participants’ perspectives (physicians and pharmacists) were triangulated.
Interviews indicated a challenge amongst AMS leaders to recommend adjustments to prescribers’ antimicrobial treatments. A sense of defensiveness on the part of prescribers was reported, hindering pharmacists’ confidence to practice relevant AMS leadership. Objectively, surveyed Pharm-H demonstrated in a significantly lesser proportion to have knowledge gaps of multi-drug resistance and AMS-concordant treatment than Pharm-R (Table 1). However, subjectively, surveyed Pharm-H reported in significantly greater proportion the need to improve their knowledge and skills, compared to Pharm-R (Table 1). Amongst survey respondents with an AMS program in place, half (55%) reported that a pharmacist was part of the committee. A minority (35%) of surveyed Pharm-H reported “never/rarely/sometimes” incorporating AMS principles, compared to 100% of Pharm-R (X2(1)=19.259, p<0.001).
Pharm-H expressed challenges in their ability to demonstrate leadership in AMS and are critical of their own knowledge and skill improvement needs. As antimicrobial resistance continues to be a rising global threat, beyond hospitals, we call for all pharmacists to advocate for their expertise to contribute to antimicrobial-related decisions in line with AMS principles.
For the table that belongs with this abstract, please see Appendix 2.
Puri K1, Riahi S1, Simao S1
1Pharmacy Department, Mackenzie Health, Richmond Hill, ON
The Institute for Safe Medication Practices recommends the use of bidirectional infusion pump interoperability to provide a highly effective medication safeguard. Through bidirectional transmission of information between infusion pumps and the electronic health record (EHR), dose error reduction software (DERS) can be leveraged to promote highly reliable practices in medication administration. Mackenzie Health is the first hospital in Canada to implement bidirectional interoperability.
Bidirectional infusion pump interoperability leverages barcode scanning and wi-fi to seamlessly transfer medication order parameters from the EHR directly into the infusion pump, eliminating the need for manual programming. Additionally, the system automatically transmits administered volume data from the pump back to the EHR. Bidirectional interoperability offers several benefits including, reduced pump programming errors, accurate and timely documentation and streamlined clinical workflows to promote highly reliable practices.
Strategic collaboration across multiple organizational tiers—including clinical informatics, operations, and pharmacy—formed the foundation for successful project execution. Through targeted process improvement workshops, the team mapped workflows and conducted comprehensive risk assessments. The initiative’s core workstreams centered on standardizing DERS data set parameters, preparing device infrastructure, and optimizing clinical workflow integration. A robust training and go-live support model ensured sustainability of this year-long implementation.
Success of this transformational change was driven by strong governance with key stakeholders, early engagement from clinical teams and focus on sustainability. Challenges included variability between EHR medication builds and medication administration practices. Over 95% of all infusions are administered using DERS. Staff compliance with leveraging interoperability when enabled, has been maintained at 85% since implementation. Interoperability has demonstrated a reduction in infusion programming incidents and the risk of alerts warning of severe harm incidents.
Bidirectional infusion pump interoperability enhances medication safety in hospitals. Other organizations may adopt these implementation learnings.
Sih K1,2, Lee A1, Wong T3
1Pharmacy Department, Children’s and Women’s Hospital of BC, Vancouver, BC
2Division of Emergency Medicine-Department of Pediatrics, BC Children’s Hospital, Vancouver, BC
3Division of Allergy-Department of Pediatrics, BC Children’s Hospital, Vancouver, BC
There is a discrepancy between the rates of perceived penicillin allergies and true allergies. The majority of suspected allergies can be de-labelled after proper assessment. Having a penicillin allergy label often results in being prescribed second line antibiotics which are less effective, have more side effects and cost more. A decision-support allergy assessment tool was created by pediatric allergists. The pediatric emergency department (ED) is an ideal location for allergy assessment due to its accessibility, multiple points of patient contact and antibiotic prescribing practices.
The aim was to increase the proportion of patients in the pediatric ED with a reported penicillin allergy that receive an allergy risk assessment to 80% within 1 year.
Pre-survey was circulated to emergency physicians to elucidate current practices in identifying and managing penicillin allergies. Change ideas were generated resulting in a new workflow to have pediatric emergency clinical pharmacist initiate penicillin allergy assessments. During a plan, do, study, act (PDSA) cycle it was noted that many patients were presenting to ED outside of clinical pharmacy hours therefore nurses were trained to use tool for assessment. Patient/caregiver educational handouts were also created.
Pre-survey results showed 0% of ED physicians were assessing penicillin allergies. Following implementation of clinical pharmacist workflows, risk assessments increased to 30%. After nurse education and application, assessments increased to 40%. Overall, 108 patients were assessed over 1 year. Over 38 patients were successfully de-labelled in ED which took an average of 7 minutes per patient.
Integrating a penicillin allergy assessment into ED patient assessment was easy and time appropriate. Not achieving aim was presumed to be due to ED acuity, pharmacist coverage and nurse turnover. Sustainability of workflows have been maintained. Patients were able to be de-label from penicillin allergies during their ED visit.
Ramesh S1, Carter A1,2
1Toronto General Hospital, Toronto, ON
2Leslie Dan Faculty of Pharmacy, Toronto, ON
The outpatient cardiology pharmacist at Toronto General Hospital (TGH) has a hybrid role, covering outpatient clinics and inpatient cardiology units (0.5:0.5 FTE). With over 100 specialty cardiac clinics at TGH and limited pharmacy resources, there was an opportunity to streamline services.
A novel workflow for the clinic pharmacist was developed, with a primary focus on new heart function clinic (HFC) patients referred for medication optimization/titration. A secondary focus was scoped referrals from other cardiac clinics.
Providers were engaged to identify which patient populations would benefit the most from pharmacist review. The HFC was prioritized due to complex medication regimens with a high propensity for drug-therapy problems (DTPs) in this population. After engaging with other HFC clinic pharmacists, we identified that most pharmacists prioritize new patients and attempt to collect a Best Possible Medication History (BPMH) prior to physician assessment. Using these learnings, a workflow was developed where the pharmacist proactively phones patients 1–2 weeks prior to their first HFC visit. A BPMH is collected as well as an assessment of heart failure symptoms, safety concerns and medication adherence. Recommendations are documented and communicated to the provider prior to the patient’s appointment. An additional focus was a consult service based on specific referral criteria.
From July 2023 to June 2024, 181 HFC patients were reviewed by the pharmacist. The average number of home medications was 10.1 (0–26), and 1.4 DTPs (0–6) identified per patient. Upon reviewing documentation after the clinic visit, pharmacist-identified DTPs were addressed or resolved in 55% of cases. The pharmacist also received 39 consults from other cardiac clinics, with drug information and adverse drug reaction management as most common referral types.
This new workflow has optimized use of pharmacist resources in our cardiac clinics and has received positive feedback from clinic providers.
Pedneault V1,2, Lebel A1, Anténor A-F1, Labrie LE1,2, Courchesne M1,2, Bernier L1,2, Homsi-Zaidan A1,2, Bergeron M3, Tanguay C1, Bussières J-F1,2
1Unité de Recherche en Pratique Pharmaceutique, Département de pharmacie, Centre hospitalier universitaire Sainte-Justine, Montréal, QC
2Faculté de pharmacie, Université de Montréal, Montréal, QC
3Centre intégré de santé et de services sociaux des Laurentides, St-Jérôme, QC
Historical dictionaries define terms and contextualize them within the evolution of a profession. There is currently no historical dictionary for pharmacy in Canada. This project aims to fill that gap by creating a comprehensive resource that highlights the development and contributions of pharmacy practice in Canada.
Our team developed the Grand dictionnaire canadien de l’histoire de la pharmacie. A documentary review and interviews with specialists were conducted to support the development of procedures. The dictionary aims to provide detailed definitions and historical context for terms related to pharmacy practice.
A standardized operating procedure was developed to guide the selection of terms, documentary research, writing, and publication. A collaborative approach was adopted, allowing multiple participants to contribute simultaneously. To date, 15 people have contributed, and 200 terms have been researched. The use of ChatGPT was explored to accelerate and standardize writing based on documentary research. A pilot website was developed, featuring an intuitive interface for easy navigation and access to information.
The Grand dictionnaire canadien de l’histoire de la pharmacie (http://gdchp.ca) was launched on November 25, 2024. It features an alphabetical index and six tabs to describe the project, the team, the method, and related work. Documentary work collaboration is based on the Teams platform. At launch, the dictionary contained 50 historical entries, three of which included an interview with a witness of the term’s use. Each entry includes a definition, historical elements, references, and the dates and contributors. Development is based on a continuous improvement process to optimize research and publication. The addition of a bilingual interface is planned.
This first dictionary offers a unique opportunity to highlight the Canadian context and the evolution of pharmacy practice. It was designed to facilitate collaboration and frequent updates. The procedures ensures consistency and efficiency in content creation.
Lee M1, Liu E1, Srimurugathasan N1, Chan D1
1North York General Hospital, North York, ON
In 2022, North York General Hospital (NYGH) created the NYGH Environmental Sustainability Committee to spearhead environmentally focused initiatives. To align with this project, the NYGH pharmacy department established a green initiatives workgroup.
A workgroup consisting of pharmacy management, pharmacists, pharmacy technicians and administrative staff met regularly to brainstorm, develop and implement environmental stewardship strategies in the inpatient and outpatient pharmacies at the hospital. A liaison ensured coordination with the corporate committee.
Initiatives were generated by gathering ideas from pharmacy staff as well as from themes originating from the corporate committee and organizations such as Choosing Wisely. Feasibility and impact assessment guided implementation, with metrics collected where possible.
Green strategies were focused on four key areas: medications, paper waste, recycling, and energy savings. Removing desflurane from the formulary and selecting environmentally-friendly metered dose inhalers were examples of reducing carbon footprint through medication selection. Transitioning from paper to electronic format of reports and medication requests saved over 40,000 sheets of paper annually. Additional measures, such as cancelling unnecessary faxes and magazines, repurposing used paper, promoting double-sided printing, and using scan-to-email features, further minimized paper use. Customized recycling signage developed with waste management companies improved compliance with waste and recycle sorting protocols. Reminder labels added to selected electronic devices and lights encouraged energy conservation after hours. Barriers to implementing additional ideas included limited coordination with other hospital departments, insufficient resources, factors beyond the department’s control such as packaging design of pharmaceuticals, and a strict commitment to maintaining safety standards.
It is essential for hospital pharmacies to actively prioritize environmental sustainability as part of their operations, adopt practices that minimize waste, and explore sustainable alternatives in pharmaceutical processes and waste management in an effort to contribute to preserve the planet for future generations.
Soubolsky A1, Albers L2, Koller-Klimosko K1
1Saskatchewan Health Authority, Saskatoon, SK
2Saskatchewan Health Authority, Regina, SK
Patients in Saskatoon living with mental health conditions experience long wait times for psychiatric care and face medication therapy management challenges. Pharmacists have a well-established and valuable role in optimizing medication therapy for patients with mental health conditions and enhancing healthcare system capacity. In Canada, the integration of a pharmacist on an outpatient psychiatric referral management team is a novel practice model requiring continual program evaluation to demonstrate value and determine how to most effectively impact patient care.
Patients are referred to psychiatry through a pooled referral system. Psychiatric nurses on the referral management team assess and direct patients to appropriate care pathways.
To further support access to care, targeted funding added a clinical pharmacist specialist to the referral management team to develop and provide a consultative service. The pharmacist performs medication assessments for patients who have been referred to psychiatry and provides medication therapy recommendations to referring practitioners.
Data is prospectively collected to describe the pharmacist consultations, medication therapy management recommendations, and subsequent referral outcomes. An evaluation of the first year of service implementation reports a total of 147 consults completed for referring primary care providers and 300 medication therapy recommendations. The most common recommendations were dose change (39%), start medication (21%), and discontinue medication (21%). Upon completion of the medication assessment, the psychiatry referral was inactivated by the referral management team for 37% (54/147) of patients after only seeing the pharmacist, and an additional 40% (59/147) were transitioned from the waitlist into the care of a psychiatrist.
Service evaluation demonstrates a pharmacist clinical specialist can improve access to care by addressing medication therapy management needs of mental health outpatients. Results also reveal opportunities and generate ideas for increased utilization of pharmacists and developing similar consultative models in mental health care.
Randhawa A1,2, Roy C2
1College of Pharmacy and Nutrition, University of Saskatchewan, Saskatoon, SK
2Department of Pharmacy Services, Saskatchewan Health Authority, Regina, SK
Single-use plastics are widely used for administering intravenous (IV) drugs. This study examines the environmental impact of administering cefazolin via ready-to-use IV bags versus syringes prepared from bulk powder bags and glass vials.
In response to a request to review the environmental impact of cefazolin commercially manufactured IV bags versus syringes prepared onsite by pharmacy departments, this study qualitatively compares options to determine which is more environmentally sustainable.
Information about current practices for preparing IV doses was sourced from hospital pharmacy technicians. This was assessed in the context of life-cycle-analyses literature of the drug, packaging, transportation, and other considerations
The primary sources of emissions differ between ready-to-use IV bags and syringes. For IV bags, emissions mainly come from distribution and cold chain storage requirements through transportation and on-site storage, which is energy-intensive and potentially carbon-intensive depending on jurisdiction. In contrast, syringe doses’ most significant emissions are from reconstitution inside laminar hoods. Syringes can be prepared efficiently when drawn from bulk bags (40 seconds per syringe). However, syringes drawn from glass vials require much longer reconstitution times (15 minutes per vial), contributing to additional laminar hood time.
Syringes from bulk powder bags are more environmentally sustainable than ready-to-use IV bags, as they avoid energy-intensive cold chain storage. They also benefit from more efficient reconstitution processes compared to doses from glass vials. Efforts towards promoting environmental sustainability in healthcare are expected to improve patient health outcomes. The findings are specific to local practice, but they are applicable to similarly equipped facilities and offer insights for smaller healthcare settings. Environmental impact is only one factor in product selection; other considerations include costs, skilled personnel availability, and NAPRA compliance requirements.
Fontaine J1, Robitaille C1, Abittan G1, Cohen E1, Boudjemai R1
1Pharmacy Department, CIUSSS Centre-Ouest-de-l’Île-de-Montréal, Montréal, QC
Within the Pharmacy Department of the CIUSSS Centre-Ouest-de-l’Île-de-Montréal, an assistant chief pharmacist and a clinical pharmacist review and analyze medication-related incidents at the Jewish General Hospital (JGH), a 600-bed acute tertiary care hospital center, according to incidents reported on provincial AH-223 forms tagged to the Pharmacy Department, as well as direct reports. This analysis is presented to the JGH pharmacists 2 to 3 times per year, as part of the Pharmacy Morbidity & Mortality rounds. Interventions to help prevent medication-related incidents are discussed, and implemented.
We describe the medication-related incidents presented at Pharmacy Morbidity and Mortality Rounds between 2021 and 2024, as well as the interventions implemented in response to these incidents to improve safety of care.
A descriptive analysis of medication-related incident reports and of interventions implemented to prevent future incidents was carried out. The following variables were collected: number of reports, severity classification, contributing factors, and type of actions taken.
A total of 151 reports were analyzed. Preliminary results show that the majority of reported incidents were of severity A or B on a scale adapted from The National Coordinating Council for Medication Error Reporting and Prevention. Frequent contributing factors were prescribing or order entry, validation, and stocking or dispensing errors. The type of actions taken most often were staff or patient education, and software integration or equipment management. Examples of actions taken are implementing an electronic medication reconciliation procedure, implementing double verification of drug protocol programming in our electronic prescribing tool, and reviewing barcodes for scanning in automated cabinets.
Descriptive analysis of incident reports can highlight trends in contributing factors, and the types of interventions implemented, which can better inform future decision-making when reviewing pharmacy-related processes and operations.
Newman P1, Dhaliwall S1, Polyakova O1, McDonald K1
1Northwest Telepharmacy Solutions, The Northwest Company, Winnipeg, MB
Medication incidents are the leading cause of injury and avoidable harm in health care systems. Up to 70% of medication errors are prescribing errors, occurring in 50% of hospital admissions. International, North American regulatory bodies, standards and associations endorse pharmacist review of all prescriptions before administration of the first dose. Despite this, most Canadian hospitals do not have 24/7 prospective pharmacist medication order review. Research on the documentation and analysis of prescribing errors, to support continuous quality improvement and pharmacist after-hours resources is lacking in Canada.
Despite long-standing implementation of computer prescriber order entry, prescribing errors remain prevalent. Causation and effective interventions to prevent prescribing errors remains understudied. Due to the prevalence of errors, and the time required to report, medication incident reporting systems within health care facilities and external platforms are underused.
Literature was reviewed to identify studies that observed pharmacist interventions while performing prospective medication order review in health care facilities. A search for documentation tools for good catches was also explored. Microsoft Office Forms, a cloud-based documentation tool was created to permit pharmacists to efficiently document good catches during order verification. A cause-based classification system for analyzing good catches was tested for feasibility.
Pharmacist good catches documentation via a cloud-based form was successful. The drug-related problem classification system for classifying severe medication errors was deemed a feasible tool for categorizing and analyzing prescribing errors according to cause.
Pharmacists play a critical role during medication order verification in clinically evaluating patients to ensure appropriate medication use and prevent patient harm. Consistent documentation and classification of good catches via an on-line user-friendly platform enables creation of a comprehensive database, establish causation and to analyze, identify trends, areas of risk, system improvements, shared learning, continuous quality improvement and further substantiate 24/7 pharmacist medication order review.
Levine A1, Clark T1, Evans A1, Hemrajani Malkani R1, Kandage M1, Orr S1, Paterson C1, Reford L1, Hebert E2, Ududuc O2, Dao D2, Soper F1, Lam S2
1McGill University, Bieler School of Environment. Montréal, QC
2Jewish General Hospital, Pharmacy Department, Montréal, QC
Pharmacy waste contributes to 25% of healthcare’s greenhouse gas emissions. The Jewish General Hospital Pharmacy (JGHP) in Montréal, QC, established the Pharmacy Green Committee to improve sustainability.
The Committee invited McGill University students to identify opportunities to reduce the environmental impact of JGHP operations. We evaluated the current state of sustainability at the JGHP and implemented interventions for improvement.
Intervention points within the pharmacy lifecycle were identified through a literature review, a waste audit of the JGHP, meetings with pharmacy and housekeeping personnel, and consultations with expert sustainable health organizations (e.g., CASCADES). We facilitated collaboration across JGH departments, ensuring support for interventions within the hospital. The initiatives considered patient safety, pharmacy operations, and departmental budget. Students made recommendations and spearheaded the implementation of temporally feasible interventions. We divided interventions into categories: short (Fall 2024), medium (Winter 2025), and long-term (Spring 2025+). In the short term, we developed clearer waste signage, introduced clips to replace single-use plastic bags for medication dispensing and transport, and delivered a staff training program on recycling and sustainable prescribing. Medium and long-term interventions will improve sustainable prescribing practices at JGH.
We sent a post-training feedback survey to participants and analyzed the results. We will continue gathering feedback on the signage developed. A second waste audit will quantitatively assess changes in waste sorting practices following the updated signage and training program.
This project demonstrates 1) Hospital pharmacies can significantly reduce plastic and render pharmacy operations more sustainable without affecting patient safety, and 2) the value of involving non-healthcare personnel as key facilitators in improving sustainable healthcare delivery. Due to students being in an unpaid research course and the interventions requiring limited purchases, the project demanded minimal financial investment, highlighting the viability of incorporating sustainable practices irrespective of a hospital’s budget constraints.
Wang D1, Hanna C1, Man D1, Kay S2
1Global Clinical Supply, Pfizer, Toronto, ON
2Global Clinical Supply, Pfizer, Andover, MA, USA
Ontario is the leading province in conducting clinical trials and ranked first in active clinical trials per capita among G7 nations. As drug development continues to grow in complexity and scope, it is critical for study sponsors and trial sites to work collaboratively to enhance investigational product (IP) handling experience.
The Clinical Research Pharmacist (CRP) is a specialty pharmacist role that was implemented in Canada in 2023 under Pfizer’s Global Clinical Supply (GCS) organization. CRPs are pharmacists with extensive clinical and/or clinical trials background. Their primary role is to provide pharmacy expertise in the development and management of IP while offering a pharmacy voice to connect scientists and clinical trial site personnel globally.
CRPs perform the following key responsibilities:
Canadian CRPs have successfully integrated within Pfizer’s GCS organization. They collaborate with clinical site personnel globally establishing themselves as subject matter experts in clinical pharmacy and clinical trials. Their presence in Canada strengthens local study site connections and provides clinical sites a platform to exchange IP related insights.
Canadian CRPs have demonstrated their role to be beneficial. Future directions include:
Chan D1
1North York General Hospital, Toronto, ON
Clinical decision supports (CDS) in electronic medication order systems help identify important alerts for clinicians. However, CDS may cause alert fatigue which is the tendency for clinicians to ignore prompts presented by CDS due to excessiveness and/or perceived clinical unimportance. This may increase the risk of missing important alerts and decrease work efficiency.
At North York General Hospital, pharmacists managed over one third of all medication CDS alerts amounting to approximately 34 alerts per day per pharmacist and resulting in an override rate of over 95%. As such, we attempted to reduce pharmacists’ alert fatigue.
Utilizing a visual analytics dashboard, high frequency alerts were tabulated with type, volume and override rate. Review of this data with pharmacists resulted in the design of three filters for suppressing non-significant alerts: (i) drug-drug interactions or duplicate alerts in specific order sets, (ii) hyperkalemia-related alerts based on patient’s current potassium level, and (iii) ceftriaxone-intravenous calcium alerts in certain age groups.
Alerts were reduced by 11,896 per month when comparing 1 month prior to filter implementation to 5 months after implementation, which represents a 33.8% decrease. This translates to a reduction from 34.1 to 22.3 alerts per day per pharmacist. However, override rate was minimally reduced from 96.1% to 95.2%. No increase in medication incidents or adverse drug reactions (ADRs) were reported after the changes. Review of potentially relevant medication incidents and all ADRs did not find any relation to the implemented filters.
The use of clinically contextualized CDS filters can be an effective and safe strategy to reduce non-meaningful alerts. Further investigation is required to determine why override rates were minimally impacted. Nevertheless, it is imperative that hospital pharmacies review alert data and reassess CDS settings and filters periodically to manage non-clinically significant alerts.
Chan S1
1Oak Valley Health, Markham, ON
Antimicrobial stewardship in surgical units is essential for preventing infections, controlling antimicrobial resistance, and optimizing patient care. Clinical rounds is one strategy used by antimicrobial stewardship programs (ASP) that has been shown to improve antimicrobial use. However, at Oak Valley Health, there is currently no formal ASP rounds implemented on the inpatient surgical units.
The ASP team designed and implemented a once weekly interdisciplinary rounds on the surgical units to review inpatient antimicrobial use. The rounds involved the infectious disease physician, pharmacist, and surgical nurse practitioners.
Initial stakeholder engagement, including infectious disease specialists, pharmacy management, and surgical nurse practitioners was crucial in determining the feasibility of conducting rounds and assembling an interdisciplinary team. The structure of rounds and how interventions would be communicated to the surgeon who is the most responsible provider (MRP) were then discussed. An intervention is defined as any antimicrobial related recommendation to optimize therapy (e.g. discontinuing unnecessary antibiotic, de-escalating antibiotic based on a review of a patient’s cultures and sensitivities etc.). Finally, any interventions made as a result of rounds were then documented in the patient’s electronic medical record.
A total of 39 ASP interventions were made after implementation of surgery rounds between March to June 2024, compared to 20 ASP interventions documented prior between July 2023 to February 2024. The most common types of ASP interventions made during rounds included intravenous to oral antimicrobial conversions, discontinuing antibiotics, and optimizing antimicrobial duration. A large majority (88%) of the recommendations made from rounds were accepted by the MRP.
Implementing antimicrobial stewardship rounds in the surgical units were sustainable and effective at optimizing antimicrobial appropriateness on these wards. There was positive feedback from the interdisciplinary round team in terms of building rapport with ASP and improved learning opportunities.
Balasubramaniam S1, Harris V1,2, Fleischer E2,3, St.Pierre K3, Mullowney T2,3, Price A2,3
1Department of Pharmacy Services, London Health Sciences Centre, London, ON
2Schulich School of Medicine & Dentistry, Western University, London, ON
3Children’s Hospital at London Health Sciences Centre, London, ON
Cystic fibrosis is characterized by mutations in the cystic fibrosis transmembrane conductance regulator gene, with the most common mutation in Canada being a deletion of the phenylalanine residue at position 508 (F508del). Elexacaftor/tezacaftor/ivacaftor (ETI) was recently approved for patients who are 2 years of age or older with certain mutations, including those with at least one copy of F508del.
A 2-year-old female with cystic fibrosis, homozygous for F508del, was started on ETI granules. After a month, transaminases were >5 times the upper limit of normal. ETI was stopped and transaminases normalized in one month. A gradual titration schedule, based on a desensitization protocol for adult patients who developed a rash on ETI, was created. The patient started on a reduced dose, with a plan to titrate up every 8 days with blood work scheduled every 2 weeks. The patient had further episodes of increased transaminases with the second occurring during a concurrent illness leading to changes in the titration schedule. However, she derived clinical benefit even with a sub-therapeutic dose of ETI.
Elevated transaminases are reported secondary to ETI. The Naranjo score of 8 indicates a probable adverse drug reaction. Transaminases decreased when ETI was held. Illness could have contributed to the second increase in liver enzymes.
Elevations in transaminases in studies resulted in no change in therapy, interruption, dose modification, or discontinuation of ETI. No specific details regarding dose modification were identified.
ETI has demonstrated benefits for patients with cystic fibrosis. There is no clear guidance on dose modification in response to elevated transaminases. This case illustrates titration of ETI and benefit from therapy in a patient who was unable to tolerate the full dose due to elevated transaminases.
Kim S1, Harris V1,2, Haylor J3, Babalola F2,3
1Department of Pharmacy, London Health Sciences Centre (LHSC), London, ON
2Schulich School of Medicine and Dentistry, Western University, London, ON
3Department of Pediatrics, Children’s Hospital, LHSC, London, ON
Central Diabetes Insipidus (CDI) is characterized by the inability to concentrate urine due to vasopressin deficiency. Treatment consists of increased fluid intake and desmopressin (DDAVP), a synthetic vasopressin, which carries a heightened risk of water intoxication and hyponatremia.
These three case reports describe the use of DDAVP oral suspension in neonates with CDI:
Case 1: An 8-day-old initiated on DDAVP 15 mcg oral suspension, then 10 mcg once daily given when urine output exceeded 4 mL/kg/hr, resulting in improved urine output and serum sodium.
Case 2: A 9-day-old started on DDAVP 1 mcg daily, titrated to DDAVP 0.5 mcg BID. Additional DDAVP 0.2 mcg oral was given for breakthrough polyuria.
Case 3: A 13-day-old started on DDAVP 0.5 mcg oral suspension, titrated to 1 mcg every 8 hours, with effective reduction in serum sodium. This patient lived where compounded DDAVP liquid would be difficult to obtain, thus was switched to DDAVP 0.2 mcg subcutaneously BID. Due to burden of subcutaneous dosing, transitioned to 20 mcg oral BID using 60 mcg DDAVP melts dissolved in 6 mL water and titrated to a final dose of 30 mcg oral BID.
A clear dose-response was demonstrated through monitoring urine output and serum sodium during dose titration of DDAVP.
Other case reports highlight successful management of CDI in 2 infants who achieved stable sodium concentrations using desmopressin lyophilisate melts. Another case described dissolving DDAVP melts in 10 mL water, with dose titration based on urine output, weight, and serum sodium.
With the removal of DDAVP rhinyl nasal solution from the market, alternative dosage forms are required. A compounded DDAVP suspension using tablets or melts can be used successfully in neonates, with dose titration to achieve the desired urine output and serum sodium.
Wang M1, Takahashi V1, Harris V1, Halabi M2
1Department of Pharmacy, London Health Sciences Centre, London, ON
2Department of Pediatric Emergency Medicine, London Health Sciences Centre, London, ON
Methemoglobinemia is a rare condition that reduces hemoglobin’s oxygen-carrying capacity. Neonates are especially vulnerable due to underdeveloped enzymatic pathways. Prilocaine can act as a trigger for this condition. Neonates can also have congenital methemoglobinemia, genetic disorders that predispose these neonates to methemoglobinemia.
A 5-day-old term male neonate underwent a frenotomy for tongue tie at a dental clinic, receiving both topical and injected prilocaine. Approximately two hours post-procedure, he developed pallor, lethargy, and progressive central cyanosis that was unresponsive to oxygen therapy. Pulse oximetry readings ranged from the high 80s to low 90s. Upon arrival at the emergency department, he was centrally cyanotic with an oxygen saturation of 91%. Arterial blood gas analysis revealed a critically elevated methemoglobin level of 30%. He was also evaluated for inherited causes of methemoglobinemia, but the results were negative. Intravenous methylene blue (2 mg/kg) was administered. Although the patient initially experienced further desaturation (an expected effect), he subsequently demonstrated rapid clinical improvement and normalization of methemoglobin levels.
Based on the Naranjo Adverse Drug Reaction Probability Scale (score of 5), prilocaine was deemed the probable cause of methemoglobinemia. The close temporal relationship between prilocaine administration and symptom onset, absence of other potential oxidizing agents, and positive response to methylene blue support this causality.
Many published cases highlight the effectiveness of intravenous methylene blue (1–2 mg/kg) in reversing cyanosis and restoring oxygen saturation in neonates. Alternatively, intravenous ascorbic acid has been used in resource-constrained settings. Neonates are particularly susceptible to prilocaine-induced methemoglobinemia due to immature enzymatic pathways, which elevate methemoglobin levels and cause cyanosis.
Practitioners should use prilocaine with caution, particularly in neonates, due to the risk of methemoglobinemia. Early identification and treatment are critical to prevent severe hypoxia and reduce mortality.
Lee R1, Harris V1, Sudbury B1, Tole S2
1Department of Pharmacy, London Health Sciences Centre, London, ON
2Department of Pediatrics, Division of Hematology/Oncology, Children’s Hospital at London Health Sciences Centre, London, ON
Children with congenital heart disease and increased risk of thrombosis often require secondary thromboprophylaxis with anticoagulation. Recently, rivaroxaban has been approved for this indication. While routine monitoring is not typically recommended, periodic monitoring of rivaroxaban concentrations may help identify dosing issues.
An 8-year-old male with hypoplastic left heart syndrome post Fontan surgery, plastic bronchitis, and protein-losing enteropathy (PLE) was on warfarin for primary thromboprophylaxis. He was transitioned to a therapeutic dose of rivaroxaban liquid (5 mg BID) due to labile INR values, frequent admissions, and challenges with enoxaparin subcutaneous administration. Due to concerns of malabsorption secondary to PLE, rivaroxaban serum concentrations were obtained, revealing an undetectable trough (< 25 ng/mL) and a subtherapeutic peak level (36 ng/mL). Investigation revealed a dosing error cause by a smaller pack size and syringe (1 mL) supplied by the manufacturer, leading to a 10-fold underdose (0.5 mg instead of 5 mg). Counselling was provided by the pharmacist to caregivers to ensure accurate rivaroxaban doses were administered. Subsequent serum concentrations drawn after 2-week and 4-weeks confirmed therapeutic concentrations of rivaroxaban.
An accidental underdose was deemed the probable cause of the subtherapeutic anticoagulation and confirmed by normalization of rivaroxaban serum concentrations following correct administration.
This appears to be the first reported instance of a dosing error attributed to packaging changes in pediatric rivaroxaban administration. The EINSTEIN-JR trial established rivaroxaban as a safe, and effective option for pediatric patients requiring thromboprophylaxis.
This case study highlights the critical role of pharmacists in identifying and addressing dosing errors to prevent a potentially life-threatening complication. Incorporating pharmacist counselling as a standard of care in pediatric direct oral anticoagulant initiation can improve efficacy, safety and reduce healthcare costs.
Heitzner J1, Martin S1
1Pharmacy Department, London Health Sciences Centre, London, ON
Alteplase is a treatment option for prosthetic valve thrombosis (PVT) instead of surgery in high-risk patients. There is limited evidence supporting a particular regimen for alteplase administration in this setting. Ultraslow infusion low-dose (USILD) alteplase was given to an elderly female with a PVT of the aortic valve (AV).
An 80-year-old female with rheumatic heart disease requiring St. Jude mechanical aortic and mitral valve replacements (2008), presented with class IV heart failure. Echocardiography revealed severe AV stenosis and moderately severe aortic insufficiency. Fluoroscopy confirmed markedly reduced bi-leaflet movement. Home medications include ASA and warfarin with therapeutic INR (2.5–3.0). Upon review by a cardiovascular surgeon, medical management was recommended. Alteplase was initiated at 25 mg IV over 25 hours and heparin was held. Subjective patient improvement occurred, with reduced AV mean gradient and trace insufficiency. Alteplase was well-tolerated with a mild headache and no hemorrhage on CT. After alteplase treatment, heparin was resumed, ASA 81 mg continued, with bridging to warfarin (target INR 3.0–3.5) and patient was discharged. Patient was readmitted 23 days later with worsening AV parameters and new reduced left ventricular function. Repeat fluoroscopy was unchanged, and the patient expired from cardiogenic shock secondary to mechanical AV thrombosis.
Alteplase reduced the AV gradient and improved symptoms, but readmission suggested unsustained benefit. Pannus formation may have been predominant, favoring surgery over thrombolysis.
Thrombolytic therapy for PVT is well described, but data on USILD alteplase, especially in elderly patients, is sparse. Evidence is largely non-randomized and lacks representation of class IV heart failure presentations.
This case highlights the safety and potential efficacy of a novel alteplase regimen for PVT in an elderly non-surgical candidate. It underscores the need for early and frequent post-thrombolysis monitoring to assess for sustained benefit.
Len S1, Elder C1, O’Byrne S1, Rieder Underwood M1, Whittemore K2
1Department of Pharmacy – Critical Care, Saskatchewan Health Authority, Regina, SK
2Department of Critical Care, Saskatchewan Health Authority, Regina, SK
Valproic acid (VPA) is a broad spectrum antiepileptic agent with off-label uses. In acute toxicity, activated charcoal, levocarnitine, and intermittent hemodialysis (IHD) are appropriate treatment. This case report describes adjunctive use of meropenem in the management of VPA toxicity.
A 35 year old, 58 kg male presented to acute care following a massive ingestion of VPA (88 grams). The patient’s initial VPA level was 3,518 μmol/L. Four hours later, his level was 11,425 μmol/L and he required intubation. Activated charcoal and levocarnitine were given per protocol. The patient became hypotensive and Poison Control suggested IHD, requiring transfer to a tertiary intensive care unit (ICU). The patient’s inadequate response to treatment as well as the delayed initiation of IHD prompted the clinical team to initiate carbapenem therapy. Meropenem 1 gram intravenous every 8 hours and IHD were initiated on arrival to ICU. Dialysis completed after 6 hours; a post dialysis VPA level was 952 μmol/L. Meropenem continued for 24 hours. The patient was eligible for transfer out of the ICU on day 2.
The serum level of VPA was not within the normal therapeutic range after completion of IHD. The first VPA level within therapeutic range (599 μmol/L) was achieved after the third dose of meropenem. After 24 hours of meropenem therapy, the patient was successfully extubated to room air.
Carbapenems have been featured in multiple case reports as potential adjunctive treatment for VPA toxicity due to a drug interaction that rapidly reduces VPA serum levels.
Carbapenems may have an earlier role in VPA toxicity management, particularly when imminent hemodialysis is not feasible. Further research is needed to fully understand their role in therapy.
The texts of poster abstracts are published exactly as submitted by the authors and have not undergone any copyediting by the Canadian Journal of Hospital Pharmacy.
Table for Newman P, Dhaliwall S, Polyakova O, McDonald K. Canadian pharmacy key performance indicators: where are we with clinical services prioritization, utilization and documentation in hospitals supported by telepharmacists in Canada? [abstract]. Can J Hosp Pharm. 2025;78(3):e3850, page 4.

Table for Augustyniak M, Ariano R, Bonnar P, Dixon A, Sheth P, Péloquin S, et al. Where are pharmacists at with regards to their antimicrobial stewardship professional practice? A mixed-methods needs assessment in Canada [abstract]. Can J Hosp Pharm. 2025;78(3):e3850, page 13.
TABLE 1 Reported knowledge and skill gaps of pharmacists in hospitals (Pharm-H) versus retail (Pharm-R) settings
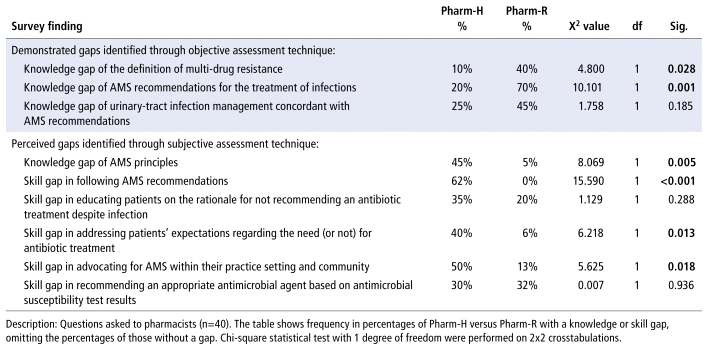
© 2025 Canadian Society of Healthcare-Systems Pharmacy | Société canadienne de pharmacie dans les réseaux de la santé
Canadian Journal of Hospital Pharmacy, VOLUME 78, NUMBER 3, 2025